Abstract
Background
Both propofol and volatile anesthetics are commonly used for maintenance of anesthesia in patients undergoing neurosurgical procedures. The effects of these two classes of drugs on cerebral hemodynamics have been compared in many clinical trials The objectives of this review were to evaluate the cerebral hemodynamic effects, operative conditions, recovery profiles, postoperative complications, and neurological outcomes of propofol-based vs volatile-based anesthesia for craniotomy.
Methods
MEDLINE®, EMBASE™, Cochrane, and other relevant databases were searched for randomized controlled trials that compared propofol-maintained anesthesia with volatile-maintained anesthesia in adult patients undergoing elective craniotomy. The primary outcome measure was the intraoperative brain relaxation score. Secondary outcome measures included intraoperative cerebral hemodynamics (intracranial pressure [ICP], cerebral perfusion pressure [CPP]), cardiovascular changes, recovery profiles, postoperative complications, and clinical outcomes (neurological morbidity, mortality, quality of life). A meta-analysis was conducted using a random effects model to compare the outcomes of the two anesthetic techniques.
Results
Fourteen studies (1,819 patients) met inclusion criteria and were analyzed. Brain relaxation scores were similar between the two groups after dural opening; however, ICP was lower (weighted mean difference of −5.2 mmHg; 95% confidence interval −6.81 to −3.6) and CPP was higher (weighted mean difference of 16.3 mmHg; 95% confidence interval 12.2 to 20.46) in patients receiving propofol-maintained anesthesia. Postoperative complications and recovery profiles were similar between the two groups, except for postoperative nausea and vomiting being less frequent with propofol-maintained anesthesia. There were inadequate data to perform a meta-analysis on clinical outcome.
Conclusion
Propofol-maintained and volatile-maintained anesthesia were associated with similar brain relaxation scores, although mean ICP values were lower and CPP values higher with propofol-maintained anesthesia. There are inadequate data to compare clinically significant outcomes such as neurological morbidity or mortality.
Résumé
Contexte
Le propofol et les anesthésiques volatils sont des agents fréquemment utilisés pour maintenir l’anesthésie chez les patients subissant des interventions neurochirurgicales. Les effets de ces deux classes de médicaments sur l’hémodynamie cérébrale ont été comparés dans de nombreuses études cliniques. Les objectifs de cette revue étaient d’évaluer les effets sur l’hémodynamie cérébrale, les conditions opératoires, les profils de rétablissement, les complications postopératoires et les pronostics neurologiques de l’anesthésie au propofol comparativement aux agents volatils lors de craniotomie.
Méthode
Nous avons effectué une recherche dans les bases de données MEDLINE®, EMBASE™, Cochrane et les autres bases de données pertinentes pour en extraire les études randomisées contrôlées comparant le propofol aux agents volatils pour le maintien de l’anesthésie chez les patients adultes subissant une craniotomie non urgente. Le critère d’évaluation principal était le score de relaxation cérébrale peropératoire. Les critères d’évaluation secondaires comprenaient l’hémodynamie cérébrale peropératoire (pression intracrânienne [PIC], pression de perfusion cérébrale [PPC]), les changements cardiovasculaires, les profils de rétablissement, les complications postopératoires et les devenirs cliniques (morbidité neurologique, mortalité, qualité de vie). Une méta-analyse a été réalisée en utilisant un modèle à effets aléatoires afin de comparer les pronostics des deux techniques d’anesthésie.
Résultats
Quatorze études (1819 patients) respectaient les critères d’inclusion et ont été analysées. Les scores de relaxation cérébrale étaient semblables dans les deux groupes après ouverture de la dure-mère; toutefois, la PIC était plus basse (différence moyenne pondérée de −5,2 mmHg; intervalle de confiance 95 % −6,81 à −3,6) et la PPC plus élevée (différence moyenne pondérée de 16,3 mmHg; intervalle de confiance 95 % 12,2 à 20,46) chez les patients dont l’anesthésie était maintenue avec du propofol. Les complications postopératoires et les profils de rétablissement étaient semblables dans les deux groupes, à l’exception des nausées et vomissements postopératoires, moins fréquents lorsque l’anesthésie était maintenue au propofol. Les données étaient insuffisantes pour faire une méta-analyse des devenirs cliniques.
