Abstract
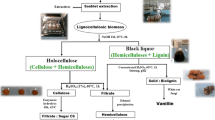
This paper focuses on valorization alternatives of several fractions of new feedstock as Juncus maritimus and Retama raetam through a specific laboratory-scale part of biorefinery way implying organosolv and chemical pretreatments as well as cellulose saccharification and alcoholic fermentation to obtain acceptable yields. After organosolv pretreatment, the obtained cellulosic fraction was used as substrate for cellulase catalyzed-saccharification followed by fermentation using Saccharomyces cerevisiae for ethanol production. The maximum obtained ethanol yields were (41.7 ± 0.85)% and (40.57 ± 1.18)% (g ethanol g−1 glucose) using respectively J. maritimus and R. raetam. The liquid hemicellulosic fraction collected after pretreatment was used as a carbon source for Aspergillus niger culture in order to produce xylanolytic enzymes. The highest xylanase activity obtained was 0.44 U mL−1 using the hemicellulosic fraction of J. maritimus. When using chemical pretreatment, the cellulose obtained in the solid fraction was converted into ethanol with yields reaching 37.28 ± 0.81% and 38.35 ± 1.76% respectively from R. reatam and J. maritimus biomasses. The lignin from solid phase separated from cellulose was analyzed by Fourier transform infrared spectroscopy (FT-IR). It shows potential interest for use in aromatic chemicals production.





Similar content being viewed by others
References
Pinatti, D.G., Conte, R.A., Soares, A.G., Pereia, M.L.G., Romão, E.L., Ferreira, J.C., Oliveira, I., Marton, L.F.M.: Biomass refinery as a renewable complement to the petroleum refinery. Int. J. Chem. Reactor Eng. 8, 1–17 (2010)
Kamm, B., Kamm, M., Gruber, P.R., Kromus, S.: Biorefinery systems. An overview. In: Kamm, B., Gruber, P.R., Kamm, M. (eds.) Biorefineries. Industrial processes and products. Statu quo and future directions, vol. 1, pp. 3–40. Wiley-VCH Verlag GmbH & Co, Weinheim (2006)
Carvalheiro, F., Duarte, L.C., Girio, F.M.: Hemicellulose biorefineries: a review on biomass pretreatments. J. Sci. Ind. Res. 67(11), 849–864 (2008)
Zhang, Y.H.P.: Reviving the carbohydrate economy via multi-product lignocellulose biorefinerie. J. Ind. Microbiol. Biotechnol. 35(5), 367–375 (2008)
Cheng, S.M., Zhu, S.D.: Lignocellulosic feedstock biorefinery: the future of the chemical and energy industry. Bioresources. 4(2), 456–457 (2009)
Luo, L., Van Der Voet, E., Huppes, G.: Biorefining of lignocellulosic feedstock: technical, economic and environmental considerations. Bioresour.Technol. 101(13), 5023–5032 (2010)
Kumar, M.N.S., Mohanty, A.K., Erickson, L., Misra, M.: Lignin and its applications with polymers. J. Biobased Mater. Bioenergy 3(1), 1–24 (2009)
Mosier, N., Wyman, C., Dale, B., Elander, R., Lee, Y.Y., Holtzapple, M., Ladisch, M.: Features of promising technologies for pretreatment of lignocellulosic biomass. Bioresour.Technol. 96(6), 673–686 (2005)
Fengel, D., Wegener, G.: Wood: chemistry, ultrastructure and reactions. Walterde Gruyter Publisher, Berlin (1984)
Wild, P.J., Huijgen, W.J.J., Heeres, H.J.: Pyrolysis of wheat straw-derived organosolv lignin. J. Anal. Appl. Pyrol. 93, 95–103 (2012)
Ksouri, R., Megdiche, W., Koyro, H. W., Abdelly, C.: Responses of halophytes to environmental stresses with special emphasis to salinity advances in botanical research. Adv. Bot. Res. 53, 117–145 (2010)
Pasha, C., Valli, N., Rao, L.V.: Lantana camara for fuel ethanol production using thermotolerant yeast. Lett. Appl. Microbiol. 44, 666–672 (2007)
Sarkar, N., Ghosh, S.K., Bannerjee, S., Aikat, K.: Bioethanol production from agricultural wastes: an overview. Renewable Energy 37, 19–27 (2012)
Dagnino, E.P., Chamorro, E.R., Romano, S.D., Felissia, F.E., Area, M.C.: Optimization of the acid pretreatment of rice hulls to obtain fermentable sugars for bioethanol production. Ind. Crops Prod. 42, 363–368 (2013)
Hendriks, A., Zeeman, G.: Pretreatments to enhance the digestibility of lignocellulosic biomass. Bioresour. Technol. 100(1), 10–18 (2009)
