Abstract
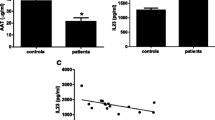
Amyotrophic lateral sclerosis (ALS) is a progressive and fatal motor neuron disease of unknown pathogenesis. The kynurenine pathway (KP), activated during neuroinflammation, is emerging as a possible contributory factor in ALS. The KP is the major route for tryptophan (TRP) catabolism. The intermediates generated can be either neurotoxic, such as quinolinic acid (QUIN), or neuroprotective, such as picolinic acid (PIC), an important endogenous chelator. The first and inducible enzyme of the pathway is indoleamine 2,3-dioxygenase (IDO). The present study aimed to characterize the expression of the KP in cerebrospinal fluid (CSF), serum and central nervous system (CNS) tissue of ALS patients. Using high performance liquid chromatography, we analysed the levels of TRP and kynurenine (KYN), and, with gas chromatography/mass spectrometry, the levels of PIC and QUIN, in the CSF and serum of ALS patients and control subjects. Immunohistochemistry was employed to determine the expression of QUIN, IDO and human leukocyte antigen-DR (HLA-DR) in sections of brain and spinal cord from ALS patients. There were significantly increased levels of CSF and serum TRP (P < 0.0001), KYN (P < 0.0001) and QUIN (P < 0.05) and decreased levels of serum PIC (P < 0.05) in ALS samples. There was a significant increase in activated microglia expressing HLA-DR (P < 0.0001) and increased neuronal and microglial expression of IDO and QUIN in ALS motor cortex and spinal cord. We show the presence of neuroinflammation in ALS and provide the first strong evidence for the involvement of the KP in ALS. These data point to an inflammation-driven excitotoxic-chelation defective mechanism in ALS, which may be amenable to inhibitors of the KP.








Similar content being viewed by others
References
Beninger RJ, Colton AM, Ingles JL, Jhamandas K, Boegman RJ (1994) Picolinic acid blocks the neurotoxic but not the neuroexcitant properties of quinolinic acid in the rat brain: evidence from turning behaviour and tyrosine hydroxylase immunohistochemistry. Neuroscience 61:603–612
Bensimon G, Lacomblez L, Meininger V, ALS/Riluzole Study Group (1994) A controlled trial of riluzole in amyotrophic lateral sclerosis. N Engl J Med 330:585–591
Blasi E, Mazzolla R, Pitzurra L, Barluzzi R, Bistoni F (1993) Protective effect of picolinic acid on mice intracerebrally infected with lethal doses of Candida albicans. Antimicrob Agents Chemother 37:2422–2426
Bosco MC, Rapisarda A, Massazza S, Melillo G, Young H, Varesio L (2000) The tryptophan catabolite picolinic acid selectively induces the chemokines macrophage inflammatory protein-1 alpha and -1 beta in macrophages. J Immunol 164:3283–3291
Brooks BR (2009) Managing amyotrophic lateral sclerosis: slowing disease progression and improving patient quality of life. Ann Neurol 65:S17–S23
Brooks BR, Miller RG, Swash M, Munsat TL (2000) El Escorial revisited: revised criteria for the diagnosis of amyotrophic lateral sclerosis. Amyotroph Lateral Scler Other Motor Neuron Disord 1:293–299
Bruijn LI, Houseweart MK, Kato S, Anderson KL, Anderson SD, Ohama E, Reaume AG, Scott RW, Cleveland DW (1998) Aggregation and motor neuron toxicity of an ALS-linked SOD1 mutant independent from wild-type SOD1. Science 281:1851–1854
Bruijn LI, Miller TM, Cleveland DW (2004) Unraveling the mechanisms involved in motor neuron degeneration in ALS. Annu Rev Neurosci 27:723–749
Cockhill J, Jhamandas K, Boegman RJ, Beninger RJ (1992) Action of picolinic acid and structurally related pyridine carboxylic acids on quinolinic acid-induced cortical cholinergic damage. Brain Res 599:57–63
Feksa LR, Latini A, Rech VC, Feksa PB, Koch GD, Amaral MF, Leipnitz G, Dutra-Filho CS, Wajner M, Wannmacher CM (2008) Tryptophan administration induces oxidative stress in brain cortex of rats. Metab Brain Dis 23:221–233
Fernandez-Pol JA, Klos DJ, Hamilton PD (2001) Antiviral, cytotoxic and apoptotic activities of picolinic acid on human immunodeficiency virus-1 and human herpes simplex virus-2 infected cells. Anticancer Res 21:3773–3776
Forrest CM, Mackay GM, Stoy N, Egerton M, Christofides J, Stone TW, Darlington LG (2004) Tryptophan loading induces oxidative stress. Free Radic Res 38:1167–1171
