Abstract
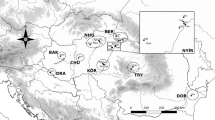
The pattern of genetic variation in a butterfly species depends on the past history of the given species and also on recent evolutionary processes affecting its populations. The aim of the present study was (i) to analyse the enzyme polymorphism in the Clouded Apollo populations of the Carpathian Basin to reveal the contemporary pattern of their genetic differentiation and (ii) to compare it with an expanded mitochondrial DNA (mtDNA) haplotype network of the SE European populations. Allozyme polymorphism was analysed in 22 populations of four geographic regions: Transdanubian (TM) and North Hungarian Mountains (NM), Körös (KÖR) and Bereg–Apuseni–East Carpathian regions (BEAC). The results of the Bayesian clustering analyses based on allozymes supported the presence of three main genetic lineages in the Carpathian Basin: One was typical for TM, another was characteristic for NM and the third cluster was predominant in KÖR. The populations of BEAC harboured a mixture of two clusters. The mtDNA haplotype network suggested a fairly similar distribution: The peri-Alpine clade together with the West Balkan clade was detected in TM, while the East Balkan clade occurred in NM, partly in TR and in the two eastern regions of the Basin (KÖR and BAEC). The incongruities between the results of the mtDNA and allozyme studies can be explained by the different timescales represented by the two markers. The mtDNA haplotype network provided strong evidence concerning the existence of two Balkan lineages, which probably formed a ‘zone of admixture’ in the Transdanubian and North Hungarian Mountains. The possibility of Last Glacial survival of Parnassius mnemosyne in the Carpathian Basin and the conservation implications of these results are discussed.





Similar content being viewed by others
References
Allendorf, F. W., & Luikart, G. (2009). Conservation and the Genetics of Populations. John Wiley & Sons.
Antao, T., Lopes, A., Lopes, R. J., Beja-Pereira, A., & Luikart, G. (2008). LOSITAN: a workbench to detect molecular adaptation based on a F(st)-outlier method. BMC Bioinformatics, 9, 323. doi:10.1186/1471-2105-9-323.
Babik, W., Branicki, W., Sandera, M., Litvinchuk, S., Borkin, L. J., Irwin, J. T., & Rafiński, J. (2004). Mitochondrial phylogeography of the moor frog, Rana arvalis. Molecular Ecology, 13(6), 1469–1480. doi:10.1111/j.1365-294X.2004.02157.x.
Beaumont, M. A., & Nichols, R. A. (1996). Evaluating loci for use in the genetic analysis of population structure. Proceedings of the Royal Society of London B: Biological Sciences, 263(1377), 1619–1626. doi:10.1098/rspb.1996.0237.
Bereczki, J., Pecsenye, K., Peregovits, L., & Varga, Z. (2005). Pattern of genetic differentiation in the Maculinea alcon species group (Lepidoptera, Lycaenidae) in Central Europe. Journal of Zoological Systematics and Evolutionary Research, 43(2), 157–165. doi:10.1111/j.1439-0469.2005.00305.x.
Bereczki, J., Tóth, J. P., Sramkó, G., & Varga, Z. (2014). Multilevel studies on the two phenological forms of Large Blue (Maculinea arion) (Lepidoptera: Lycaenidae). Journal of Zoological Systematics and Evolutionary Research, 52(1), 32–43. doi:10.1111/jzs.12034.
Besold, J., & Schmitt, T. (2015). More northern than ever thought: refugia of the woodland ringlet butterfly Erebia medusa (Nymphalidae: Satyrinae) in Northern Central Europe. Journal of Zoological Systematics and Evolutionary Research, 53, 67–75. doi:10.1111/jzs.12076.
Bilton, D. T., Mirol, P. M., Mascheretti, S., Fredga, K., Zima, J., & Searle, J. B. (1998). Mediterranean Europe as an area of endemism for small mammals rather than a source for northwards postglacial colonization. Proceedings of the Royal Society of London B: Biological Sciences, 265(1402), 1219–1226. doi:10.1098/rspb.1998.0423.
Cassel-Lundhagen, A., Tammaru, T., Windig, J. J., Ryrholm, N., & Nylin, S. (2009). Are peripheral populations special? Congruent patterns in two butterfly species. Ecography, 32(4), 591–600. doi:10.1111/j.1600-0587.2008.05685.x.
