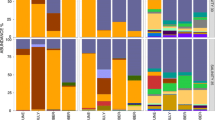
Abstract
Diatoms are a species-diverse phylum of microalgae often presenting high biomass in aquatic habitats. This makes them excellent ecological indicators in rivers and lakes. They are routinely used to assess ecological quality of rivers and lakes using microscopy, which is time consuming. An alternative is to determine species in samples based on short DNA barcodes and high-throughput sequencing (HTS). Former studies showed that community structure and water quality assessments based on diatoms deliver similar results with both methods. But, none evaluated if diversities were assessed in the same way despite the importance of this ecological metric. Based on littoral benthic samplings carried out in 56 pristine alpine lakes, we compared different diversity indices measured with microscopy and metabarcoding. Each lake was sampled in three different places of its littoral. We showed that α (diversity measured in a single sampling site of a given lake) and ϒ (total diversity in a lake where three independent samples were considered) diversities obtained with HTS were higher than those obtained with microscopy. This may be explained by the capacity of HTS to detect morphologically cryptic species and to better detect rare taxa. On the other hand, β diversity obtained with HTS was smaller, which may be explained by the capacity of HTS to detect very rare species and free-floating extracellular DNA. Nevertheless, diversity indices obtained with both methodologies were well correlated each other. This study validates the possibility to assess diatom diversity with HTS in a comparable way to the classical microscopic analysis.






Similar content being viewed by others
References
Abarca, N., Jahn, R., Zimmermann, J., & Enke, N. (2014). Does the cosmopolitan diatom Gomphonema parvulum (Kutzing) Kutzing have a biogeography? PLoS One, 9, 1–18.
Amend, A. S., Seifert, K. A., & Bruns, T. D. (2010). Quantifying microbial communities with 454 pyrosequencing: does read abundance count? Molecular Ecology, 19, 5555–5565.
Apothéloz-Perret-Gentil, L., Cordonier, A., Straub, F., Iseli, J., Esling, P., & Pawlowski, J. (2017). Taxonomy-free molecular diatom index for high-throughput eDNA biomonitoring. Molecular Ecology Resources. https://doi.org/10.1111/1755-0998.12668.
Baird, D. J., & Hajibabaei, M. (2012). Biomonitoring 2.0: a new paradigm in ecosystem assessment made possible by next-generation DNA sequencing. Molecular Ecology, 1–6.
Berthon, V., Marchetto, A., Rimet, F., Dormia, E., Jenny, J. P., Pignol, C., & Perga, M. E. (2013). Trophic history of French sub-alpine lakes over the last ~150 years: phosphorus reconstruction and assessment of taphonomic biases. Journal of Limnology, 72, 417–429.
Bray, J. R., & Curtis, J. T. (1957). An ordination of upland forest communities of southern Wisconsin. Ecological Monographs, 27, 325–349.
Cantonati, M., & Lowe, R. L. (2014). Lake benthic algae: toward an understanding of their ecology. Freshwater Science, 33(2), 475–486.
Chonova, T., Keck, F., Labanowski, J., Montuelle, B., Rimet, F., & Bouchez, A. (2016). Seperate treatment of hospital and urban wastewaters: a real scale comparison of effluents and their effect on microbial communities. Science of the Total Environment, 542, 965–975.
Civade, R., Dejean, T., Valentini, A., Roset, N., Raymond, J.-C., Bonin, A., et al. (2016). Spatial representativeness of environmental DNA metabarcoding signal for fish biodiversity assessment in a natural freshwater system. PLoS One, 11(6), e0157366. https://doi.org/10.1371/journal.pone.0157366.
Copeland, C. (2016). Clean Water Act: A Summary of the Law. Congressional Research Service, CRS report, number 7–5700 RL30030, pp. 1–10.
Debroas, D., Hugoni, M., & Domaizon, I. (2015). Evidence for an active rare biosphere within freshwater protists community - Debroas - 2015 - Molecular Ecology - Wiley Online Library. Molecular Ecology, 24, 1236–1247. https://doi.org/10.1111/mec.13116.
Debroas, D., Domaizon, I., Humbert, J.-F., Jardillier, L., Lepère, C., Oudart, A., & Taïb, N. (2017). Overview of freshwater microbial eukaryotes diversity: a first analysis of publicly available metabarcoding data. FEMS Microbiology Ecology, 93(4). https://doi.org/10.1093/femsec/fix023.