Frumento G, Rotondo R, Tonetti M, Damonte G, Benatti U, Ferrara GB (2002) Tryptophan-derived catabolites are responsible for inhibition of T and natural killer cell proliferation induced by indoleamine 2,3-dioxygenase. J Exp Med 196:459–468
Fukui S, Schwarcz R, Rapoport SI, Takada Y, Smith QR (1991) Blood-brain barrier transport of kynurenines: implications for brain synthesis and metabolism. J Neurochem 56:2007–2017
Guillemin GJ, Smith DG, Kerr SJ, Smythe G, Kapoor V, Armati PJ, Brew BJ (2000) Characterisation of kynurenine pathway metabolism in human astrocytes and implications in neuropathogenesis. Redox Rep 5:108–111
Guillemin GJ, Kerr SJ, Smyth PG, Smith DG, Kapoor V, Armati PJ, Croitoru J, Brew BJ (2001) Kynurenine pathway metabolism in human astrocytes: a paradox for neuronal protection. J Neurochem 78:842–853
Guillemin GJ, Croitoru-Lamoury J, Dormont D, Armati PJ, Brew BJ (2003) Quinolinic acid upregulates chemokine production and chemokine receptor expression in astrocytes. Glia 41:371–381
Guillemin GJ, Kerr SJ, Brew BJ (2004) Involvement of quinolinic acid in AIDS dementia complex. Neurotox Res 7:103–124
Guillemin GJ, Meininger V, Brew BJ (2005a) Implications for the kynurenine pathway and quinolinic acid in amyotrophic lateral sclerosis. Neurodegener Dis 2:166–176
Guillemin GJ, Brew BJ, Noonan CE, Takikawa O, Cullen KM (2005b) Indoleamine 2,3 dioxygenase and quinolinic acid immunoreactivity in Alzheimer’s disease hippocampus. Neuropathol Appl Neurobiol 31:395–404
Guillemin GJ, Cullen KM, Lim CK, Smythe GA, Garner B, Kapoor V, Takikawa O, Brew BJ (2007) Characterization of the kynurenine pathway in human neurons. J Neurosci 27:12884–12892
Hartai Z, Klivenyi P, Janaky T, Penke B, Dux L, Vecsei L (2005) Kynurenine metabolism in plasma and in red blood cells in Parkinson’s disease. J Neurol Sci 239:31–35
Heyes MP, Achim CL, Wiley CA, Major EO, Saito K, Markey SP (1996) Human microglia convert l-tryptophan into the neurotoxin quinolinic acid. Biochem J 320(Pt 2):595–597
Ilzecka J, Kocki T, Stelmasiak Z, Turski WA (2003) Endogenous protectant kynurenic acid in amyotrophic lateral sclerosis. Acta Neurol Scand 107:412–418
Jhamandas K, Boegman RJ, Beninger RJ, Bialik M (1990) Quinolinate-induced cortical cholinergic damage: modulation by tryptophan metabolites. Brain Res 529:185–191
Jhamandas KH, Boegman RJ, Beninger RJ, Flesher S (1998) Role of zinc in blockade of excitotoxic action of quinolinic acid by picolinic acid. Amino Acids 14:257–261
Kalisch BE, Jhamandas K, Boegman RJ, Beninger RJ (1994) Picolinic acid protects against quinolinic acid-induced depletion of NADPH diaphorase containing neurons in the rat striatum. Brain Res 668:1–8
Lacomblez L, Bensimon G, Leigh PN, Guillet P, Meininger V, Amyotrophic Lateral Sclerosis/Riluzole Study Group II (1996) Dose-ranging study of riluzole in amyotrophic lateral sclerosis. Lancet 347:1425–1431
Leuthauser SW, Oberley LW, Oberley TD (1982) Antitumor activity of picolinic acid in CBA/J mice. J Natl Cancer Inst 68:123–126
Lyons TJ, Liu H, Goto JJ, Nersissian A, Roe JA, Graden JA, Cafe C, Ellerby LM, Bredesen DE, Gralla EB, Valentine JS (1996) Mutations in copper-zinc superoxide dismutase that cause amyotrophic lateral sclerosis alter the zinc binding site and the redox behavior of the protein. Proc Natl Acad Sci USA 93:12240–12244
Matsuo H, Tsukada S, Nakata T, Chairoungdua A, Kim DK, Cha SH, Inatomi J, Yorifuji H, Fukuda J, Endou H, Kanai Y (2000) Expression of a system L neutral amino acid transporter at the blood-brain barrier. Neuroreport 11:3507–3511
McGeer PL, McGeer EG (2002) Inflammatory processes in amyotrophic lateral sclerosis. Muscle Nerve 26:459–470
Muller AJ, DuHadaway JB, Donover PS, Sutanto-Ward E, Prendergast GC (2005) Inhibition of indoleamine 2,3-dioxygenase, an immunoregulatory target of the cancer suppression gene Bin1, potentiates cancer chemotherapy. Nat Med 11:312–319
Munn DH, Zhou M, Attwood JT, Bondarev I, Conway SJ, Marshall B, Brown C, Mellor AL (1998) Prevention of allogeneic fetal rejection by tryptophan catabolism. Science 281:1191–1193
Nagano S, Satoh M, Sumi H, Fujimura H, Tohyama C, Yanagihara T, Sakoda S (2001) Reduction of metallothioneins promotes the disease expression of familial amyotrophic lateral sclerosis mice in a dose-dependent manner. Eur J Neurosci 13:1363–1370
