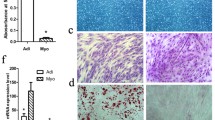
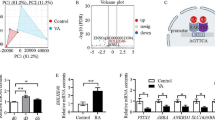
Abstract
The action of FTO on myoblasts proliferation and differentiation and molecular mechanism underlying it were investigated by transfecting with FTO lentiviral overexpression vector and gene expression profile sequencing. Compared with the control group, myoblasts with FTO transfection was significantly enhanced proliferation; the expression of MYOG and MYOD mRNA was significantly increased. In cells transfected with FTO, 129 differentially expressed genes were determined compared with control group, with 104 up-regulated and 25 down-regulated genes. Twelve pathways (Phagosome, Focal adhesion, Adrenergic signaling in cardiomyocytes, Endocytosis, Cardiac muscle contraction, Toll-like receptor, Ribosome, Tight junction, Regulation of actin cytoskeleton, Cytokine–cytokine receptor interaction, Adrenergic signaling in cardiomyocytes and MAPK) were significantly enriched. Eight genes known to be directly or indirectly related to skeletal muscle development (LAMA5, SPP1, CAV3, RASGRF1, FAK, PDGFB, PDGFRα, and RAC2) were enriched in the focal adhesion and expressed differentially. Altogether, these data suggested that FTO stimulated differentiation of myoblasts through regulation of eight genes enriched in the focal adhesion.






Similar content being viewed by others
References
Bisht B, Srinivasan K, Dey CS (2008) vivo inhibition of focal adhesion kinase causes insulin resistance. J Physiol 586:3825–3837. https://doi.org/10.1113/jphysiol.2008.157107
Block J, Bonilla L, Hansen PJ (2009) Effect of addition of hyaluronan to embryo culture medium on survival of bovine embryos in vitro following vitrification and establishment of pregnancy after transfer to recipients. Theriogenology 71:1063–1071. https://doi.org/10.1016/j.theriogenology.2008.11.007
Bloise FF, Cordeiro A, Ortiga-Carvalho TM (2018) Role of thyroid hormone in skeletal muscle physiology. J Endocrinol 236:R57–r68. https://doi.org/10.1530/joe-16-0611
Caruso V, Chen H, Morris MJ (2011) Early hypothalamic FTO overexpression in response to maternal obesity–potential contribution to postweaning hyperphagia. PLoS ONE 6:e25261. https://doi.org/10.1371/journal.pone.0025261
Cebrian-Serrano A, Salvador I, Silvestre MA (2014) Beneficial effect of two culture systems with small groups of embryos on the development and quality of in vitro-produced bovine embryos. Anat Histol Embryol 43:22–30. https://doi.org/10.1111/ahe.12043
Church C et al (2010) Overexpression of Fto leads to increased food intake and results in obesity. Nat Genet 42:1086–1092. https://doi.org/10.1038/ng.713
Dina C et al (2007) Variation in FTO contributes to childhood obesity and severe adult obesity. Nat Genet 39:724–726. https://doi.org/10.1038/ng2048
Frayling TM et al (2007) A common variant in the FTO gene is associated with body mass index and predisposes to childhood and adult obesity. Science (New York, NY) 316:889–894. https://doi.org/10.1126/science.1141634
Galbiati F, Razani B, Lisanti MP (2001) Emerging themes in lipid rafts and caveolae. Cell 106:403–411. https://doi.org/10.1016/s0092-8674(01)00472-x
Gao X, Shin YH, Li M, Wang F, Tong Q, Zhang P (2010) The fat mass and obesity associated gene FTO functions in the brain to regulate postnatal growth in mice. PLoS ONE 5:e14005. https://doi.org/10.1371/journal.pone.0014005
Ge L et al (2018) Deletion of exon 4 in LAMA2 is the most frequent mutation in Chinese patients with laminin alpha2-related muscular dystrophy. Sci Rep 8:14989. https://doi.org/10.1038/s41598-018-33098-3
Graham ZA, Gallagher PM, Cardozo CP (2015) Focal adhesion kinase and its role in skeletal muscle. J Muscle Res Cell Motil 36:305–315. https://doi.org/10.1007/s10974-015-9415-3
Guo F, Zhang Y, Zhang C, Wang S, Ni Y, Zhao R (2015) Fatmass and obesity associated (FTO) gene regulates gluconeogenesis in chicken embryo fibroblast cells. Comp Biochem Physiol A Mol Integr Physiol 179:149–156. https://doi.org/10.1016/j.cbpa.2014.10.003
Hashemi-Gorji F, Yassaee VR, Dashti P, Miryounesi M (2018) Novel LAMA2 gene mutations associated with merosin-deficient congenital muscular dystrophy. Iran Biomed J 22:408–414
Huang HY et al (2015) Brain natriuretic peptide stimulates lipid metabolism through its receptor NPR1 and the glycerolipid metabolism pathway in chicken adipocytes. Biochemistry 54:6622–6630. https://doi.org/10.1021/acs.biochem.5b00714
Ito T et al (2013) Imatinib attenuates severe mouse dystrophy and inhibits proliferation and fibrosis-marker expression in muscle mesenchymal progenitors. Neuromuscul Disord NMD 23:349–356. https://doi.org/10.1016/j.nmd.2012.10.025
Lassiter DG et al (2018) FAK tyrosine phosphorylation is regulated by AMPK and controls metabolism in human skeletal muscle. Diabetologia 61:424–432. https://doi.org/10.1007/s00125-017-4451-8
Luo W et al (2014) The transient expression of miR-203 and its inhibiting effects on skeletal muscle cell proliferation and differentiation. Cell Death Dis 5:e1347. https://doi.org/10.1038/cddis.2014.289