Conclusion
Le propofol et les agents volatils comme maintien de l’anesthésie sont associés à des scores de relaxation cérébrale semblables, bien que les valeurs de PIC moyennes aient été plus basses et les valeurs de PPC moyennes plus élevées avec une anesthésie maintenue au propofol. Les données sont insuffisantes pour comparer les devenirs significatifs d’un point de vue clinique tels que la morbidité neurologique ou la mortalité.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
The well-accepted goals for the anesthetic management of patients undergoing neurosurgical procedures include the delivery of smooth and hemodynamically stable anesthesia with provision of good operating conditions and a smooth but rapid emergence that allows for early neurological assessment. The ideal anesthetic agents should maintain cerebral perfusion pressure (CPP), preserve cerebral autoregulation and carbon dioxide reactivity, confer neuroprotection, and not interfere with neurophysiological monitoring. Numerous agents, both intravenous and inhalational, as well as different techniques have been used and studied.1,2
Over the past three decades, multiple studies have compared propofol-maintained and volatile-based anesthesia in patients undergoing elective craniotomy.1–8 Footnote 1 Most of these studies have looked at either perioperative cerebral hemodynamic effects or recovery profiles. Few studies have evaluated clinically significant outcomes such as neurological morbidity, mortality, or quality of life. In addition, an intrinsic problem of research in neuroanesthesia is the small sample size of many studies. Thus far, a lack of systematic reviews have been undertaken to compare propofol-maintained anesthesia vs volatile-maintained anesthesia for neurosurgical procedures. The objectives of this review were to evaluate the cerebral hemodynamic effects, operative conditions, recovery profiles, postoperative complications, and neurological outcomes of propofol-based vs volatile-based anesthesia for craniotomy.
Methods
This systematic review was carried out using the recommended guidelines provided by the Cochrane Handbook for Systematic Reviews of InterventionsFootnote 2 and reported according to the PRISMA 2009 checklist.Footnote 3
Search strategy
A professional librarian searched the databases, MEDLINE® (from 1946 to November 10, 2013), MEDLINE In-Process & other Non-Indexed Citations (Ovid) (November 10, 2013), EMBASE™ (from 1974 to November 10, 2013), Cochrane Central Register of Controlled Trials (CENTRAL) in the Cochrane Library (November 10, 2013), and Cochrane Database of Systematic Reviews (CDSR) in the Cochrane Library (November 10, 2013), and the procedure was reviewed by two authors (L.V., J.M.). Searches were conducted using three different components 1) terms related to inhalational anesthetics 2) terms related to intravenous anesthetics, and 3) terms related to craniotomy. The following MeSH terms were used in searching MEDLINE (Appendix 1; available as Electronic Supplementary Material) and The Cochrane Register of Controlled Trials: “Anesthetics, Inhalation”, “Enflurane”, “Ethyl Ether”, “Halothane”, “Isoflurane”, “Methoxyflurane”, “Nitrous Oxide”, “Trichloroethylene”, “Xenon”, “Sevoflurane”, “Desflurane”, “Intravenous anesthetics”, “Total intravenous anaesthesia”, “Alfentanil”, “Chloralose”, “Diazepam”, “Etomidate”, “Fentanyl”, “Methohexital”, “Midazolam”, “Propanidid”, “Propofol”, “Sodium Oxybate”, “Sufentanil”, “Thiamylal”, “Thiopental”, “Urethane”, “lorazepam”, “meperidine”, “remifentanil”, “ketamine”, “droperidol”, “Craniotomy”, “volatile induction/maintenance anaesthesia”, “intracranial surgery”, “intracranial operation”, “Supratentorial surgery”, “Neurosurgical Procedures”, “Neurosurgery”, “Brain surgery”, “Brain resection”, and “Neurosurgery”. Corresponding EMTREE terms were used in searching EMBASE (Appendix 2; available as Electronic Supplementary Material). Randomized controlled trials (RCT) was not used as a search term. A search of trial registries and a manual search of the reference lists from the selected articles were conducted to identify additional trials. The searches were restricted to the English language and human studies.
