Abstract
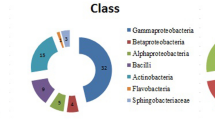
The rhizobacteria that promote the growth of plants can have a positive effect on the productivity of crops, especially in stress conditions. Among the plant -growth -promoting (PGP) rhizobacteria (PGPR) cluster, Bacillus spp. are among the genera with most potential due to their spore forming ability, thereby increasing the adaptation of Bacillus strains to commercial formulation and field application. Due to their intrinsic properties, the Bacilli have several mechanisms conferring beneficial effects on plants. Thirty-five strains of Bacillus isolated from the rhizosphere of wheat from three different soils in arid and semi-arid areas of Algeria were tested for properties involved in the promotion of plant growth. The PGP ability of the 35 strains was evaluated by determining their biofertilisation (phosphate solubilisation), biostimulation [indole acetic acid (IAA) production] and biocontrol [cyanhydric acid (HCN), siderophores, 2,3-butanediol production and antifungal activity] activities. Of the 35 strains, 78 % had the ability to solubilise phosphates at rates of 16.65 μg/mL for strain D13, 15.60 μg/mL for D7 and 15.05 μg/mL for D6. These strains were the most successful and were isolated from arid and alkaline soils. The highest concentrations of IAA were produced by strains D4 and D7 to values ranging from 10 to 19 μg/mL. All strains inhibited at least one fungal strain tested, and 75 % had activity against three fungi or more. More than half of Bacillus strains produced 2.3- butanediol but only a single strain produced HCN. Only three strains (B25, D11 and BA11) were efficient in the production of siderophores. Also, four strains (B21, D4, B10 and B25) possessed ACCdeaminase and were considered regulators of stress. Phylogenetic diversity of the strains was analysed by 16S rDNA sequencing. The results identified all strains as being similar to the Bacillus sp. cluster, and divided separately into five groups. The majority of strains (n = 28) were assigned to the species Bacillus thuringiensis and Bacillus subtilis. The Bacillus species isolated in this study showing PGP abilities have the potential to be used as PGPR.





Similar content being viewed by others
References
Ahemad M, Kibret M (2014) Mechanisms and applications of plant growth promoting rhizobacteria: current perspective. J King Saud Univ Sci 26:1–20
Amar JD, Kumar M, Kumar R (2013) Plant growth promoting rhizobacteria (PGPR): an alternative of chemical fertilizer for sustainable, environment friendly agriculture. Res J Agric For Sci 1(4):21–23
Ash CJ, Farrow AE, Wallbanks S, Collins MD (1991) Phylogenetic heterogeneity of the genus Bacillus revealed by comparative analysis of small subunit ribosomal RNA sequences. Lett Appl Microbiol 13:202–206. doi:10.1111/j.1472-765X.1991.tb00608.x
Barazani O, Friedman J (1999) Is IAA the major root growth factor secreted from plant-growth-mediating bacteria. J Chem Ecol 25:2397–2406. doi:10.1023/A:1020890311499
Cappuccino JC, Sherman N (1992) Microbiology: a laboratory manual, 3rd edn. Benjamin/Cummings, New York, pp 125–179
Castric PA (1977) Glycine metabolism by Pseudomonas aeruginosa: hydrogen cyanide biosynthesis. J Bacteriol 130:826–831, PMCID: PMC235287
Ding Y, Wang J, Liu Y, Chen S (2005) Isolation and identification of nitrogen-fixing bacilli from plant rhizospheres in Beijing region. J Appl Microbiol 99:1271–1281. doi:10.1111/j.1365-2672.2005.02738.x
Dworkin M, Foster J (1958) Experiments with some microorganisms which utilize ethane and hydrogen. J Bacteriol 75:592–601, PMCID: PMC290115