Owe-Young R, Webster NL, Mukhtar M, Pomerantz RJ, Smythe G, Walker D, Armati PJ, Crowe SM, Brew BJ (2008) Kynurenine pathway metabolism in human blood-brain barrier cells: implications for immune tolerance and neurotoxicity. J Neurochem 105:1346–1357
Pardridge WM, Fierer G (1990) Transport of tryptophan into brain from the circulating, albumin-bound pool in rats and in rabbits. J Neurochem 54:971–976
Perkins MN, Stone TW (1982) An iontophoretic investigation of the actions of convulsant kynurenines and their interaction with the endogenous excitant quinolinic acid. Brain Res 247:184–187
Rosen DR, Siddique T, Patterson D, Figlewicz DA, Sapp P, Hentati A, Donaldson D, Goto J, O’Regan JP, Deng HX et al (1993) Mutations in Cu/Zn superoxide dismutase gene are associated with familial amyotrophic lateral sclerosis. Nature 362:59–62
Rothstein JD (2009) Current hypotheses for the underlying biology of amyotrophic lateral sclerosis. Ann Neurol 65:S3–S9
Salter M, Pogson CI (1985) The role of tryptophan 2,3-dioxygenase in the hormonal control of tryptophan metabolism in isolated rat liver cells. Effects of glucocorticoids and experimental diabetes. Biochem J 229:499–504
Santamaria A, Galvan-Arzate S, Lisy V, Ali SF, Duhart HM, Osorio-Rico L, Rios C, St’astny F (2001) Quinolinic acid induces oxidative stress in rat brain synaptosomes. Neuroreport 12:871–874
Schrocksnadel K, Wirleitner B, Winkler C, Fuchs D (2006) Monitoring tryptophan metabolism in chronic immune activation. Clin Chim Acta 364:82–90
Shaw PJ (2005) Molecular and cellular pathways of neurodegeneration in motor neurone disease. J Neurol Neurosurg Psychiatry 76:1046–1057
Smith AP, Lee NM (2007) Role of zinc in ALS. Amyotroph Lateral Scler 8:131–143
Smythe GA, Braga O, Brew BJ, Grant RS, Guillemin GJ, Kerr SJ, Walker WD (2002) Concurrent quantification of quinolinic, picolinic, and nicotinic acids using electron-capture negative-ion gas chromatography-mass spectrometry. Anal Biochem 301:21–26
Stone TW, Perkins MN (1981) Quinolinic acid: a potent endogenous excitant at amino acid receptors in CNS. Eur J Pharmacol 72:411–412
Streit WJ (2002) Microglia as neuroprotective, immunocompetent cells of the CNS. Glia 40:133–139
Sullivan SM, Lee A, Bjorkman ST, Miller SM, Sullivan RK, Poronnik P, Colditz PB, Pow DV (2007) Cytoskeletal anchoring of GLAST determines susceptibility to brain damage: an identified role for GFAP. J Biol Chem 282:29414–29423
Takikawa O, Yoshida R, Kido R, Hayaishi O (1986) Tryptophan degradation in mice initiated by indoleamine 2,3-dioxygenase. J Biol Chem 261:3648–3653
Watanabe M, Dykes-Hoberg M, Culotta VC, Price DL, Wong PC, Rothstein JD (2001) Histological evidence of protein aggregation in mutant SOD1 transgenic mice and in amyotrophic lateral sclerosis neural tissues. Neurobiol Dis 8:933–941
Widner B, Leblhuber F, Walli J, Tilz GP, Demel U, Fuchs D (2000) Tryptophan degradation and immune activation in Alzheimer’s disease. J Neural Transm 107:343–353
Widner B, Leblhuber F, Fuchs D (2002) Increased neopterin production and tryptophan degradation in advanced Parkinson’s disease. J Neural Transm 109:181–189
Yoshida R, Hayaishi O (1978) Induction of pulmonary indoleamine 2,3-dioxygenase by intraperitoneal injection of bacterial lipopolysaccharide. Proc Natl Acad Sci USA 75:3998–4000
Zhang D (2007) Early activation of an interferon signaling pathway in a mouse model of amyotrophic lateral sclerosis. J Neurovirol 13:48 (abstract S.85)
Zhang H, Andrekopoulos C, Joseph J, Chandran K, Karoui H, Crow JP, Kalyanaraman B (2003) Bicarbonate-dependent peroxidase activity of human Cu, Zn-superoxide dismutase induces covalent aggregation of protein: intermediacy of tryptophan-derived oxidation products. J Biol Chem 278:24078–24089
Acknowledgements
This study was funded by the Motor Neuron Disease Research Institute Association (Australia). The NSW Tissue Resource Centre is supported by The University of Sydney, Schizophrenia Research Institute, National Institutes on Alcohol Abuse and Alcoholism (NIAAA-grant no: R01AAA01272508), Sydney South Western Area Health Service (SSWAHS).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, Y., Stankovic, R., Cullen, K.M. et al. The Kynurenine Pathway and Inflammation in Amyotrophic Lateral Sclerosis. Neurotox Res 18, 132–142 (2010). https://doi.org/10.1007/s12640-009-9129-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-009-9129-7