Crandall, K. A., Bininda-Emonds, O. R. P., Mace, G. M., & Wayne, R. K. (2000). Considering evolutionary processes in conservation biology. Trends in Ecology & Evolution, 15(7), 290–295. doi:10.1016/S0169-5347(00)01876-0.
de Guia, A. P. O., & Saitoch, T. (2011). Evolutionarily significant units of gray-sided vole (Myodes rufocanus) in Hokkaido, Japan. Philippine Journal of Science, 140(1), 41–50.
Earl, D. A., & vonHoldt, B. M. (2011). STRUCTURE HARVESTER: a website and program for visualizing STRUCTURE output and implementing the Evanno method. Conservation Genetics Resources, 4(2), 359–361. doi:10.1007/s12686-011-9548-7.
Evanno, G., Regnaut, S., & Goudet, J. (2005). Detecting the number of clusters of individuals using the software structure: a simulation study. Molecular Ecology, 14(8), 2611–2620. doi:10.1111/j.1365-294X.2005.02553.x.
Filipi, K., Marková, S., Searle, J. B., & Kotlík, P. (2015). Mitogenomic phylogenetics of the bank vole Clethrionomys glareolus, a model system for studying end-glacial colonization of Europe. Molecular Phylogenetics and Evolution, 82(Part A), 245–257. doi:10.1016/j.ympev.2014.10.016.
Filz, K. J., Engler, J. O., Stoffels, J., Weitzel, M., & Schmitt, T. (2013). Missing the target? A critical view on butterfly conservation efforts on calcareous grasslands in south-western Germany. Biodiversity and Conservation, 22, 2223–2241. doi:10.1007/s10531-012-0413-0.
Francis, B., Green, M., & Payne, C. (1994). GLIM 4. The statistical system for generalised linear interactive modelling. New York: Oxford University Press.
Fraser, D. J., & Bernatchez, L. (2001). Adaptive evolutionary conservation: towards a unified concept for defining conservation units. Molecular Ecology, 10, 2741–2752.
Freese, A., Benes, J., Bolz, R., Cizek, O., Dolek, M., Geyer, A., et al. (2006). Habitat use of the endangered butterfly Euphydryas maturna and forestry in Central Europe. Animal Conservation, 9, 388–397.
Hewitt, G. M. (1999). Post-glacial re-colonization of European biota. Biological Journal of the Linnean Society, 68(1–2), 87–112. doi:10.1111/j.1095-8312.1999.tb01160.x.
Goudet, J. (1995). FSTAT (Version 1.2): a computer program to calculate F-statistics. Journal of Heredity, 86(6), 485–486.
Gratton, P., Konopiński, M. K., & Sbordoni, V. (2008). Pleistocene evolutionary history of the Clouded Apollo (Parnassius mnemosyne): genetic signatures of climate cycles and a “time-dependent” mitochondrial substitution rate. Molecular Ecology, 17(19), 4248–4262. doi:10.1111/j.1365-294X.2008.03901.x.
Habel, J. C., Schmitt, T., & Müller, P. (2005). The fourth paradigm pattern of post-glacial range expansion of European terrestrial species: the phylogeography of the Marbled White butterfly (Satyrinae, Lepidoptera). Journal of Biogeography, 32(8), 1489–1497. doi:10.1111/j.1365-2699.2005.01273.x.
Hammer, Ø., Harper, D. A. T., & Ryan, P. D. (2001). PAST: paleontological statistics software package for education and data analysis. Palaeontologia Electronica, 4(1), 9.
Hampe, A., & Petit, R. J. (2005). Conserving biodiversity under climate change: the rear edge matters. Ecology Letters, 8(5), 461–467. doi:10.1111/j.1461-0248.2005.00739.x.
Heath, J. (1981). Threatened Rhopalocera (butterflies) in Europe. Veroffentlichungen fur Naturschutz und Landschaftspflege in Baden-Wurttemberg. Beiheft. http://agris.fao.org/agris-search/search.do?recordID=US201302579729. Accessed 14 December 2015
Hewitt, G. (2000). The genetic legacy of the Quaternary ice ages. Nature, 405(6789), 907–913. doi:10.1038/35016000.
Hewitt, G. M. (1996). Some genetic consequences of ice ages, and their role in divergence and speciation. Biological Journal of the Linnean Society, 58(3), 247–276. doi:10.1006/bijl.1996.0035.