Selection criteria
Two independent reviewers (J.C., R.M.) evaluated the search results and identified the eligible studies for possible inclusion using predefined selection criteria. Adult patients with both supratentorial and infratentorial pathologies undergoing elective neurosurgical procedures were included in the review. There was no restriction on the type of neurosurgical pathology, but patients with traumatic brain injury were excluded. Only randomized and quasi-randomized controlled trials that compared propofol-maintained with volatile-maintained anesthesia were included. Propofol-maintained anesthesia was defined as using propofol infusion, either by manual controlled or targeted controlled infusion (TCI), for maintenance of anesthesia. Volatile-maintained anesthesia included sevoflurane, desflurane, or isoflurane, with or without nitrous oxide. The primary outcome of this systemic review was the intraoperative brain relaxation score. Secondary outcomes included other intraoperative cerebral hemodynamics (intracranial pressure [ICP] and cerebral perfusion pressure), intraoperative hemodynamic events, recovery profiles, postoperative complications, and neurological outcomes. Studies reporting any of the abovementioned outcome variables were included for evaluation. Any disagreements in selection between the two authors were resolved by discussion or by consulting with the senior author (L.V.).
Data extraction and quality assessment
The data were extracted by J.C. and R.M. individually and validated by the senior author (L.V.) and by double data entry. Details of the study population, interventions, and outcomes were extracted using a standardized electronic data extraction form. When necessary, authors of the selected articles were contacted to obtain missing information for quantitative analysis. Two reviewers (J.C., R.M.) graded the eligible studies independently using the Cochrane risk of bias assessment scale. The Cochrane risk of bias assessment scale is a domain-based evaluation tool developed by Cochrane Collaboration. It assesses seven different domains of bias in the primary studies: random sequence generation, adequacy of concealment of allocation, blinding of participants and healthcare providers, blinding of outcome assessors, incomplete outcome data, risk of selective reporting bias, and other sources of bias.C
Outcome measurements
The primary outcome of this review was the brain relaxation score. The brain relaxation score is a reflection of the degree of brain swelling as evaluated by the surgeon after opening the dura and is usually reported on a four-point scale (slack brain, mild, moderate, and severe brain herniation). Secondary outcomes included other intraoperative cerebral hemodynamics (ICP and CPP), intraoperative hemodynamic events (hypotension, hypertension, tachycardia, bradycardia), recovery profiles (time to eye opening, extubation, orientation, obeying commands, and an Aldrete score > 9), postoperative complications (nausea, vomiting, seizures, shivering, agitation, pain), and clinical outcomes (postoperative neurological morbidity, mortality, and quality of life). There were many variations with regard to timing and the method of ICP measurement. Only studies using either epidural or subdural ICP measurement at the time of the first burr hole in the craniotomy were included in this review. Similarly, only studies where CPP was calculated with the formula using mean arterial pressure (MAP), (CPP = MAP-ICP), at the time of the first burr hole in the craniotomy were included in the analysis. Outcome variables reported in different scales or using different measuring methods or different timing were not used for analysis.
Statistical analysis
The meta-analysis was performed using Review Manager, version 5.1 software (The Cochrane Collaboration, Oxford, UK) with a random effects model. Considering the clinical heterogeneity and the potential variations in effect size amongst the studies, a random effects model was chosen instead of a fixed effects model. The results are presented as a risk ratio (RR) for dichotomous data and as mean difference (MD) for continuous data with corresponding 95% confidence intervals. The percentage of variability across studies attributable to heterogeneity rather than chance was estimated using the I2 statistic. The strategies used to deal with heterogeneity in this systemic review include i) conducting a subgroup analysis to explore the heterogeneity in the use of anesthetic agents (propofol infusion techniques, different types/combinations of volatile agents, and types of opioids used) and ii) performing the sensitivity analysis according to the methodological quality of the primary studies. Publication bias was assessed using Begg’s funnel plots.
Results
Characteristics of included trials
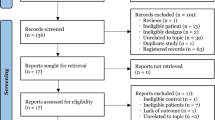
The search strategy resulted in an initial yield of 1,599 citations. Thirty-six potential articles were selected after thorough examination of titles and abstracts, and further examination led to the exclusion of 19 studies from analysis, as shown in Fig. 1. Finally, 14 studies (n = 1,819) were included in the analysis.9–22 The characteristics of the study populations are summarized in Table 1. The sample size of these studies varies from 40-411 patients. All the included studies were RCTs that were conducted in either a single centre or multiple centres. Eight of the 14 studies involved supratentorial craniotomies, and the remaining six studies involved both supratentorial and infratentorial surgery. The indications for craniotomy included brain tumour resection, aneurysm clipping, excision of arteriovenous malformations, vessel ligation for trigeminal neuralgia, and evacuation of hematomas. Most of the study population consisted of young fit neurologically intact elective neurosurgical patients (Glasgow Coma Score of 15 and American Society of Anesthesiologists physical status I-III) with a mean age of 52.1 (range 18-70). The distribution of sex was quite homogeneous across different studies.
