Abstract
Introduction
Physostigmine fell out of widespread use in the 1980s due to safety concerns; however, more recent research has demonstrated that its safety profile is better than previously thought. These studies have mainly included adults. We theorized that improved safety data may lead to more acceptance. Our objectives, therefore, were to characterize current frequency of use of physostigmine in pediatric patients as well as to study adverse effect rates in a national pediatric patient population.
Methods
The National Poison Data System was queried for cases of patients aged 0–18 years that involved single-substance exposures to antimuscarinic xenobiotics that were reported to a poison center between January 1, 2000, and December 31, 2020. Cases were stratified into groups by therapy received: benzodiazepines alone, benzodiazepines and physostigmine, physostigmine alone, or no physostigmine or benzodiazepines. Patient demographics, clinical effects, and medical outcomes were analyzed.
Results
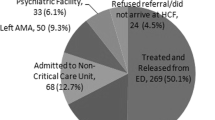
A total of 694,132 cases were reviewed, and 150,075 were included for analysis. Nearly 5% (7562/150,075) of patients received specific pharmacological therapy with benzodiazepines, physostigmine, or both. A benzodiazepine as a single agent was the most frequently used pharmacologic therapy (92% of 7562). Among patients receiving any pharmacological therapy, only 8.3% (n = 627) of patients received physostigmine. Frequency of serious outcomes significantly increased across the study period among patients receiving benzodiazepines alone or with physostigmine. There was no increase in serious outcomes among patients receiving only physostigmine.
Conclusions
Physostigmine frequency of use was low overall, but when used, was associated with less severe outcomes when compared to benzodiazepines.


Similar content being viewed by others
Change history
28 May 2024
A Correction to this paper has been published: https://doi.org/10.1007/s13181-024-01007-y
References
Dawson AH, Buckley NA. Pharmacological management of anticholinergic delirium - theory, evidence and practice. Br J Clin Pharmacol. 2016;81(3):516–24.
Frascogna N. Physostigmine: is there a role for this antidote in pediatric poisonings? Curr Opin Pediatr. 2007;19(2):201–5.
Hoffman RS, Goldfrank LR. The poisoned patient with altered consciousness: controversies in the use of a ‘coma cocktail.’ JAMA. 1995;274(7):562–9.
Pentel P, Peterson CD. Asystole complicating physostigmine treatment of tricyclic antidepressant overdose. Ann Emerg Med. 1980;9(11):588–90.
Suchard JR. Assessing physostigmine’s contraindication in cyclic antidepressant ingestions. J Emerg Med. 2003;25(2):185–91.
Watkins JW, Schwarz ES, Arroyo-Plasencia AM, Mullins ME, Toxicology Investigators Consortium Case Registry I. The use of physostigmine by toxicologists in anticholinergic toxicity. J Med Toxicol. 2015;11(2):179–84.
Arens AM, Kearney T. Adverse effects of physostigmine. J Med Toxicol. 2019;15(3):184–91.
Arens AM, Shah K, Al-Abri S, Olson KR, Kearney T. Safety and effectiveness of physostigmine: a 10-year retrospective review. Clin Toxicol (Phila). 2018;56(2):101–7.
Boley SP, Olives TD, Bangh SA, Fahrner S, Cole JB. Physostigmine is superior to non-antidote therapy in the management of antimuscarinic delirium: a prospective study from a regional poison center. Clin Toxicol (Phila). 2019;57(1):50–5.
Boley SP, Stellpflug SJ. A comparison of resource utilization in the management of anticholinergic delirium between physostigmine and nonantidote therapy. Ann Pharmacother. 2019;53(10):1026–32.
Burns MJ, Linden CH, Graudins A, Brown RM, Fletcher KE. A comparison of physostigmine and benzodiazepines for the treatment of anticholinergic poisoning. Ann Emerg Med. 2000;35(4):374–81.
Beaver KM, Gavin TJ. Treatment of acute anticholinergic poisoning with physostigmine. Am J Emerg Med. 1998;16(5):505–7.
Rasimas J, Sachdeva KK, Donovan JW. Revival of an antidote: bedside experience with physostigmine. Toxicology Communications. 2018;2(1):85–101.
Schneir AB, Offerman SR, Ly BT, Davis JM, Baldwin RT, Williams SR, et al. Complications of diagnostic physostigmine administration to emergency department patients. Ann Emerg Med. 2003;42(1):14–9.
Cole JB, Stellpflug SJ, Ellsworth H, Harris CR. Reversal of quetiapine-induced altered mental status with physostigmine: a case series. Am J Emerg Med. 2012;30(6):950–3.
Nguyen TT, Armengol C, Wilhoite G, Cumpston KL, Wills BK. Adverse events from physostigmine: an observational study. Am J Emerg Med. 2018;36(1):141–2.
Isbister GK, Oakley P, Dawson AH, Whyte IM. Presumed angel’s trumpet (Brugmansia) poisoning: clinical effects and epidemiology. Emerg Med. 2003;15(4):376–82.
Rumack BH. Anticholinergic poisoning: treatment with physostigmine. Pediatrics. 1973;52(3):449.
Funk W, Hollnberger H, Geroldinger J. Physostigmine and anaesthesia emergence delirium in preschool children: a randomized blinded trial. Eur J Anaesthesiol. 2008;25(1):37–42.