Hewitt, G. M. (2001). Speciation, hybrid zones and phylogeography—or seeing genes in space and time. Molecular Ecology, 10(3), 537–549. doi:10.1046/j.1365-294x.2001.01202.x.
Hewitt, G. M. (2004). Genetic consequences of climatic oscillations in the Quaternary. Philosophical Transactions of the Royal Society of London B: Biological Sciences, 359(1442), 183–195. doi:10.1098/rstb.2003.1388.
Hughes, J. B., Daily, G. C., & Ehrlich, P. R. (1997). Population diversity: its extent and extinction. Science, 278(5338), 689–692. doi:10.1126/science.278.5338.689.
Jaarola, M., & Searle, J. B. (2002). Phylogeography of field voles (Microtus agrestis) in Eurasia inferred from mitochondrial DNA sequences. Molecular Ecology, 11(12), 2613–2621. doi:10.1046/j.1365-294X.2002.01639.x.
Konvička, M., & Kuras, T. (1999). Population structure, behaviour and selection of oviposition sites of an endangered butterfly, Parnassius Mnemosyne, in Litovelské Pomoravíl. Czech Republic. Journal of Insect Conservation, 3(3), 211–223. doi:10.1023/A:1009641618795.
Kotlík, P., Deffontaine, V., Mascheretti, S., Zima, J., Michaux, J. R., & Searle, J. B. (2006). A northern glacial refugium for bank voles (Clethrionomys glareolus). Proceedings of the National Academy of Sciences, 103(40), 14860–14864. doi:10.1073/pnas.0603237103.
Maes, D., & Dyck, H. V. (2005). Habitat quality and biodiversity indicator performances of a threatened butterfly versus a multispecies group for wet heathlands in Belgium. Biological Conservation, 123(2), 177–187. doi:10.1016/j.biocon.2004.11.005.
Maes, D., & Van Dyck, H. (2001). Butterfly diversity loss in Flanders (north Belgium): Europe’s worst case scenario? Biological Conservation, 99(3), 263–276. doi:10.1016/S0006-3207(00)00182-8.
Maes, D., Vanreusel, W., Talloen, W., & Dyck, H. V. (2004). Functional conservation units for the endangered Alcon Blue butterfly Maculinea alcon in Belgium (Lepidoptera: Lycaenidae). Biological Conservation, 120(2), 229–241. doi:10.1016/j.biocon.2004.02.018.
Magri, D. (2008). Patterns of post-glacial spread and the extent of glacial refugia of European beech (Fagus sylvatica). Journal of Biogeography, 35(3), 450–463. doi:10.1111/j.1365-2699.2007.01803.x.
Magri, D., Vendramin, G. G., Comps, B., Dupanloup, I., Geburek, T., Gömöry, D., et al. (2006). A new scenario for the Quaternary history of European beech populations: palaeobotanical evidence and genetic consequences. New Phytologist, 171(1), 199–221. doi:10.1111/j.1469-8137.2006.01740.x.
Magyari, E. (2002). Holocene biogeography of Fagus sylvatica L. and Carpinus betulus L. in the Carpathian-Alpine Region. Folia Historico – Naturalia Musei Matrensis, 26, 15–35.
Malicky, H., Ant, H., Aspöck, H., De Jong, R., Thaler, K., & Varga, Z. (1983). Argumente zur Existenz und Chorologie mitteleuropäischer (extramediterran-europäischer) Faunen-Elemente. Entomologia generalis, 9(1–2), 101–119.
Mantel, N. (1967). The Detection of Disease Clustering and a Generalized Regression Approach. Cancer Research, 27(2 Part 1), 209–220.
Meglécz, E., Nève, G., Pecsenye, K., & Varga, Z. (1999). Genetic variations in space and time in Parnassius mnemosyne (L.) (Lepidoptera) populations in north-east Hungary: implications for conservation. Biological Conservation, 89(3), 251–259. doi:10.1016/S0006-3207(99)00006-3.
Meglécz, E., Pecsenye, K., Peregovits, L., & Varga, Z. (1997). Allozyme variation in Parnassius mnemosyne (L.) (Lepidoptera) populations in North-East Hungary: variation within a subspecies group. Genetica, 101(1), 59–66. doi:10.1023/A:1018368622549.
Meglécz, E., Pecsenye, K., Varga, Z., & Solignac, M. (1998). Comparison of differentiation pattern at allozyme and microsatellite loci in Parnassius Mnemosyne (Lepidoptera) populations. Hereditas, 128(2), 95–103. doi:10.1111/j.1601-5223.1998.00095.x.