Anesthesia management is shown in Table 1. For propofol-maintained anesthesia, the manual infusion of propofol at rates of 10-8-6 mg·kg−1·hr−1 was the most commonly used technique in the primary studies. Only two studies used TCI for the administration of propofol. With regard to volatile-maintained anesthesia, sevoflurane alone was used in seven studies, one study used either sevoflurane or isoflurane alone, and six studies used isoflurane with nitrous oxide. Desflurane was not used in any study. Mannitol was used in only eight studies. The majority of studies reported the titration of anesthetic agents against blood pressure not according to depth of anesthesia monitors. In addition, in most studies, only the proposed dosing regimen was reported and not the actual total doses of anesthetics used.
Methodological quality of the studies
A risk of bias graph and summaries are shown in Figs 2 and 3. In eight studies, the randomization sequences were generated by computer-generated random numbers, and in six studies, the methods of randomization were not specified. An open-label design was commonly used in many studies, and only six studies provided good documentation of adequate allocation concealment. Blinding of participants was documented in two studies, and in four studies, blinding of outcome assessors was reported. Selective reporting was identified in four studies. No study was rated “yes” in all domains.
Meta-analysis results
The results of the meta-analysis of all outcome variables are summarized in Table 2. All forest plots and funnel plots are available online (Figs 4 to 35) as Electronic Supplementary Material.
Cerebral hemodynamics
Brain relaxation score
The majority of studies used a four-point scale (slack brain, mild, moderate, and severe brain herniation). Some studies used a three-point scale, whereas others reported only the number of events of brain herniation that required treatment with mannitol. For this meta-analysis, only the studies with complete data for a four-point scale were included, and the RR for each category was calculated.9,11,16,19,22 The propofol-maintained group had lower RR for moderate herniation (RR = 0.75; 95% CI 0.58 to 0.97; P = 0.03) than the volatile-maintained group, but there were no differences amongst the groups in other categories.
Intracranial pressure and cerebral perfusion pressure
In the pooled analysis, propofol-maintained anesthesia had lower ICP values than volatile-maintained anesthesia; the overall weighted mean difference was −5.2 mmHg (95% CI −6.81 to −3.6; P < 0.0001).16,19,22 Similar to the ICP comparison, there were significant variations in the CPP measurement in different studies. The propofol-maintained group had higher CPP values than the volatile-maintained group; the combined CPP weighted mean difference was 16.33 mmHg (95% CI 12.2 to 20.46; P < 0.001).16,19
Intraoperative hemodynamic events
The hemodynamic stability of the two anesthetic techniques was evaluated in most of the primary studies and was frequently reported as the number of events of hypertension, hypotension, bradycardia, and tachycardia. Propofol-maintained anesthesia was associated with fewer intraoperative hypotensive events than volatile-maintained anesthesia (RR= 0.74; 95% CI 0.61 to 0.90; P = 0.002). There were no significant differences in the incidence of intraoperative hypertension, bradycardia and tachycardia between the groups.9,11,13–15,17
Recovery profiles
An objective comparison of the two anesthetic techniques was difficult due to the variations in the variables used to quantify the recovery profiles of the patients. Commonly used recovery variables included mean times to: eye opening, tracheal extubation, obeying verbal commands, orientation, and to achieve an Aldrete score > 9. Due to the presence of the clinical and methodological heterogeneity in the primary studies, not all recovery profile outcomes could be combined and quantitative analysis was not possible. Semi-qualitative evaluation of the data, the mean difference in time to eye opening11,14,15,18 and extubation11,14,15,19 between the groups, ranged from −4 to 2 min and from 0.1 to 2 min, respectively. Similarly, the mean difference in time to achieve an Aldrete score > 9 ranged from −3.75 to 0.13 min.9,11,19 The times to obey commands14,19 and orientation19,20 were faster in the propofol-maintained group with the mean difference ranging from −5.5 to 0 min and from −7.5 to −23.6 min, respectively.