Tao, L., Aden, A., Elander, R., Pallapolu, V., Lee, Y., Garlock, R., Balan, V., Dale, B., Kim, Y., Mosier, N., Ladisch, M., Falls, M., Holtzapple, M., Sierra, R., Shi, J., Ebrik, M., Red-mond, T., Yang, B., Wyman, C., Hames, B., Thomas, S., Warner, R.: Process and technoeconomic analysis of leading pretreatment technologies for lignocellulosic ethanol production using switchgrass. Bioresour. Technol. 102, 11105–11114 (2011)
Alvira, P., Tomás-Pejó, E., Ballesteros, M., Negro, M.J.: Pretreatment technologies for an efficient bioethanol production process based on enzymatic hydrolysis: a review. Bioresour. Technol. 101(13), 4851–4861 (2010)
Pan, X., Arato, C., Gilkes, N., Gregg, D., Mabee, W., Pye, K., Xiao, Z., Zhang, X., Saddler, J.: Biorefining of softwoods using ethanol organosolv pulping: preliminary evaluation of process streams for manufacture of fuel-grade ethanol and coproducts. Biotechnol.Bioeng. 90(4), 473–481 (2005)
Hoopman, T., Birch, G., Serghat, S., Portmann, M.O., Mathlouthi, M.: Solute-solvent interactions and the sweet taste of small carbohydrates. Part II: sweetness intensity and persistence in ethanol-water mixtures. Food Chem. 46, 147–153 (1993)
Holladay, J.E., Bozell, J.J., White, J.F., Johnson, D.: Top value added chemicals from biomass, volume II—results of screening for potential candidates from biorefinery lignin. Pacific Northwest National Laboratory and the National Renewable Energy Laboratory. Prepared for the US Department of Energy under contract number DE-ACOS-76RL01830 (2007)
Goering, H.K., Van Soest, P.J.: Forage fiber analysis (apparatus, reagents, procedures and some applications), agricultural hand book 379, 1737–1741 (1970)
Miller, G.L.: Use dinitrosalicylic acid reagent for the determination of reducing sugars. Anal. Chem. 31(3), 426–429 (1959)
Smichi, N., Messaoudi, Y., Moujahed, N., Gargouri, M.: Ethanol production from halophyte Juncus maritimus using freezing and thawing biomass pretreatment. Renewable Energy (2015)
Aita, G., Salvi, D.: Lignocellulose as a source for fuels and chemicals. LaAgr 52(4), 12–13 (2009)
Perez, L., Teymouri, F., Alizadeh, H., Dale, B.E.: Understanding factors that limit enzymatic hydrolysis of biomass :characterization of pretreated corn stover. Appl. Biochem. Biotech. 121(124), 1081–1099 (2005)
Smichi, N., Messaoudi, Y., Ksouri, R., Abdelly, C., Gargouri, M.: Pretreatment and enzymatic saccharification of new phytoresource for bioethanol production from halophyte species. Renewable Energy 63, 544–549 (2014)
Chen, Y., Stevens, M.A., Zhu, Y., Holmes, J., Moxley, G., Xu, H.: Reducing acid in dilute acid pretreatment and the impact on enzymatic saccharification. J. Ind. Microbiol. Biot. 39(5), 691–700 (2012)
Zhang, Y.H.P, Ding, S.Y., Mielenz, J.R., Cui, J.B., Elander, R.T., Laser, M., Himmel, M.E., McMillan, J.R., Lynd, L.R.: Fractionating recalcitrant lignocellulose at modest reaction conditions. Biotechnol.Bioeng. 97(2), 214–223 (2007)
Shigemasa, Y., Kishimoto, Y., Sashiwa, H., Saimoto, H.: Dissolution of cellulose in dimethyl sulfoxide. Effect of thiamine hydrochloride. Polym. J. 22, 1101–1103 (1990)
Heinze, T., Dicke, R., Koschella, A., Henning Kull, A., Klohr, E.A., Koch, W.: Effective preparation of cellulose derivatives in a new simple cellulose solvent. Macromol. Chem. Phys. 201(6), 627–631 (2000)
Boullagui, H., Touhami, Y., Hanafi, N., Ghariani, A., Hamdi, M.: Performance comparison between three technologies for continous ethanol production from molasses. Biomass Bioenergy 48, 25–32 (2013)
Yoswathana, N., Phuriphipat, P., Treyawutthiwat, P., Eshtiaghi, M.N.: Bioethanol production from rice straw. Energy Res. J. 1(1), 26–31 (2010)
Hamdy, M.K., Kim, K., Rudtke, C.A.: Continuous ethanol production by yeast immobilized on to channeled alumina beads. Biomass 21(3), 189–206 (1990)
Novozymes CellicR CTec2 and HTec2 – Enzymes for hydrolysis of lignocellulosic—Application sheet