Edwards U, Rogall T, Blocker H, Emde M, Bottger EC (1989) Isolation and direct complete nucleotide determination of entire genes. Characterization of a gene coding for 16S ribosomal RNA. Nucleic Acids Res 17:7843–7853. doi:10.1093/nar/17.19.7843
Egamberdiyera D (2007) The effect of plant growth and nutrient uptake of maize in two different Soils. Appl Soil Ecol 36:184–189. doi:10.1016/j.apsoil.2007.02.005
Erturk Y, Ercisli S, Haznedar A, Cakmakci R (2010) Effects of Plant Growth Promoting Rhizobacteria (PGPR) on rooting and root growth of kiwifruit (Actinidia deliciosa) stem cuttings. Biol Res 42:91–98. doi:10.4067/S0716-97602010000100011
Gamalero E, Berta G, Massa N, Glick BR, Lingua G (2010) Interactions between Pseudomonas putida UW4 and Gigaspora rosea BEG9 and their consequences on the growth of cucumber under salt stress conditions. J Appl Microbiol 108:236–45. doi:10.1111/j.1365-2672.2009.04414.x
Garbeva P, Van Veen JA, van Elsas JD (2003) Assessment of the diversity, and antagonism towards Rhizoctoniasolani AG3, of Pseudomonas species in soil from different agricultural regimes. FEMS Microbiol Ecol 47:51–64
Gaur AC (1990) Phosphate solubilizing microorganisms as biofertilizers, 1st edn. Omega Scientific, New Delhi, India. ISBN 81-85399-09-3
Glazebrook J, ChenW EB, Chang HS, Nawrath C, Metraux JP, Zhu T, Katagiri F (2003) Topology of the network integrating salicylate and jasmonate signal transduction derived from global expression phenotyping. Plant J 31:217–228, PMID:12694596
Glick BR, Jacobson CB, Schwarze MMK, Pasternak JJ (1994) 1-Aminocyclopropane-1- carboxylic acid deaminase mutants of the plant growth promoting rhizobacterium Pseudomonas putida GR12-2 do not stimulate canola root elongation. Can J Microbiol 40:911–915. doi:10.1139/m94-146
Goswami D, Dhandhukia P, Patel P, Thakker JN (2014) Screening of PGPR from saline desert of Kutch: growth promotion in Arachis hypogea by Bacillus licheniformis A2. Microbiol Res 169:66–75. doi:10.1016/j.micres.2013.07.004
Holl FB, Chanway SP, Turkington R, Radley RA (1988) Response of cresied wheatgrss (Agropyron cristatum L.) perennial ryegrass (Lolium perrenne) and white clover (Trifolium repens L.) to inoculation with Bacillus polymyxa. Soil Biol Biochem 20:19–24, http://dx.doi.org/ 10.1016/0038-0717(88)90121-6
Illmer P, Schinner F (1992) Solubilization of inorganic phosphates by microorganisms isolated from forest soil. Soil Biol Biochem 24:389–395, http://dx.doi.org/ 10.1016/0038-0717(92)90199-8
Jacobson CB, Pasternak JJ, Glick BR (1994) Partial purification and characterization of 1-aminocyclopropane-1-carboxylate deaminase from the plant growth promoting rhizobacterium Pseudomonas putida GR12-2. Can J Microbiol 40:1019–1025, http://dx.doi.org/ 10.1139/m94-162
Joseph B, Patra RR, Lawrence R (2007) Characterization of plant growth promoting rhizobacteria associated with chickpea (Cicer arietinum L.). Int J Plant Prod 2:141–152
Kloepper JW, Litshitz R, Zablotowicz RM (1989) Free living bacterial inocula for enhancing crop producitivity. Trends Biotechnol 7:39–43. doi:10.1016/0167-7799(89)90057-7
Kumar NR, Arasu VT, Gunasekaran P (2002) Genotyping of antifungal compounds producing plant growth-promoting rhizobacteria Pseudomonas fluorescens. Curr Sci 82:1463–1466
Loper JE, Schroth MN (1986) Influence of bacterial sources of indole-3-acetic acid on root elongation of sugar beet. Phys Chem Chem Phys 76:386–389, http://dx.doi.org/ 10.1094/Phyto-76-386
Lorck H (1948) Production of hydrocyanic acid by bacteria. Physiol Plant 142-146 doi: 10.1111/j.1399-3054.1948.tb07118.x
