Abstract
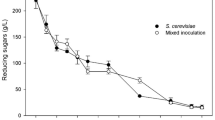
A study was conducted to concentrate the betacyanin in red pitahaya extracts by removing the coexisting sugars by fermentation. Four lactic acid bacteria (Lactobacillus acidophilus, L. casei, L. rhamnosus and L. plantarum) and a yeast species (Saccharomyces cerevisiae) were screened to determine their efficiency to reduce sugar content in red pitahaya extracts for concentration of their betacyanin content. A reduction of sugar content (19.8–56.4%) and increase in the yield of betacyanins were observed in all extracts as compared to the control, which was not innoculated with any microorganisms after 1 day of fermentation. The lowest total sugar content (26.40 g/L) was observed in extracts fermented by S. cerevisiae. Extracts fermented by S. cerevisiae also showed greater numbers of microbial cells (10.75 log CFU/mL) and a lower pH value (3.54) compared to those (6.89–8.48 log CFU/mL and pH 4.64–5.42) of the Lactobacillus spp. after 1 day of fermentation. An optimization step using response surface methodology (RSM) was then conducted using S. cerevisiae. Temperature, time and agitation speed were found to have a significant effect on the total sugar content and BC of concentrated betacyanins from red pitahaya, while the yield of betacyanins was significantly influenced by temperature and agitation speed.



Similar content being viewed by others
Code availability
Not applicable.
References
Badwaik LS, Prasad K, Deka SC (2012) Optimization of extraction conditions by response surface methodology for preparing partially defatted peanut. Int Food Res J 19:341–346
Castellar MR, Obόn JM, Alacid M, Fernández-López JA (2003) Colour properties and stability of betacyanins from Opuntia fruits. J Agric Food Chem 51:2772–2776
Castellar MR, Obόn JM, Alacid M, Fernández-López JA (2008) Fermentation of Opuntia stricta (Haw.) fruits for betalains concentration. J Agric Food Chem 56:4253–4257. https://doi.org/10.1021/jf703699c
Chethana S, Nayak CA, Raghavarao KSMS (2007) Aqueous two phase extraction for purification and concentration of betalains. J Food Eng 81:679–687. https://doi.org/10.1016/j.jfoodeng.2006.12.021
Chhabra RP (2003) Fluid-dynamics and heat transfer with non-Newtonian liquids in mechanically agitated vessels. Adv Heat Transf 37:77–178. https://doi.org/10.1016/S0065-2717(03)37002-9
Chien YL, Ho CT, Chiang BH, Hwang LS (2011) Effect of fermentation time on antioxidative activities of Ganoderma lucidum broth using leguminous plants as part of the liquid fermentation medium. Food Chem 126:1586–1592. https://doi.org/10.1016/j.foodchem.2010.12.024
Choo WS (2018) Betalains: Application in functional foods. In: Merillon J-M, Ramawat KG (eds) Bioactive molecules in food. Springer, Cham, pp 1–28
Costa MGM, Fonteles TV, Jesus ALT, Rodrigues S (2013) Sonicated pineapple juice as substrate for L. casei cultivation for probiotic beverage development: process optimisation and product stability. Food Chem 139:261–266. https://doi.org/10.1016/j.foodchem.2013.01.059
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356. https://doi.org/10.1021/ac60111a017
Faravash RS, Ashtiani FZ (2008) The influence of acid volume, ethanol-to-extract ratio and acid washing time on the yield of pectic substances extraction from peach pomace. Food Hydrocoll 22:196–202. https://doi.org/10.1016/j.foodhyd.2007.04.003
Gengatharan A, Dykes G, Choo WS (2015) Betalains: natural plant pigments with potential application in functional foods. LWT Food Sci Technol 64:645–649. https://doi.org/10.1016/j.lwt.2015.06.052
Gengatharan A, Dykes G, Choo WS (2016) Stability of betacyanin from red pitahaya (Hylocereus polyrhizus) and its potential application as a natural colourant in milk. Int J Food Sci Technol 51:427–434. https://doi.org/10.1111/ijfs.12999
Gengatharan A, Dykes G, Choo WS (2017) The effect of pH treatment and refrigerated storage on natural colourant preparations (betacyanins) from red pitahaya and their potential application in yoghurt. LWT Food Sci Technol 80:437–445. https://doi.org/10.1016/j.lwt.2017.03.014
Gengatharan A, Dykes G, Choo WS (2020) Betacyanins from Hylocereus polyrhizus: pectinase-assisted extraction and application as a natural food colourant in ice cream. J Food Sci Technol 58:1401–1410. https://doi.org/10.1007/s13197-020-04651-8
Harivaindran KV, Rebecca OPS, Chandran S (2008) Study of optimal temperature, pH and stability of dragon fruit (Hylocereus polyrhizus) peel for use as potential natural colourant. Pak J Biol Sci 11:2259–2263. https://doi.org/10.3923/pjbs.2008.2259.2263
Herbach KM, Stintzing FC, Carle R (2004) Impact of thermal treatment on colour and pigment pattern of red beet (Beta vulgaris L.) preparations. J Food Sci 69:C491–C498. https://doi.org/10.1111/j.1365-2621.2004.tb10994.x
Herbach KM, Rohe M, Stintzing FC, Carle R (2006a) Structural and chromatic stability of purple pitahaya (Hylocereus polyrhizus [Weber] Britton and Rose) betacyanins as affected by the juice matrix and selected additives. Food Res Int 39:667–677. https://doi.org/10.1016/j.foodres.2006.01.004