Dejean, T., Valentini, A., Duparc, A., Pellier-Cuit, S., Pompanon, F., Taberlet, P., & Miaud, C. (2011). Persistence of environmental DNA in freshwater ecosystems. PLoS One, 6(8), e23398. https://doi.org/10.1371/journal.pone.0023398.
European Commission. (2000). Directive 2000/60/EC of the European Parliament and of the Council of 23rd October 2000 establishing a framework for Community action in the field of water policy. Official Journal of the European Communities, 327, 1–72.
European Committee for Standardization (2014a) EN 13946 Water quality—guidance for the routine sampling and preparation of benthic diatoms from rivers and lakes. CEN standard, 1–18.
European Committee for Standardization (2014b). EN 14407 Water quality—guidance for the identification and enumeration of benthic diatom samples from rivers and lakes. CEN standard, 1–13.
Evans, K. M., Wortley, A. H., Simpson, G. E., Chepurnov, V. A., & Mann, D. G. (2008). A molecular systematic approach to explore diversity within the Sellaphora pupula species complex (Bacillariophyta). Journal of Phycology, 44(1), 215–231.
Feret, L., Bouchez, A., & Rimet, F. (2017). Benthic diatom communities in high altitude lakes: a large scale study in the French Alps. International Journal of Limnology. https://doi.org/10.1051/limn/2017026.
Ferreira da Silva, E., Almeida, S. F. P., Nunes, M. L., Luís, A. T., Borg, F., Hedlund, M., et al. (2009). Heavy metal pollution downstream the abandoned Coval da Mó mine (Portugal) and associated effects on epilithic diatom communities. Science of the Total Environment, 407(21), 5620–5636. https://doi.org/10.1016/j.scitotenv.2009.06.047.
Fontana, G., Ugland, K. I., Gray, J. S., Willis, T. J., & Abbiati, M. (2008). Influence of rare species on beta diversity estimates in marine benthic assemblages. Journal of Experimental Marine Biology and Ecology, 366(1), 104–108. https://doi.org/10.1016/j.jembe.2008.07.014.
Hammer, O., Harper, D. A. T., & Ryan, P. D. (2001). PAST: paleontological statistics software package for education and data analysis. Palaeontologia Electronica, 4, 1–9.
Hebert, P., Cywinska, A., Ball, S. L., & deWaard, J. R. (2003). Biological identifications through DNA barcodes. Proceedings of the Royal Society B-Biological Sciences, 270, 313–321.
Hoagland, K. D. (1981). Diatom colonization, community structure and succession on artificial substrate in freshwater. phD, University of Nebraska, Lincoln, USA.
Hoffman, G., Werum, M., & Lange-Bertalot, H. (2011). Diatomeen im Susswasser-benthos von Mitteleuropa. Rugell: A.R.G. Gantner Verlag K.G..
Kebschull, J. M., & Zador, A. M. (2015). Sources of PCR-induced distortions in high-throughput sequencing data sets. Nucleic Acids Research, 43, e143.
Keck, F., Vasselon, V., Tapolczai, K., Rimet, F., & Bouchez, A. (2017). Freshwater biomonitoring in the Information Age. Frontiers in Ecology and the Environment, 15(5), 266–274. https://doi.org/10.1002/fee.1490.
Kermarrec, L., Bouchez, A., Rimet, F., & Humbert, J. F. (2012). First evidence of the existence of semi-cryptic species and of a phylogeographic structure in the Gomphonema parvulum (Kützing) Kützing complex (Bacillariophyta). Protist, 164, 686–705.
Kermarrec, L., Franc, A., Rimet, F., Chaumeil, P., Humbert, J. F., & Bouchez, A. (2013). Next-generation sequencing to inventory taxonomic diversity in eukaryotic communities: a test for freshwater diatoms. Molecular Ecology Resources, 13(4), 607–619.
Kermarrec, L., Franc, A., Rimet, F., Chaumeil, P., Frigerio, J. M., Humbert, J. F., & Bouchez, A. (2014). A next-generation sequencing approach to river biomonitoring using benthic diatoms. Freshwater Science, 33(1), 349–363.