Postoperative complications
Postoperative complications were commonly reported as the incidence (i.e., number) of events that occurred per total patients. There were no significant differences between propofol-maintained and volatile-maintained techniques with regard to the incidence of postoperative pain, seizure, and agitation. Nevertheless, the incidence of postoperative nausea and vomiting (PONV) was significantly lower with propofol-maintained anesthesia. There was a significant heterogeneity encountered in combining the results (Fig 13; available as Electronic Supplementary Material); hence, subgroup analysis was completed, which showed that nitrous oxide is an effect modifier of propofol-maintained anesthesia in preventing PONV. The protective effect of propofol-maintained anesthesia on PONV was more prominent in the subgroup where nitrous oxide was used (OR 0.1; 95% CI 0.03 to 0.35; P < 0.0003)17,19,21 compared with the group where no nitrous oxide was used (OR 0.65; 95% CI 0.38 to 1.10; P = 0.14).9,11,13–15
Neurological outcomes
One primary study reported the 24-hr postoperative neurological complications as a secondary outcome and did not find differences between the two anesthetic techniques.20 None of the included studies investigated other clinical outcomes in the two anesthetic techniques, such as long-term neurological morbidity, mortality, or quality of life; consequently, quantitative analysis could not be performed.
Subgroup analysis
Subgroup analysis was performed for each outcome parameter with different combinations of anesthetic regimens (propofol infusion techniques, different types/combination of volatile agents, and types of opioids used). With propofol-maintained anesthesia, there are no differences in the pooled results with either manual controlled infusions or targeted-controlled infusions. Similarly, in the volatile-maintained group, there are no differences in the pooled results with use of either isoflurane or sevoflurane as the choice of volatile agent. There are no differences in the subgroup analysis of other outcome parameters with the addition of nitrous oxide.
Discussion
This meta-analysis showed that brain relaxation scores were similar when comparing propofol-maintained vs volatile-maintained anesthesia; however, with propofol-maintained anesthesia, the initial ICP was lower and CPP was higher when compared with volatile-maintained anesthesia. This ICP lowering effect is not associated with less brain swelling or better operative conditions after dural opening, as shown by the similar brain relaxation score with both techniques. Nevertheless, all studies that assessed the brain relaxation scores were limited to patients with a Glasgow Coma Score >15 undergoing supratentorial excision of tumours. The preoperative ICP would be expected to be normal or only slightly elevated in this group of patients. Therefore, the effect of propofol-maintained anesthesia on brain relaxation might be expected to be small. The intraoperative hemodynamic events and perioperative complications were similar with the two techniques, except for the incidence of PONV. Due to heterogeneity, we were unable to combine the variables to assess the recovery profiles; however, mean differences in certain important recovery variables were small, e.g., the magnitude of minutes, and they were also of minimal clinical significance.
Currently, there are no consensus guidelines or recommendations suggesting the best anesthesia technique for neurosurgical procedures. Most clinical trials assessed perioperative cerebral and hemodynamic variables and recovery profiles as their major outcome measures. The major clinically relevant outcomes, such as neurological function, morbidity, mortality, and the quality of life, were not evaluated in most studies comparing the two anesthetic techniques. For purposes of applying the results of this systemic review into practice, there were no differences in the majority of outcome variables, and due to lack of data on clinically significant outcomes, we are unable to recommend one anesthetic technique over the others in patients undergoing elective craniotomy.
This systematic review has several limitations. First, all primary studies included only relatively healthy and neurologically intact patients; hence, the extrapolation of our results to all neurosurgical patients is inappropriate. Second, in this systematic review, we have segregated the interventions into either propofol-based or volatile-based anesthesia. This classification is generally understood and was commonly used in the previous narrative reviews for discussion. Nevertheless, there could be concerns about oversimplification and generalization of different anesthetic regimens. The volatile-based anesthesia group, in particular, included different combinations of volatile agents (sevoflurane, isoflurane, with or without nitrous oxide); however, subgroup analysis did not reveal any differences in the pooled results with different combinations of volatile anesthetic regimens. Third, because of the significant clinical and methodological heterogeneity amongst the primary studies, we were unable to combine all the outcome variables for recovery profiles. Fourth, some other important considerations in neuroanesthesia include preservation of autoregulation and carbon dioxide reactivity, the neuroprotective effect, and the interference with neuromonitoring, but these factors were not included in this meta-analysis due to the heterogeneity of the data. Finally, cerebral ischemia, postoperative neurological deficits, and perioperative mortality are the important outcome parameters, but none of the primary studies measured these important outcomes. Instead, CPP and ICP were used as surrogate outcomes for cerebral ischemia.