Moritz, C. (1994). Defining “evolutionarily significant units” for conservation. Trends in Ecology & Evolution, 9(10), 373–375. doi:10.1016/0169-5347(94)90057-4.
New, T. R. (1993). Introduction to the biology and conservation of the Lycaenidae. In Conservation biology of Lycaenidae (Butterflies). (Vol. 8, pp. 1–21.). Gland, Switzerland: IUCN.
Paradis, E. (2010). pegas: an R package for population genetics with an integrated–modular approach. Bioinformatics, 26(3), 419–420. doi:10.1093/bioinformatics/btp696.
Parmesan, C., Ryrholm, N., Stefanescu, C., Hill, J. K., Thomas, C. D., Descimon, H., et al. (1999). Poleward shifts in geographical ranges of butterfly species associated with regional warming. Nature, 399, 579–583.
Peakall, R., & Smouse, P. E. (2006). genalex 6: genetic analysis in Excel. Population genetic software for teaching and research. Molecular Ecology Notes, 6(1), 288–295. doi:10.1111/j.1471-8286.2005.01155.x.
Petit, R. J., Aguinagalde, I., de Beaulieu, J.-L., Bittkau, C., Brewer, S., Cheddadi, R., et al. (2003). Glacial refugia: hotspots but not melting pots of genetic diversity. Science (New York, N.Y.), 300(5625), 1563–1565. doi:10.1126/science.1083264.
Pinceel, J., Jordaens, K., Pfenninger, M., & Backeljau, T. (2005). Rangewide phylogeography of a terrestrial slug in Europe: evidence for Alpine refugia and rapid colonization after the Pleistocene glaciations. Molecular Ecology, 14(4), 1133–1150. doi:10.1111/j.1365-294X.2005.02479.x.
Pritchard, J. K., Stephens, M., & Donnelly, P. (2000). Inference of population structure using multilocus genotype data. Genetics, 155(2), 945–959.
QGIS Development Team. (2014). Quantum GIS Geographic Information System. Open Source Geospatial Foundation Project. http://qgis.osgeo.org.
R Core Team. (2014). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL http://www.R-project.org/.
Ryder, O. A. (1986). Species conservation and systematics: the dilemma of subspecies. Trends in Ecology and Evolution, 1(1), 9–10. doi:10.1016/0169-5347(86)90059-5.
Schmitt, T., Giessl, A., & Seitz, A. (2002). Postglacial colonisation of western Central Europe by Polyommatus coridon (Poda 1761) (Lepidoptera: Lycaenidae): evidence from population genetics. Heredity, 88(1), 26–34. doi:10.1038/sj.hdy.6800003.
Schmitt, T., & Hewitt, G. M. (2004). The genetic pattern of population threat and loss: a case study of butterflies. Molecular Ecology, 13(1), 21–31.
Schmitt, T., Rákosy, L., Abadjiev, S., & Müller, P. (2007). Multiple differentiation centres of a non-Mediterranean butterfly species in south-eastern Europe. Journal of Biogeography, 34(6), 939–950. doi:10.1111/j.1365-2699.2006.01684.x.
Schmitt, T., & Seitz, A. (2001). Intraspecific allozymatic differentiation reveals the glacial refugia and the postglacial expansions of European Erebia medusa (Lepidoptera: Nymphalidae). Biological Journal of the Linnean Society, 74(4), 429–458. doi:10.1111/j.1095-8312.2001.tb01404.x.
Schmitt, T., & Varga, Z. n. (2012). Extra-Mediterranean refugia: the rule and not the exception? Frontiers in Zoology, 9(1), 22–33. doi:10.1186/1742-9994-9-22.
Stewart, J. R., & Lister, A. M. (2001). Cryptic northern refugia and the origins of the modern biota. Trends in Ecology & Evolution, 16(11), 608–613. doi:10.1016/S0169-5347(01)02338-2.
Stinchcombe, J. R., & Hoekstra, H. E. (2007). Combining population genomics and quantitative genetics: finding the genes underlying ecologically important traits. Heredity, 100(2), 158–170. doi:10.1038/sj.hdy.6800937.
Taberlet, F., Wust-Saucy, C., & Taberlet. (1998). Comparative phylogeography and postglacial colonization routes in Europe. Molecular Ecology, 7(4), 453–464.