Maplestone PA, Campbell R (1989) Colonization of roots of wheat seedlings by bacilli proposed as biocontrol agents against take-all. Soil Biol Biochem 21(4):543–550. doi: 10.1016/0038-0717(89)90128-4
Nautiyal CS (1999) An efficient microbiological growth medium for screening phosphate solubilizing microorganisms. FEMS Microbiol Lett 170:260–270
Olsen SR, Sommers LE (1982) Phosphorus. In: Page AL, Miller RH, Keeney DR (eds) Methods of soil analysis, part 2, chemical and microbial properties, 2nd edn. American Society of Agronomy, Madison, pp 403–430
Qiao JQ, Wu HJ, Rong Huo R, Gao XW, Borriss R (2014). Stimulation of plant growth and biocontrol by Bacillus amyloliquefaciens subsp. plantarum FZB42 engineered for improved action. Chemical and Biological Technologies in Agriculture 1:12
Ramette A, Frapolli M, Defago G, Moenne-Loccoz Y (2003) Phylogeny of HCN synthase-encoding hcn BC genes in biocontrol fluorescent pseudomonads and its relationship with host plant species and HCN synthesis ability. Mol Plant-Microbe Interact 16:525–535. doi:10.1094/MPMI.2003.16.6.525
Ryu CM, Farag MA, Hu CH, Reddy MS, Wei HX, Paré PW, Kloepper JW (2003) Bacterial volatiles promote growth in Arabidopsis. Proc Natl Acad Sci USA 100:4927–4932. doi:10.1104/pp.103.026583
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425, PMID: 3447015
Shwyn B, Neilands JB (1987) Universal chemical assay for the detection and determination of siderophores. Annu Rev Plant Physiol Plant Mol Biol 160:47–56
Silini-Cherif H, Silini A, Ghoul M, Yadav S (2012) Isolation and characterization of plant growth promoting traits of a rhizobacteria: Pantoea agglomerans lma2. Pak J Biol Sci 15:267–276. doi:10.3923/pjbs.2012.267.276
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28(10):2731–2739. doi:10.1093/molbev/msr121
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 11(22):4673–4680, PMID: 7984417
Tian F, Ding Y, Zhu H, Yao L, Du B (2009) Genetic diversity of siderophore-producing bacteria of tobacco rhizosphere. Braz J Microbiol 40:276–284. doi:10.1590/S1517-83822009000200013
Vacheron J, Desbrosses G, Bouffaud M-L et al (2013) Plant growth-promoting rhizobacteria and root system functioning. Front Plant Sci 4:356. doi:10.3389/fpls.2013.00356
Valdebinito M, Crumbliss AL, Winkelman G, Hantke K (2006) Environmental factors influence the production of enterobactin, salmochelin, aerobactin and yersiniabactin in Escherichia coli strainNissle 1917. Int J Med Microbiol 296:513–520, PMID:17008127
Wilson K (1990) Preparation of genomic DNA from bacteria. In: Ausubel FM, Brent R (eds) Current protocols in molecular biology. Greene/Wiley Interscience, New York, pp 241–245
Yadav S, Kaushik R, Saxena AK, Arora DK (2011) Diversity and phylogeny of plant growth-promoting bacilli from moderately acidic soil. J Basic Microbiol 51(1):98–106. doi:10.1002/jobm.201000098
Young JPW, Downer HL, Eardly BD (1991) Phylogeny of the phototrophic Rhizobium strain BTAil by polymerase chain reaction-based sequencing of a 16S rRNA gene segment. J Bacteriol 173:2271–2277, PMCID: PMC207778
Zahir ZA, Arshad M, Frankenberger WT (2004) Plant growth promoting rhizobacteria: applications and perspectives in agriculture. Adv Agron 81:97–168. doi:10.1016/S0065-2113(03)81003-9
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cherif-Silini, H., Silini, A., Yahiaoui, B. et al. Phylogenetic and plant-growth-promoting characteristics of Bacillus isolated from the wheat rhizosphere. Ann Microbiol 66, 1087–1097 (2016). https://doi.org/10.1007/s13213-016-1194-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13213-016-1194-6