Based on this systemic review, there are several study design features that would be beneficial for future studies. First, blinding of patients and the assessors (surgeon) is practically feasible in the operating theatre, and it is important to reduce reporting and observer bias. Second, the importance of reporting integrity should be emphasized, as the quality of reporting across studies is equally important to conducting the study. The CONSORT statement, which is endorsed by the Journal, is an evidence-based minimum set of recommendations for reporting RCTs.Footnote 4 Third, clinical outcomes such as neurological morbidity and mortality should be important in choosing anesthetic techniques and should be the focus of future studies.
In conclusion, this systematic review compared the effects of propofol-maintained vs volatile-maintained anesthesia in patients undergoing elective craniotomy. There were similarities between propofol-maintained and volatile-maintained anesthesia in the majority of outcomes measured. The initial mean ICP values were lower and CPP values were higher with propofol-maintained anesthesia than with volatile anesthesia, but this ICP lowering effect was not associated with less brain swelling or better operative conditions after dural opening, as shown by similar brain relaxation scores with both techniques. As no clinically significant outcome differences in neurological morbidity or mortality have been shown, conclusive recommendations based on the results of this systemic review cannot be made.
Notes
Ayrian E, Zelman V. Total intravenous anesthesia: Advantages for intracranial surgery - Commentary. Neurosurgery 2007; 61(5 Suppl. 2): ONS377.
Higgins JP, Green S. Cochrane Handbook for Systematic Reviews of Interventions. Version 5.1.0., March 2011. Available from URL: http://handbook.cochrane.org (accessed (August 2013)).
Preferred Reporting Items for Systematic Reviews and Meta-Analyses; Available from URL: http://www.prisma-statement.org (accessed (August 2013)).
Consolidated Standards of Reporting Trials: http://www.consort-statement.org (accessed August 2013).
References
Engelhard K, Werner C. Inhalational or intravenous anesthetics for craniotomies? Pro inhalational. Curr Opin Anaesthesiol 2006; 19: 504-8.
Hans P, Bonhomme V. Why we still use intravenous drugs as the basic regimen for neurosurgical anaesthesia. Curr Opin Anaesthesiol 2006; 19: 498-503.
Dahyot-Fizelier C, Frasca D, Debaene B. Inhaled agents in neuroanaesthesia for intracranial surgery: pro or con (French). Ann Fr Anesth Reanim 2012; 31: e229-34.
Mizumoto N, De Oliveira EP. Total intravenous anesthesia: advantages for intracranial surgery - Commentary. Neurosurgery 2007; 61(5 Suppl. 2): 377-8.
Weglinski MR, Perkins WJ. Inhalational versus total intravenous anesthesia for neurosurgery: theory guides, outcome decides. J Neurosurg Anesthesiol 1994; 6: 290-3.
Archer DP. TIVA versus inhalational anesthesia for neurosurgery. J Neurosurg Anesthesiol 1994; 6: 284.
Ravussin P, de Tribolet N, Wilder-Smith OH. Total intravenous anesthesia is best for neurological surgery. J Neurosurg Anesthesiol 1994; 6: 285-9.
Cole CD, Gottfried ON, Gupta DK, Couldwell WT. Total intravenous anesthesia: advantages for intracranial surgery. Neurosurgery 2007; 61(5 Suppl 2): 369-77.
Citerio G, Pesenti A, Latini R, et al. A multicentre, randomised, open-label, controlled trial evaluating equivalence of inhalational and intravenous anaesthesia during elective craniotomy. Eur J Anaesthesiol 2012; 29: 371-9.
Banevicius G, Rugyte D, Macas A, Tamasauskas A, Stankevicius E. The effects of sevoflurane and propofol on cerebral hemodynamics during intracranial tumors surgery under monitoring the depth of anesthesia. Medicina (Kaunas) 2010; 46: 743-52.