Tamura, K., Stecher, G., Peterson, D., Filipski, A., & Kumar, S. (2013). MEGA6: Molecular Evolutionary Genetics Analysis Version 6.0. Molecular Biology and Evolution, 30(12), 2725–2729. doi:10.1093/molbev/mst197.
Thomas, J. A. (2005). Monitoring change in the abundance and distribution of insects using butterflies and other indicator groups. Philosophical Transactions of the Royal Society of London B: Biological Sciences, 360(1454), 339–357. doi:10.1098/rstb.2004.1585.
Todisco, V., Gratton, P., Cesaroni, D., & Sbordoni, V. (2010). Phylogeography of Parnassius apollo: hints on taxonomy and conservation of a vulnerable glacial butterfly invader. Biological Journal of the Linnean Society, 101(1), 169–183. doi:10.1111/j.1095-8312.2010.01476.x.
Ursenbacher, S., Carlsson, M., Helfer, V., Tegelström, H., & Fumagalli, L. (2006). Phylogeography and Pleistocene refugia of the adder (Vipera berus) as inferred from mitochondrial DNA sequence data. Molecular Ecology, 15(11), 3425–3437. doi:10.1111/j.1365-294X.2006.03031.x.
Välimäki, P., & Itämies, J. (2003). Migration of the clouded Apollo butterfly Parnassius mnemosyne in a network of suitable habitats—effects of patch characteristics. Ecography, 26(5), 679–691. doi:10.1034/j.1600-0587.2003.03551.x.
van Swaay, C. A. M. (1990). An assessment of the changes in butterfly abundance in The Netherlands during the 20th Century. Biological Conservation, 52(4), 287–302. doi:10.1016/0006-3207(90)90073-X.
van Swaay, C. M., & Warren, M. (1999). Red Data Book of European Butterflies (Rhopalocera). Council of Europe.
van Swaay, C. M., Cuttelod, A., Collins, S., Maes, D., Lopez Munguira, M., Šašić, M., et al. (2010). European red list of Butterflies. http://library.wur.nl/WebQuery/clc/1939351. http://library.wur.nl/WebQuery/clc/1939351. Accessed 16 December 2015
van Swaay, C. M., Warren, S., & Löis, G. (2006). Biotope use and trends of European butterflies. Journal of Insect Conservation, 10, 189–209.
Varga, Z. (1977). Das Prinzip der areal-analytischen Methode in der Zoogeographie und die Faunenelemente-Einteilung der europäischen Tagschmetterlinge (Lep.: Diurna). Acta Zoologica Debrecina, 14, 223–285.
Vila, M., Lundhagen, A. C., Thuman, K. A., Stone, J. R., & Bjorklund, M. (2006). A new conservation unit in the butterfly Erebia triaria (Nymphalidae) as revealed by nuclear and mitochondrial markers. Annales Zoologici Fennici, 43, 72–79.
Wahberg, N., & Saccheri, I. (2007). The effects of Pleistocene glaciations on the Phylogeography of Melitaea cinxia (Lepidoptera: Nymphalidae). European Journal of Entomology, 104, 675–684.
Wahlberg, N., & Wheat, C. W. (2008). Genomic outposts serve the phylogenomic pioneers: designing novel nuclear markers for genomic DNA extractions of Lepidoptera. Systematic Biology, 57(2), 231–242. doi:10.1080/10635150802033006.
Waples, R. S. (1991). Pacific salmon, oncorhynchus spp., and the definition of…. Marine Fisheries Review, 53(3), 11.
Warren, M. S., & Key, R. S. (1991). Woodlands: past, present and potential for insects. In The conservation of insects and their habitats. (pp. 155–212.). London: Academic Press.
Acknowledgments
The study was financed by the NKFP-3 B/023/2004 and OTKA K84071 projects. JB was supported by János Bolyai Scholarship of the Hungarian Academy of Sciences. The technical assistance of V. Mester in the electrophoretic work is very much respected. A. Crisan, L. Peregovits, L. Rákosy, S. Szabó and P. Kozma contributed to the sampling at several sites. The support of the Nature Conservation Authorities of Hungary is highly appreciated.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pecsenye, K., Tóth, J.P., Bereczki, J. et al. Genetic structure of Parnassius mnemosyne (Lepidoptera: Papilionidae) populations in the Carpathian Basin. Org Divers Evol 16, 809–819 (2016). https://doi.org/10.1007/s13127-016-0281-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13127-016-0281-7