Mamma, D., Christakopoulos, P., Koullas, D., Kekos, D., Macris, B.J., Koukios, E.: An alternative approach to the bioconversion of sweet sorghum carbohydrates to ethanol. Biomass Bioenergy 8(2), 99–103 (1995)
Sant’Ana da Silva, A., Inoue, H., Endo, T., Yano, S., Bon, E.P.: Milling pretreatment of sugarcane bagasse and straw for enzymatic hydrolysis and ethanol fermentation. Bioresour. Technol. 101(19), 7402–7409 (2010)
Izidoro, S.C., Knob, A.: Production of xylanases by an A. niger strain in wastes grain. Acta Sci. Biol. Sci. 36(3), 313–319 (2014)
Ahmed, S., Imdad, S.S., Jamil, A.: Comparative study for the kinetics of extracellular xylanases from Trichoderma harzianum and Chaetomium thermophilum. Eletronic Journal of Microbiology. 15(3), 0717–3458 (2012)
Knob, A., Carmona, E.C.: Xylanase production by Penicillium sclerotiorum and its characterization. WASJ 4(2), 277–283 (2008)
Siedenberg, D., Gerlach, S.R., Schugerl, K., Giueppin, M.L.F., Hunik, J.: Production of xylanase by Aspaergillus awamori on synthetic medium in shake flask cultures. Process Biochem. 33(4), 429–433 (1997)
Sonia, K.G., Chadha, B.S., Saini, H.S.: Sorghum straw for xylanase hyper-production by Thermomyces lanuginosus (D2W3) under solid state fermentation. Bioresour.Technol. 96, 1561–1569 (2005)
Thygeson, A., Thomson, A.B., Schmidt, A.S., Jorgenson, H., Olsson, L.: Production of cellulose and hemicellulose degrading enzymes by filamentous fungus cultivated on wet oxidized wheat straw. Enzyme Microb. Technol. 32, 606–615 (2003)
Shah, A.R., Shah, R.K., Madamwar, D.: Improvement of the quality of whole wheat bread by supplementation of xylanase from Aspergillus foetidus. Bioresour. Technol. 97, 2047–2053 (2006)
Polizeli, M.L.T.M., Rizzatti, A.C.S., Monti, R., Terenzi, H.F., Jorge, J.A., Amorim, D.S.: Xylanases from fungi: properties and industrial applications. Appl. Microbiol. Biotechnol. 67, 577–591 (2005)
Cpeljnik, T., Krizaj, I., Marinsek-Logar, R.: Isolation and characterization of the Pseudo butyriovibrio xylanivorans Mz5T xylanase XynT—the first family 11 endoxylanase from rumen Butyriovibrio-related bacteria. Enzyme Microb. Technol. 34, 219–227 (2004)
Ohara H.: Biorefinery. Appl. Microbiol. Biotechnol. 62, 474–477 (2003)
Beg, Q.K., Kapoor, M., Mahajan, L., Hoondal, G.S.: Microbial xylanases and their industrial applications: a review. Appl. Microbiol. Biotechnol. 56, 326–338 (2001)
Hortling, B., Tarja, T., Kentta, E.: Determination of carboxyland non-conjugated carbonyl groups in dissolved and residual lignins by IR spectroscopy. Holzforschung 51, 405–410 (1997)
Gilarranz, M., Rodrıguez, F., Oliet, M., Garcıa, J., Alonso, V., Phenolic, O.H.: group estimation by FTIP and UV spectroscopy. Application to organosolv lignins. J. Wood Chem. Technol. 21, 387–395 (2001)
Durie, R., Lynch, B., Sternhell, S.: Comparative studies of brown coal and lignin. I. Infra-red spectra. Aust. J. Chem. 13, 156–168 (1960)
Bolker, H.I., Somerville, N.G.: Infrared spectroscopy of lignins. Pulp. Paper. Can. Mag. 64, 187–194 (1963)
Colthup, N., Daly, L., Wiberley, S.: Introduction to infrared and Raman spectroscopy. Academic Press Limited, London (1990)
Xu, F., Sun, J., Sun, R., Fowler, P., Baird, M.S.: Comparative study of organosolv lignins from wheat straw. Ind. Crops Prod. 23, 180–193 (2006)
Faix, O.: Classification of lignins from different botanical origins by FT-IR spectroscopy. Holzforschung 45, 21–27 (1991)
Haveren, J., Scott, E.L., Sanders, J.P.M.: Review: bulk chemicals from biomass. Biofuels Bioprod. Biorefin. 2, 41–57 (2008)
Acknowledgements
The authors would like to thank the financial support provided by the Engineering Procurement & Project Management (EPPM).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Smichi, N., Messaoudi, Y. & Gargouri, M. Lignocellulosic Biomass Fractionation: Production of Ethanol, Lignin and Carbon Source for Fungal Culture. Waste Biomass Valor 9, 947–956 (2018). https://doi.org/10.1007/s12649-017-9859-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12649-017-9859-3