King, L., Clarke, G., Bennion, H., Kelly, M., & Yallop, M. (2006). Recommendations for sampling littoral diatoms in lakes for ecological status assessments. Journal of Applied Phycology, 18(1), 15–25. https://doi.org/10.1007/s10811-005-9009-3.
Kociolek, J. P. (2005). Taxonomy and ecology: further considerations. Proceedings of the California Academy of Sciences, 56, 99–106.
Kociolek, J. P., Sabbe, K., Vandepitte, L., Decock, W., & Vanhoorn, B. (2016). Catalogue of diatom names resurrected: DiatomBase will be the new authority resource for diatom names and more. 24th International Diatom Symposium, 100.
Krammer, K., & Lange-Bertalot, H. (1986). Bacillariophyceae 1. Teil: Naviculaceae. Susswasserflora von Mitteleuropa. Stuttgart: Gustav Fischer Verlag.
Krammer, K., & Lange-Bertalot, H. (1988). Bacillariophyceae 2. Teil: Bacillariaceae, Epithemiaceae, Surirellaceae. Susswasserflora von Mitteleuropa. Stuttgart: Gustav Fischer Verlag.
Krammer, K., & Lange-Bertalot, H. (1991a). Bacillariophyceae 3. Teil: Centrales, Fragilariaceae, Eunotiaceae. Susswasserflora von Mitteleuropa. Stuttgart: Gustav Fischer Verlag.
Krammer, K., & Lange-Bertalot, H. (1991b). Bacillariophyceae 4. Teil: Achnanthaceae. Kritische Erganzungen zu Navicula (Lineolatae) und Gomphonema. Gesamtliteraturverzeichnis Teil 4. Susswasserflora von Mitteleuropa. Stuttgart: Gustav Fischer Verlag.
Lange-Bertalot, H., & Metzeltin, D. (1996). Indicators of oligotrophy. 800 taxa representative of three ecologically distinct lakes. Carbonate buffered - Oligodystrophic - Weakly buffered soft water. Konigstein: Koeltz scientific books.
Lejzerowicz, F., Esling, P., Pillet, L., Wilding, T. A., Black, K. D., & Pawlowski, J. (2015). High-throughput sequencing and morphology perform equally well for benthic monitoring of marine ecosystems. Scientific Reports, 5. https://doi.org/10.1038/srep13932.
Levkov, Z., Krstic, S., Metzeltin, D., & Nakov, T. (2007). Diatoms of Lakes Prespa and Ohrid. about 500 taxa from ancient lake system. Iconographia Diatomologica, volume 16 (p. 613). ARG Gantner Verlag KG.
Loman, N. J., Misra, R. V., Dallman, T. J., Constantinidou, C., Gharbia, S. E., Wain, J., & Pallen, M. J. (2012). Performance comparison of benchtop high-throughput sequencing platforms. Nature Biotechnology, 30, 434–439.
Luís, A. T., Teixeira, P., Almeida, S. F. P., Ector, L., Matos, J. X., & Silva, E. A. F. d. (2009). Impact of acid mine drainage (AMD) on water quality, stream sediments and periphytic diatom communities in the surrounding streams of Aljustrel mining area (Portugal). Water, Air, and Soil Pollution, 200(1–4), 147–167. https://doi.org/10.1007/s11270-008-9900-z.
Mangot, J. F., Domaizon, I., Taib, N., Marouni, N., Duffaud, E., Bronner, G., & Debroas, D. (2013). Short-term dynamics of diversity patterns: evidence of continual reassembly within lacustrine small eukaryotes. Environmental Microbiology, 15, 1745–1758.
Mann, D. G., & Vanormelingen, P. (2013). An inordinate fondness? The number, distributions and origins of diatom species. Journal of Eukaryotic Microbiology, 60, 1–26.
Mann, D. G., McDonald, S. M., Bayer, M. M., Droop, S. J. M., Chepurnov, V. A., Loke, R. E., et al. (2004). The Sellaphora pupula species complex (Bacillariophyceae): morphometric analysis, ultrastructure and mating data provide evidence for five new species. Phycologia, 43(4), 459–482.
Mantel, N. (1967). The detection of disease clustering and a generalized regression approach. Cancer Research, 27, 209–220.
Morin, S., Bonet, B., Corcoll, N., Guasch, H., Bottin, M., & Coste, M. (2015). Cumulative stressors trigger increased vulnerability of diatom communities to additional disturbances. Microbial Ecology, 70, 585–595.
