Abstract
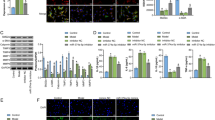
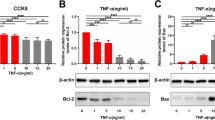
Moyamoya disease (MMD) is characterized by frequent migration and phenotypic transformation of vascular smooth muscle cells (VSMCs) in the intima layer of blood vessels. However, the underlying mechanism is unclear. Toll-like receptor (TLR) 7 is abundantly expressed in smooth muscle cells (SMCs) in multiple vascular diseases, which might be linked to the disease-associated vascular remodeling. In the present study, the expression of TLR7 in MMD vessels was examined using the superficial temporal artery (STA) and middle cerebral artery (MCA) from MMD patients. Furthermore, the effect of TLR7 activation on the VSMC phenotype switch in vitro and vascular remodeling in vivo was assessed using a 9.4Tesla MRI. Our results demonstrated that the TLR7 and microRNA Let-7c expression are upregulated in VSMCs and the plasma of MMD patients, respectively. Additionally, TLR7 stimulation by Let-7c or Imiquimod induces a synthetic phenotype switch in VSMCs. Mechanistic studies revealed that Akt/mTOR signaling is responsible for this TLR-induced VSMC phenotypic switch. The Let-7c or Imiquimod treatment also resulted in reduced blood flow of internal carotid arteries (ICAs) in an in vivo model, while TLR7 inhibition attenuated the ICA stenosis. Besides, Let-7c was also found to be elevated in the hypoxic endothelial cells. Taken together, our study demonstrates that Let-7c released by endothelial cells under hypoxic conditions may activate TLR7 on VSMCs, ultimately leading to the phenotype switch and vascular wall remodeling. These findings thus elucidate the putative mechanisms underlying progressive stenosis of blood vessels in MMD and provide prospective therapeutic targets for further exploration.






Similar content being viewed by others
Data Availability
The data used and/or analyzed in the current study are available from the corresponding author upon reasonable request.
References
Sekhon J, Rogers B, Montalvan V. Moyamoya disease: a story of multiple strokes. Chest. 2020;158:A2566.
Fox BM, Dorschel KB, Lawton MT, Wanebo JE. Pathophysiology of vascular stenosis and remodeling in moyamoya disease. Front Neurol. 2021;12:661578.
Moyamoya angiopathy and developmental abnormalities: a neurosurgical perspective. Dev Med Child Neurol. 2021:131.
Berry JA, Cortez V, Toor H, Saini H, Siddiqi J. Moyamoya: An update and review. Cureus. 2020;12(10):e10994.
Mertens R, Graupera M, Gerhardt H, Bersano A, Tournier-Lasserve E, Mensah MA, et al. The genetic basis of moyamoya disease. Transl Stroke Res. 2022:25–45.
Mineharu Y, Miyamoto S. RNF213 and GUCY1A3 in moyamoya disease: key regulators of metabolism, inflammation, and vascular stability. Front Neurol. 2021;12:687088.
Bang OY, Chung JW, Kim DH, Won HH, Yeon JY, Ki CS, et al. Moyamoya disease and spectrums of RNF213 vasculopathy. Transl Stroke Res. 2020:580–9.
Bang OY, Fujimura M, Kim SK. The pathophysiology of moyamoya disease: An update. J Stroke. 2016:12–20.
Huang S, Guo ZN, Shi M, Yang Y, Rao M, et al. Int J Stroke. 2017:246–53.
Cuoco JA, Busch CM, Klein BJ, Benko MJ, Stein R, Nicholson AD, et al. ACTA2 cerebral arteriopathy: not just a puff of smoke. Cerebrovasc Dis. 2018:159–69.
Wang G, Jacquet L, Karamariti E, Xu Q. Origin and differentiation of vascular smooth muscle cells. J Physiol. 2015;593:3013–30.
Chakraborty R, Chatterjee P, Dave JM, Ostriker AC, Greif DM, Rzucidlo EM, et al. Targeting smooth muscle cell phenotypic switching in vascular disease. JVS Vasc Sci. 2021;2:79–94.
Sorokin V, Vickneson K, Kofidis T, Woo CC, Lin XY, Foo R, et al. Role of vascular smooth muscle cell plasticity and interactions in vessel wall inflammation. Front Immunol. 2020;11:599415.
Badran A, Nasser SA, Mesmar J, El-Yazbi AF, Bitto A, Fardoun MM, et al. Reactive oxygen species: Modulators of phenotypic switch of vascular smooth muscle cells. Int J Mol Sci. 2020:1–21.
Shi J, Yang Y, Cheng A, Xu G, He F. Metabolism of vascular smooth muscle cells in vascular diseases. Am J Physiol - Hear Circ Physiol. 2020:H613–31.
Lin CJ, Hunkins BM, Roth RA, Lin CY, Wagenseil JE, Mecham RP. Vascular smooth muscle cell subpopulations and neointimal formation in mouse models of elastin insufficiency. Arterioscler Thromb Vasc Biol. 2021;41:2890–905.
