Abstract
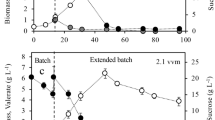
The aim of this study was to characterize the influence of the aeration conditions on the production of PHB and its molecular mass in a mutant strain of Azotobacter vinelandii (OPN), which carries a mutation on ptsN, the gene encoding enzyme IIANtr, previously shown to increase the accumulation of PHB. Cultures of A. vinelandii wild-type strain OP and its mutant derivative strain OPN were grown in 500-mL flasks, containing 100 and 200 mL of PY sucrose medium. PHB production and its molecular mass were analyzed at the end of the culture. The molecular mass (MM) was significantly influenced by the aeration conditions and strain used. A polymer with a higher molecular weight was produced under low aeration conditions for both strains. A maximal molecular mass of 2,026 kDa (equivalent to 3,670 kDa measured by GPC) was obtained with strain OPN cultured under low-aeration conditions, reaching a value two-fold higher than that obtained from the parental strain OP (MM = 1,013 kDa) grown under the same conditions. Aeration conditions and the ptsN mutation influence the molecular mass of the PHB produced by A. vinelandii affecting in turn its physico-chemical properties.



Similar content being viewed by others
References
Aldor I, Keasling J (2003) Process design for microbial plastic factories: metabolic engineering of polyhydroxyalkanoates. Curr Opin Biotechnol 14:475–483
Chen G, Page W (1994) Effect of substrate on the molecular weight of of poly-β-hydroxybutyrate produced by Azotobacter vinelandii UWD. Biotechnol Lett 16(2):155–160
Dominguez-Diaz M, Romo-Uribe A (2012) Viscoelastic behavior of biodegradable polyhydroxyalkanoates. Bioinspired Biomim Nanobiomaterials 1:214–220. doi:10.1557/opl.2011.555
Dominguez-Diaz M, Flores A, Cruz-Silva R, Romo-Uribe A (2011) Kinetics of crystallization of biodegradable PHA copolymers: a combined X-ray scattering and micro-indentation study. Mater Res Soc Symp Proc 1301
Galindo E, Peña C, Nuñez C, Segura D, Espín G (2007) Molecular and Bioengineering strategies to improve alginate and polydydroxyalkanoate production by Azotobacter vinelandii. Microbiol Cell Fact 6(7):1–16
Hahn KS, Chang KY, Kim SB, Chang NH (1994) Optimization of microbial Poly(3-hydroxybutyrate) recovery using dispersions of sodium hypochlorite solution and chloroform. Biotechnol Eng 44:256–261
Hernandez-Eligio A, Moreno S, Castellanos M, Castañeda M, Nuñez C, Muriel-Millan L, Espín G (2012) RsmA post-transcriptionally controls PhbR expression and polyhydroxybutyrate biosynthesis in Azotobacter vinelandii. Microbiology 158:1953–1963
Iwata T (2005) Strong fibers and films of microbial polyesters. Macromol Biosci 5(8):689–701
Karr D, Waters J, Emerich D (1983) Analysis of poly-β-hydroxybutyrate in Rhizobium japonicum bacteroids by ion-exclusion high-pressure liquid chromatography and UV detection. Appl Environ Microbiol 46:1339–1344
Khanna S, Srivastava A (2005) Recent advances in microbial polyhydroxyalkanoates. Process Biochem 40:607–619
Klimek J, Ollis D (1980) Extracellular microbial polysaccharides: kinetics of Pseudomonas sp. Azotobacter vinelandii, and Aureobasidium pullulans batch fermentations. Biotechnol Bioeng 22:2321–2342
Lowry OH, Rosebrough NJ, Farr AL, Randall R (1951) Protein measurement with the folin phenol reagent. J Biol Chem 193:265–275
Madison L, Huisman G (1999) Metabolic engineering of poly(3-Hydroxyalkanoates): 16 from DNA to plastic. Microbiol Mol Biol Rev 63(1):21–53
Marchessault RH, Okamura K, Su CJ (1970) Physical propierties of poly(β-Hydroxybutyrate). Macromolecules 3(6):735–740
Miller G (1959) Use of dinitrosalicylic acid reagent for determination of reducting sugars. Anal Chem 31(3):426–428