Nolte, V., Pandey, R. V., Jost, S. T., Medinger, R., Ottenwalder, O., Boengik, J., & Schlotterer, C. (2010). Contrasting seasonal niche separation between rare and abundant taxa conceals the extent of protist diversity. Molecular Ecology, 19, 2908–2915.
Pandey, L. K., Bergey, E. A., Lyu, J., Park, J., Choi, S., Lee, H., et al. (2017). The use of diatoms in ecotoxicology and bioassessment: insights, advances and challenges. Water Research, 118, 39–58.
Passy, S. I. (2008). Continental diatom biodiversity in stream benthos declines as more nutrients become limiting. Proceedings of the National Academy of Sciences of the United States of America, 105, 9663–9667.
Passy, S. I. (2009). The relationship between local and regional diatom richness is mediated by the local and regional environment. Global Ecology and Biogeography, 18(3), 383–391.
Pinto, A. J., & Raskin, L. (2012). PCR biases distort bacterial and archaeal community structure in pyrosequencing datasets. PLoS One, 7, e43093.
Pompanon, F., Coissac, E., & Taberlet, P. (2011). Metabarcoding a new way to analyze biodiversity. Biofutur, 319, 30–32.
Potapova, M., & Charles, D. F. (2007). Diatom metrics for monitoring eutrophication in rivers of the United States. Ecological Indicators, 7(1), 48–70.
Ricciardi, F., Bonnineau, A., Faggiano, L., Geiszinger, A., Guasch, H., Lopez-Doval, J. C., et al. (2009). Is chemical contamination linked to the diversity of biological communities in rivers? Trends in Analytical Chemistry, 28, 592–602.
Rimet, F. (2012). Recent views on river pollution and diatoms. Hydrobiologia, 683, 1–24.
Rimet, F., Kermarrec, L., Bouchez, A., Hoffmann, L., Ector, L., & Medlin, L. (2011). Molecular phylogeny of the family Bacillariaceae based on 18S rDNA sequences: focus on freshwater Nitzschia of the Lanceolatae section. Diatom Research, 26, 273–291.
Rimet, F., Trobajo, R., Mann, D. G., Kermarrec, L., Franc, A., Domaizon, I., & Bouchez, A. (2014). When is sampling complete? The effects of geographical range and marker choice on perceived diversity in Nitzschia palea (Bacillariophyta). Protist, 165(3), 245–259.
Rimet, F., Chaumeil, P., Keck, F., Kermarrec, L., Vasselon, V., Kahlert, M., et al. (2016). R-Syst::diatom: an open-access and curated barcode database for diatoms and freshwater monitoring. Database: The Journal of Biological Databases and Curation, 2016(1), 21. https://doi.org/10.1093/database/baw016.
Rimet, F., Abarca, N., Bouchez, A., Kusber, W. H., Jahn, R., Kahlert, M., et al. (2018). The potential of high throughput sequencing (HTS) of natural samples as a source of primary taxonomic information for reference libraries of diatom barcodes. Fottea. https://doi.org/10.5507/fot.2017.013.
Rivera, S., Vasselon, V., Jacquet, S., Ariztegui, D., Bouchez, A., & Rimet, F. (2018). Metabarcoding of lake benthic diatoms: from structure assemblages to ecological assessment. Hydrobiologia, 1–12 https://doi.org/10.1007/s10750-017-3381-2.
Routledge, R. D. (1977). On Whittaker’s components of diversity. Ecology, 58, 1120–1127.
Schloss, P. D., Westcott, S. L., Ryabin, T., Hall, J. R., Hartmann, M., Hollister, E. B., et al. (2009). Introducing mothur: open-source, platform-independent, community-supported software for describing and comparing microbial communities. Applied and Environmental Microbiology, 75, 7537–7541.
Stevenson, R. J. (2014). Ecological assessments with algae: a review and synthesis. Journal of Phycology, 50, 437–461.
Stevenson, R. J., Pan, Y., & Van Dam, H. (2010). Assessing environmental conditions in rivers and streams with diatoms. In J. P. Smol & E. F. Stoermer (Eds.), The Diatoms: Applications for the Environmental and Earth Sciences (second ed., pp. 57–85). London: Cambridge University Press.