Lauta E, Abbinante C, Del Gaudio F, et al. Emergence times are similar with sevoflurane and total intravenous anesthesia: results of a multicenter RCT of patients scheduled for elective supratentorial craniotomy. J Neurosurg Anesthesiol 2010; 22: 110-8.
Bonhomme V, Demoitie J, Schaub I, Hans P. Acid-base status and hemodynamic stability during propofol and sevoflurane-based anesthesia in patients undergoing uncomplicated intracranial surgery. J Neurosurg Anesthesiol 2009; 21: 112-9.
Magni G, La Rosa I, Gimignani S, Melillo G, Imperiale C, Rosa G. Early postoperative complications after intracranial surgery: comparison between total intravenous and balanced anesthesia. J Neurosurg Anesthesiol 2007; 19: 229-34.
Sneyd JR, Andrews CJ, Tsubokawa T. Comparison of propofol/remifentanil and sevoflurane/remifentanil for maintenance of anaesthesia for elective intracranial surgery. Br J Anaesth 2005; 94: 778-83.
Magni G, Baisi F, La Rosa I, et al. No difference in emergence time and early cognitive function between sevoflurane–fentanyl and propofol–remifentanil in patients undergoing craniotomy for supratentorial intracranial surgery. J Neurosurg Anesthesiol 2005; 17: 134-8.
Petersen KD, Landsfeldt U, Cold GE, et al. intracranial pressure and cerebral hemodynamic in patients with cerebral tumors: a randomized prospective study of patients subjected to craniotomy in propofol-fentanyl, isoflurane-fentanyl, or sevoflurane-fentanyl anesthesia. Anesthesiology 2003; 98: 329-36.
Talke P, Caldwell J, Brown R, Dodson B, Howley J, Richardson CA. A comparison of three anesthetic techniques in Patients undergoing craniotomy for supratentorial intracranial surgery. Anesth Analg 2002; 95: 430-5.
Ittichaikulthol W, Pausawasdi S, Srichintai P, Sarnvivad P. Propofol vs isoflurane for neurosurgical anesthesia in Thai patients. J Med Assoc Thai 1997; 80: 454-60.
Todd MM, Warner DS, Sokoll MD, et al. A prospective, comparative trial of three anesthetics for elective supratentorial craniotomy. Propofol/fentanyl, isoflurane/nitrous oxide, and fentanyl/nitrous oxide. Anesthesiology 1993; 78: 1005-20.
Ravussin P, Tempelhoff R, Modica PA, Bayer-Berger MM. Propofol vs. thiopental-isoflurane for neurosurgical anesthesia: comparison of hemodynamics, CSF pressure, and recovery. J Neurosurg Anesthesiol 1991; 3: 85-95.
Mishra LD, Dube SK, Singh SN, Dubey RK. Comparison between continuous propofol infusion & conventional balanced anaesthesia in neurosurgical patients. J Anaesth Clin Pharmacol 2008; 24: 399-402.
Santra S, Das B. Subdural pressure and brain condition during propofol vs isoflurane - nitrous oxide anaesthesia in patients undergoing elective supratentorial tumour surgery. Indian J Anaesth 2009; 53: 44-51.
Acknowledgement
We acknowledge the assistance of Ms. Marina Englesakis for conducting the systematic searches for this manuscript.
Funding
None.
Conflicts of interest
None declared.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is accompanied by an editorial. Please see Can J Anesth 2014; 61: this issue.
Author contributions
Jason Chui, Ramamani Mariappan, Mehta Jigesh, Pirjo Manninen, and Lashmi Venkatraghavan helped conduct the study and write the manuscript.
Electronic supplementary material
Below is the link to the electronic supplementary material.
12630_2014_118_MOESM1_ESM.doc
Appendix 1 Search terms, strategy, and results in MEDLINE database. Appendix 2 Search terms, strategy, and results in EMBASE database. (DOC 188 kb)
Rights and permissions
About this article
Cite this article
Chui, J., Mariappan, R., Mehta, J. et al. Comparison of propofol and volatile agents for maintenance of anesthesia during elective craniotomy procedures: systematic review and meta-analysis. Can J Anesth/J Can Anesth 61, 347–356 (2014). https://doi.org/10.1007/s12630-014-0118-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12630-014-0118-9