Palmer RB, Reynolds KM, Banner W, Randall Bond G, Kauffman RE, Paul IM, et al. Adverse events associated with diphenhydramine in children, 2008–2015. Clin Tox (Phila). 2019;58(2):1–8.
Derinoz O, Emeksiz HC. Use of physostigmine for cyclopentolate overdose in an infant. Pediatrics. 2012;130(3):e703–5.
Glatstein MM, Alabdulrazzaq F, Garcia-Bournissen F, Scolnik D. Use of physostigmine for hallucinogenic plant poisoning in a teenager: case report and review of the literature. Am J Therapeutics. 2012;19(5):384–8.
Kulka PJ, Toker H, Heim J, Joist A, Jakschik J. Suspected central anticholinergic syndrome in a 6-week-old infant. Anesth Analg. 2004;99(5):1376–8.
Phillips MA, Acquisto NM, Gorodetsky RM, Wiegand TJ. Use of a physostigmine continuous infusion for the treatment of severe and recurrent antimuscarinic toxicity in a mixed drug overdose. J Med Toxicol. 2014;10(2):205–9.
Rhyee SH, Pedapati EV, Thompson J. Prolonged delirium after quetiapine overdose. Pediatr Emer Care. 2010;26(10):754–6.
Schultz U, Idelberger R, Rossiant R, Buhre W. Central anticholinergic syndrome in a child undergoing circumcision. Acta Anaesthesiol Scand. 2002;46(2):224–6.
Thornton SL, Farnaes F, Minns A. Prolonged antimuscarinic delirium in a child due to benztropine exposure treated with multiple doses of physostigmine. Pediatr Emerg Care. 2016;32(4):243–5.
Slovis TL, Ott JE, Teitebaum DT, Lipscomb W. Physostigmine therapy in acute tricyclic antidepressant poisoning. Clin Toxicol (Phila). 1971;4(3):451–9.
Niewiñska K, Niewiñski Pa, Sokolowski J, Poradowska-Jeszke W, SokólOssowicz A, Wiela-Hojeñska A. Administration of acetylcholinesterase inhibitors for central anticholinergic syndrome in pediatric poisoning. Pharmacological Reports. 2007;59(1):226–31.
Wang GS, Baker K, Ng P, Janis GC, Leonard J, Mistry RD, et al. A randomized trial comparing physostigmine vs lorazepam for treatment of antimuscarinic (anticholinergic) toxidrome. Clin Toxicol (Phila). 2020;59(8):1–13.
Lovelace B. How U.S. drugmaker Akorn’s closure contributed to the escalating drug shortage crisis. NBC News [Internet]. July 16, 2023 [cited 10/24/23] Available from: https://www.nbcnews.com/health/health-news/akorn-us-drugmaker-closure-escalating-drug-shortage-rcna91402.
Whitledge JD, Soto P, Glowacki KM, Fox ER, Mazer-Amirshahi M. Shortages of agents used to treat antimuscarinic delirium. Am J Emerg Med. 2023;67:163–7.
Anticholinergic. In: Micromedex- Drug Classes [Database on the Internet]. Greenwood Village, CO, USA: IBM Watson Health. 2019; Available from: https://www.micromedexsolutions.com.
Duran CE, Azermai M, Vander Stichele RH. Systematic review of anticholinergic risk scales in older adults. European J Clinical Pharm. 2013;69(7):1485–96.
Salahudeen MS, Duffull SB, Nishtala PS. Anticholinergic burden quantified by anticholinergic risk scales and adverse outcomes in older people: a systematic review. BMC Geriatr. 2015;15(1):31.
Montastruc F, Benevent J, Touafchia A, Chebane L, Araujo M, Guitton-Bondon E, et al. Atropinic (anticholinergic) burden in antipsychotic-treated patients. Fundam Clin Pharmacol. 2018;32(1):114–9.
Howland MA. Physostigmine salicylate. In: Nelson LS, Howland MA, Lewin NA, editors. Goldfrank’s toxicologic emergencies. 11th ed. New York: McGraw Hill Education; 2019. p. 755–8.
American Association of Poison Control Centers. NPDS coding users’ manual©. 2014.
Doan UV, Wu ML, Phua DH, Mendez Rojas B, Yang CC. Datura and Brugmansia plants related antimuscarinic toxicity: an analysis of poisoning cases reported to the Taiwan poison control center. Clin Toxicol (Phila). 2018;54(4):1–8.
Acknowledgements
The authors are very grateful for the support of and guidance in statistical methodology from George Eckert, MAS, Biostatistician Supervisor in the Department of Biostatistics at the Indiana University School of Medicine and Richard M. Fairbanks School of Public Health.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by SH, RA, AO, and SP. The first draft of the manuscript was written by SH, RA, AO, and KN, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflicts of Interest
None.
Sources of Funding
There were no external sources of funding for this study.
Additional information
Supervising Editor: Howard Greller, MD.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original version of this article was revised: Table 1 contained typographical errors as originally published and has been updated, and a sentence in the 2nd paragraph of the Results section has been updated to reflect the corrected values in Table 1.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Huber, S., Avera, R., Penfound, S. et al. Safety of Physostigmine for Pediatric Antimuscarinic Poisoning. J. Med. Toxicol. (2024). https://doi.org/10.1007/s13181-024-00988-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13181-024-00988-0