Tapolczai, K., Vasselon, V., Bouchez, A., Stenger-Kovacs, C., Padisak, J., & Rimet, F. (2017). Taxonomy-free DNA biomonitoring for rivers: How to choose the sequence similarity threshold? Under revision.
Trobajo, R., Clavero, E., Chepurnov, V., Sabbe, K., Mann, D. G., Ishihara, S., & Cox, E. J. (2009). Morphological, genetic and mating diversity within the widespread bioindicator Nitzschia palea (Bacillariophyceae). Phycologia, 48, 443–459.
Trobajo, R., Mann, D. G., Clavero, E., Evans, K. M., Vanormelingen, P., & McGregor, R. C. (2010). The use of partial cox1, rbcL and LSU rDNA sequences for phylogenetics and species identification within the Nitzschia palea species complex (Bacillariophyceae). European Journal of Phycology, 45(4), 413–425.
Valentini, A., Taberlet, P., Miaud, C., Civade, R., Herder, J., Thomsen, P. F., et al. (2016). Next-generation monitoring of aquatic biodiversity using environmental DNA metabarcoding. Molecular Ecology, 25(4), 929–942. https://doi.org/10.1111/mec.13428.
Vasselon, V., Domaizon, I., Rimet, F., Kahlert, M., & Bouchez, A. (2016). Application of high-throughput sequencing (HTS) metabarcoding to diatom biomonitoring: do DNA extraction methods matter? Freshwater Science, 36(1), 162–177.
Vasselon, V., Bouchez, A., Rimet, F., Jacquet, S., Trobajo, R., Corniquel, M., et al. (2017a). A correction factor inferred from cell biovolume improves quantification in diatom metabarcoding. Methods in Ecology and Evolution, https://doi.org/10.1111/2041-210X.12960.
Vasselon, V., Rimet, F., Tapolczai, K., & Bouchez, A. (2017b). Assessing ecological status with diatoms DNA metabarcoding: scaling-up on a WFD monitoring network (Mayotte island, France). Ecological Indicators, 82, 1–12. https://doi.org/10.1016/j.ecolind.2017.06.024.
Visco, J., Apotheloz-Perret-Gentil, L., Cordonier, A., Esling, P., Pillet, L., & Pawlowski, J. (2015). Environmental monitoring: inferring the diatom index from next-generation sequencing data. Environmental Science and Technology, 49, 7597–7605.
Weaver, W., & Shannon, C. E. (1949). The mathematical theory of communication.
Werum, M., Lange-Bertalot, H., & Werum, M. (2004). Diatoms in springs from Central Europe ane elsewhere under the influence of hydrogeology and anthropogenic impacts. Iconographia Diatomologica, volume 13. A.R.G. Ruggell: Gantner Verlag K.G..
Whittaker, R. H. (1960). Vegetation of the Siskiyou Mountains, Oregon and California. Ecological Monographs, 30, 279–338.
Youssef, N. H., Couger, M. B., & Elshahed, M. S. (2010). Fine-scale bacterial beta diversity within a complex ecosystem (Zodletone Spring, OK, USA): the role of the rare biosphere. PLoS One, 5, e12414. https://doi.org/10.1371/journal.pone.0012414.
Zimmermann, J., Jahn, R., & Gemeinholzer, B. (2011). Barcoding diatoms: evaluation of the V4 subregion on the 18S rRNA gene, including new primers and protocols. Organisms Diversity and Evolution, 11, 173. https://doi.org/10.1007/s13127-011-0050-6.
Zimmermann, J., Glöckner, G., Jahn, R., Enke, N., & Gemeinholzer, B. (2014). Metabarcoding vs. morphological identification to assess diatom diversity in environmental studies. Molecular Ecology Resources, 15, 526–542.
Acknowledgements
We thank Pr. E. Dambrine for initiating this study with the students of Savoie-Mont-Blanc University, F. Arthaud for following this study and managing the data, Léa Feret and David Pobel for their participation in determining diatom samples under microscope, and Cécile Chardon, Sonia Lacroix, and Louis Jacas for processing the samples in molecular biology.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rimet, F., Vasselon, V., A.-Keszte, B. et al. Do we similarly assess diversity with microscopy and high-throughput sequencing? Case of microalgae in lakes. Org Divers Evol 18, 51–62 (2018). https://doi.org/10.1007/s13127-018-0359-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13127-018-0359-5