Mao X, Cai T, Olyarchuk JG, Wei L (2005) Automated genome annotation and pathway identification using the KEGG Orthology (KO) as a controlled vocabulary. Bioinformatics (Oxford, England) 21:3787–3793. https://doi.org/10.1093/bioinformatics/bti430
Nghiem PP et al (2017) Osteopontin is linked with AKT, FoxO1, and myostatin in skeletal muscle cells. Muscle Nerve 56:1119–1127. https://doi.org/10.1002/mus.25752
Olson LE, Soriano P (2009) Increased PDGFRalpha activation disrupts connective tissue development and drives systemic fibrosis. Dev Cell 16:303–313. https://doi.org/10.1016/j.devcel.2008.12.003
Orr-Urtreger A, Bedford MT, Do MS, Eisenbach L, Lonai P (1992) Developmental expression of the alpha receptor for platelet-derived growth factor, which is deleted in the embryonic lethal Patch mutation. Development (Cambridge, England) 115:289–303
Saeed-Zidane M et al (2019) Hyaluronic acid and epidermal growth factor improved the bovine embryo quality by regulating the DNA methylation and expression patterns of the focal adhesion pathway. PLoS ONE 14:e0223753. https://doi.org/10.1371/journal.pone.0223753
Scuteri A et al (2007) Genome-wide association scan shows genetic variants in the FTO gene are associated with obesity-related traits. PLoS Genet 3:e115. https://doi.org/10.1371/journal.pgen.0030115
Shah DS, Nisr RB, Stretton C, Krasteva-Christ G, Hundal HS (2020) Caveolin-3 deficiency associated with the dystrophy P104L mutation impairs skeletal muscle mitochondrial form and function. J Cachexia Sarcopenia Muscle. https://doi.org/10.1002/jcsm.12541
Song C, Song WT, Shu JT, Tao ZY, Zhu WQ, Di C, Li HF (2015) Tissue- and breed-specific expression of the chicken fat mass- and obesity-associated gene (FTO). Genet Mol Res GMR 14:10500–10506. https://doi.org/10.4238/2015.September.8.11
Soriano P (1997) The PDGF alpha receptor is required for neural crest cell development and for normal patterning of the somites. Development (Cambridge, England) 124:2691–2700
Tidball JG, Spencer MJ, St Pierre BA (1992) PDGF-receptor concentration is elevated in regenerative muscle fibers in dystrophin-deficient muscle. Exp Cell Res 203:141–149. https://doi.org/10.1016/0014-4827(92)90049-e
Uaesoontrachoon K, Yoo HJ, Tudor EM, Pike RN, Mackie EJ, Pagel CN (2008) Osteopontin and skeletal muscle myoblasts: association with muscle regeneration and regulation of myoblast function in vitro. Int J Biochem Cell Biol 40:2303–2314. https://doi.org/10.1016/j.biocel.2008.03.020
Wang X et al (2017) FTO is required for myogenesis by positively regulating mTOR-PGC-1alpha pathway-mediated mitochondria biogenesis. Cell Death Dis 8:e2702. https://doi.org/10.1038/cddis.2017.122
Wang Y, Rao K, Yuan L, Everaert N, Buyse J, Grossmann R, Zhao R (2012) Chicken FTO gene: tissue-specific expression, brain distribution, breed difference and effect of fasting. Comp Biochem Physiol A Mol Integr Physiol 163:246–252. https://doi.org/10.1016/j.cbpa.2012.08.009
Wu C (2007) Focal adhesion: a focal point in current cell biology and molecular medicine. Cell Adhes Migr 1:13–18. https://doi.org/10.4161/cam.1.1.4081
Wu J, Mao X, Cai T, Luo J, Wei L (2006) KOBAS server: a web-based platform for automated annotation and pathway identification. Nucleic Acids Res 34:W720–724. https://doi.org/10.1093/nar/gkl167
Zhao Y, Haginoya K, Sun G, Dai H, Onuma A, Iinuma K (2003) Platelet-derived growth factor and its receptors are related to the progression of human muscular dystrophy: an immunohistochemical study. J Pathol 201:149–159. https://doi.org/10.1002/path.1414
Acknowledgements
We thank International Science Editing (https://www.international science editing. com) for editing this manuscript.
Funding
The research was supported by the Natural Science Fund project in Jiangsu (BK20191217), China Meat-type Chicken Research System (CARS-42-Z06), Key Laboratory of Poultry Genetics and Breeding in Jiangsu (JQLAB-ZZ-201705), and the Special Funds for Transformation of Scientific and Technological Projects in Jiangsu Province (BA2019049).
Author information
Authors and Affiliations
Contributions
Conceptualization: HH and SL; validation: CL and LL; data curation: ZL; formal analysis: QW and ZH; writing the original draft: HH; supervision: SL.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
The study project was conducted in accordance with the Guidelines for Experimental Animals formulated by the Ministry of Science and Technology (Beijing, China). All experimental programs were approved by the Science Research Department (in charge of animal welfare) of the Institute of Animal Sciences, CAAS (Beijing, China).
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Huang, H., Liu, L., Li, C. et al. Fat mass- and obesity-associated (FTO) gene promoted myoblast differentiation through the focal adhesion pathway in chicken. 3 Biotech 10, 403 (2020). https://doi.org/10.1007/s13205-020-02386-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-020-02386-z