Rinaldi C, Donato L, Alibrandi S, Scimone C, D’angelo R, Sidoti A. Oxidative stress and the neurovascular unit. Life. 2021:767.
Huang Y, Zhang Q, Lubas M, Yuan Y, Yalcin F, Efe IE, et al. Synergistic toll-like receptor 3/9 signaling affects properties and impairs glioma-promoting activity of microglia. J Neurosci. 2020;40:6428–43.
Erridge C, Burdess A, Jackson AJ, Murray C, Riggio M, Lappin D, et al. Vascular cell responsiveness to toll-like receptor ligands in carotid atheroma. Eur J Clin Invest. 2008;38:713–20.
Zhang L, Zeng XX, Li YM, Chen SK, Tang LY, Wang N, et al. Keratin 1 attenuates hypoxic pulmonary artery hypertension by suppressing pulmonary artery media smooth muscle expansion. Acta Physiol. 2021;231:e13558.
Buonfiglioli A, Efe IE, Guneykaya D, Ivanov A, Huang Y, Orlowski E, et al. Let-7 microRNAs regulate microglial function and suppress glioma growth through Toll-like receptor 7. Cell Rep. 2019;29:3460-3471.e7.
Lehmann SM, Krüger C, Park B, Derkow K, Rosenberger K, Baumgart J, et al. An unconventional role for miRNA: Let-7 activates Toll-like receptor 7 and causes neurodegeneration. Nat Neurosci. 2012;15:827–35.
Zhao S, Gong Z, Zhang J, Xu X, Liu P, Guan W, et al. Elevated serum microRNA Let-7c in moyamoya disease. J Stroke Cerebrovasc Dis. 2015;24:1709–14.
Roberts JM, Maniskas ME, Fraser JF, Bix GJ. Internal carotid artery stenosis: A novel surgical model for moyamoya syndrome. PLoS One. 2018;13:e0191312.
Ifuku M, Buonfiglioli A, Jordan P, Lehnardt S, Kettenmann H. TLR2 controls random motility, while TLR7 regulates chemotaxis of microglial cells via distinct pathways. Brain Behav Immun. 2016;58:338–47.
Huang CX, Chen N, Wu XJ, He Y, Huang CH, Liu H, et al. Zebrafish let-7b acts downstream of hypoxiainducible factor-1α to assist in hypoxia-mediated cell proliferation and cell cycle regulation. Life Sci. 2017;171:21–9.
Gibadulinova A, Bullova P, Strnad H, Pohlodek K, Jurkovicova D, Takacova M, et al. CAIX-mediated control of LIN28/let-7 axis contributes to metabolic adaptation of breast cancer cells to hypoxia. Int J Mol Sci. 2020;21:1–18.
Rombouts KB, van Merrienboer TAR, Ket JCF, Bogunovic N, van der Velden J, Yeung KK. The role of vascular smooth muscle cells in the development of aortic aneurysms and dissections. Eur J Clin Invest. 2022:e13697.
Green ID, Liu R, Wong JJL. The expanding role of alternative splicing in vascular smooth muscle cell plasticity. Int J Mol Sci. 2021:10213.
Chi H, Li C, Zhao FS, Zhang L, Ng TB, Jin G, et al. Anti-tumor activity of toll-like receptor 7 agonists. Front Pharmacol. 2017:304.
Scimone C, Bramanti P, Ruggeri A, Donato L, Alafaci C, Crisafulli C, et al. CCM3/SERPINI1 bidirectional promoter variants in patients with cerebral cavernous malformations: A molecular and functional study. BMC Med Genet. 2016;17:74.
Scimone C, Bramanti P, Ruggeri A, Katsarou Z, Donato L, Sidoti A, et al. Detection of novel mutation in Ccm3 causes familial cerebral cavernous malformations. J Mol Neurosci. 2015;57:400–3.
Li M, Qian M, Kyler K, Xu J. Endothelial–vascular smooth muscle cells interactions in atherosclerosis. Front Cardiovasc Med. 2018:151.
Rodriguez D, Watts D, Gaete D, Sormendi S, Wielockx B. Hypoxia pathway proteins and their impact on the blood vasculature. Int J Mol Sci. 2021:9191.
Acknowledgements
We thank the Laboratory Core Facility of Tongji Hospital of Tongji Medical College, Wuhan, 430030, People’s Republic of China, for technical assistance. We would also like to especially acknowledge Dr. Qing Li for kindly sharing the cells used in the study with us and providing technical assistance. This study was supported by the Natural Science Foundation of Hubei Province (WJ2019Z008) and the Natural Science Foundation of Tongji Hospital (2020JZKT651), People’s Republic of China.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.

Supplementary Figure 1
qPCR (a) and Westernblot (b) assay was used to determine the transfection efficiency of TLR7 siRNA on VSMCs. (PNG 113 kb)
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ma, X., Huang, Y., He, X. et al. Endothelial Cell–Derived Let-7c-Induced TLR7 Activation on Smooth Muscle Cell Mediate Vascular Wall Remodeling in Moyamoya Disease. Transl. Stroke Res. 14, 608–623 (2023). https://doi.org/10.1007/s12975-022-01088-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12975-022-01088-3