Herbach KM, Stintzing FC, Carle R (2006b) Betalain stability and degradation-structural and chromatic aspects. J Food Sci 71:R41–R50
Holzapfel WH, Wood BJB (2014) Introduction to the LAB. In: Holzapfel WH, Wood BJB (eds) Lactic acid bacteria—biodiversity and taxonomy. Wiley, Chichester, pp 1–12
Hou JW, Yu RC, Chou CC (2000) Changes in some components of soymilk during fermentation with bifidobacteria. Food Res Int 33:393–397. https://doi.org/10.1016/S0963-9969(00)00061-2
Huang WC, Chen SJ, Chen TL (2006) The role of dissolved oxygen and function of agitation in hyaluronic acid fermentation. Biochem Eng J 32:239–243. https://doi.org/10.1016/j.bej.2006.10.011
Koubaier HBH, Essaidi I, Snoussi A, Zgoulli S, Chaabouni M, Thonart P, Bouzouita N (2013) Effect of Saccharomyces cerevisiae fermentation on the colorants of heated red beet root extracts. Afr J Biotechnol 12:728–734. https://doi.org/10.5897/AJB12.2661
Lin X, Jia Y, Li K, Hu X, Li C, Liu S (2021) Effect of the inoculation strategies of selected Metschnikowia agaves and Saccharomyces cerevisiae on the volatile profile of pineapple wine in mixed fermentation. J Food Sci Technol. https://doi.org/10.1007/s13197-021-05019-2
Mohamad M, Ali MW, Ripin AA (2013) Effect of extraction process parameters on the yield of bioactive compounds from the roots of Eurycoma longifolia. Jurnal Teknologi 60:51–57. https://doi.org/10.11113/jt.v60.1441
Narendranath NV, Power R (2005) Relationship between pH and medium dissolved solids in terms of growth and metabolism of Lactobacilli and Saccharomyces cerevisiae during ethanol production. Appl Environ Microbiol 71:2239–2243.
Pourrat A, Lejeune B, Grand A, Pourrat H (1988) Betalains assay of fermented red beet root extract by high performance liquid chromatography. J Food Sci 53:294–295. https://doi.org/10.1111/j.1365-2621.1988.tb10237.x
Rodmui A, Kongkiattikajorn J, Dandusitapun Y (2008) Optimization of agitation conditions for maximum ethanol production by co-culture. Kasetsart J Nat Sci 42:285–293
Sener A, Canbaş A, Ünal MÜ (2007) The effect of fermentation temperature on the growth kinetics of wine yeast species. Turk J Agric For 31:349–354
Siegumfeldt H, Rechinger KB, Jakobsen M (2000) Dynamic changes of intracellular pH in individual lactic acid bacterium cells in response to a rapid drop in extracellular pH. Appl Environ Microbiol 66:2330–2335.
Sravan Kumar S, Manoj P, Giridhar P (2015) A method for red-violet pigments extraction from fruits of Malabar spinach (Basella rubra) with enhanced antioxidant potential under fermentation. J Food Sci Technol 52:3037–3043. https://doi.org/10.1007/s13197-014-1335-5
Stintzing FC, Schieber A, Carle R (2003) Evaluation of colour properties and chemical quality parameters of cactus juices. Eur Food Res Technol 216:303–311. https://doi.org/10.1007/s00217-002-0657-0
Wang L, Ou MS, Nieves I, Shanmugam KT (2015) Fermentation of sweet sorghum derived sugars to butyric acid at high titer and productivity by a moderate thermophile Clostridium thermobutyricum at 50°C. Bioresour Technol 198:533–539. https://doi.org/10.1016/j.biortech.2015.09.062
Yolmeh M, Najafi MBH, Farhoosh R (2014) Optimisation of ultrasound-assisted extraction of natural pigment from annatto seeds by response surface methodology (RSM). Food Chem 155:319–324. https://doi.org/10.1016/j.foodchem.2014.01.059
Yong YY, Dykes G, Lee SM, Choo WS (2018) Effect of refrigerated storage on betacyanin composition, antibacterial activity of red pitahaya (Hylocereus polyrhizus) and cytotoxicity evaluation of betacyanin rich extract on normal human cell lines. LWT Food Sci Technol 91:491–497. https://doi.org/10.1016/j.lwt.2018.01.078
Yuan Y, Gao Y, Mao L, Zhao J (2008) Optimisation of conditions for the preparation of β carotene nanoemulsions using response surface methodology. Food Chem 107:1300–1306. https://doi.org/10.1016/j.foodchem.2007.09.015
Funding
(Information that explains whether and by whom the research was supported). This work was funded by School of Science, Monash University Malaysia.
Author information
Authors and Affiliations
Contributions
AG—original draft, writing, performed the analysis and data collection. GAD—supervision, editing and writing. WSC—conceptualization, project management, supervision, editing and writing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declares that they have no conflict of interest statement.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and material
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gengatharan, A., Dykes, G.A. & Choo, W.S. Fermentation of red pitahaya extracts using Lactobacillus spp. and Saccharomyces cerevisiae for reduction of sugar content and concentration of betacyanin content. J Food Sci Technol 58, 3611–3621 (2021). https://doi.org/10.1007/s13197-021-05116-2
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-021-05116-2