Myshkina VL, Nikolaeva DA, Makhina TK, Bonartseva AP, Filatova EV, Ruzhitsky AO, Bonartseva GA (2008) Effect of growth conditions on the molecular weight of Poly-3-hydroxybutyrate Produced By Azotobacter chroococcum 7B. Appl Biochem Microbiol 44(5):482–486
Noguez R, Segura D, Moreno S, Hernandez A, Juarez K, Espin G (2008) Enzyme INtr, NPr and IIA Ntr are involved in regulation of the poly-β-hydroxybutyrate biosynthetic genes in Azotobacter vinelandii. J Mol Microbiol Biotechnol 15:244–254
Peña C, Campos N, Galindo E (1997) Changes in alginate molecular mass distributions, broth viscosity and morphology of Azotobacter vinelandii cultured in shake flasks. Appl Microbiol Biotechnol 48:510–515
Peña C, Galindo E, Peter C, Büchs J (2007) Evolution of the specific power consumption and oxygen transfer rate in alginate-producing cultures of Azotobacter vinelandii conducted in shake flasks. Biochem Eng J 36:73–80
Peña C, Galindo E, Peter C, Büchs J (2011) The viscosifying power, degree acetylation and molecular mass of the alginate produced by Azotobacter vinelandii in shake flasks are determined by the oxygen transfer rate. Process Biochem 46:290–297
Pflüger-Grau K, Görke B (2010) Regulatory roles of the bacterial nitrogen-related phosphotransferase system. Trends Microbiol 18:205–214
Quagliano JC, Miyazaki SS (1997) Effect of aeration and carbon/nitrogen ratio on the molecular mass of the biodegradable polymer poly(3-hydroxybutyrate) obtained from Azotobacter chroococcum 6B. Appl Microbiol Biotechnol 48:662–664
Rubinstein M, Colby RH (2003) Polymer physics. Oxford University Press, New York
Ryu WH, Cho KS, Goodrich PR, Park CH (2008) Production of polyhydroxyalkanoates by Azotobacter vinelandii UWD using swine wastewater: effect of supplements glucose, yeast extract, and inorganic salts. Biotechnol Bioeng 13:651–658
Segura D, Espín G (1998) Mutational inactivation of a gene homologous to Escherichia coli ptsP affects poly-beta-hydroxybutyrate accumulation and nitrogen fixation in Azotobacter vinelandii. J Bacteriol 180:4790–4798
Segura D, Espín G (2004) Inactivation of pycA, encoding pyruvate carboxylase activity, increases poly-beta-hydroxybutyrate accumulation in Azotobacter vinelandii on solid medium. Appl Microbiol Biotechnol 65:414–418
Segura D, Guzmán J, Espín G (2003) Azotobacter vinelandii mutants that overproduce poly-beta-hydroxybutyrate or alginate. Appl Microbiol Biotechnol 63:159–163
Segura D, Vite O, Romero Y, Moreno S, Castaneda M, Espin G (2009) Isolation and characterization of Azotobacter vinelandii mutants impaired in Alkylresorcinol Synthesis: alkylresorcinols are not essential for cysts desiccation resistance. J Bacteriol 191:3142–3148
Ward IM (1971) Mechanical properties of solid polymers. Wiley, New York
Williams S, Martin D (2005) Applications of polyhydroxyalkanoates (PHA) in medicine and pharmacy. Biopolymers Online. doi:10.1002/3527600035.bpol4004
Zhang YX, Xin JY, Song H, Chun-gu X (2008) Biosynthesis of poly-3-hydroxybutyrate with a high molecular weight by methanotroph from methane and methanol. J Nat Gas Chem 17:103–109
Acknowledgments
The authors gratefully acknowledge the technical help of Maraolina Dominguez-Diaz (LNyC, UNAM) and Modesto Millán (IBT, UNAM). This work was partially financed by DGAPA-UNAM (grants IT209411-3 and IN110310), and CONACyT under CIAM2008 program (grant 107294).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Peña, C., López, S., García, A. et al. Biosynthesis of poly-β-hydroxybutyrate (PHB) with a high molecular mass by a mutant strain of Azotobacter vinelandii (OPN). Ann Microbiol 64, 39–47 (2014). https://doi.org/10.1007/s13213-013-0630-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13213-013-0630-0