Abstract
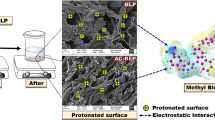
Commercial activated carbons were modified by a series of chemical or physical treatments using H2O2, NH3, and heating under N2 flow without notably changing their pore structures. The resultant carbons were characterized by N2 adsorption and Bohem titration and then used to remove Ponceau 4R, methyl orange and brilliant blue from aqueous solutions. Surface chemistry was found to play a significantly different role in removing these three compounds. The removal of anionic Ponceau 4R increases with increasing carbon surface basicity due to the predominant dispersive interaction mechanism. In contrast, surface chemistry has little effect on the removal of anionic methyl orange, which can be explained by two parallel mechanisms involving electrostatic and dispersive interactions due to the basic amine group in a dye molecule. The influence of surface chemistry on the removal of amphoteric brilliant blue dye can also be ignored due to a weak interaction between the carbons and dye molecules, which is resulted from strong cohesive energy from electrostatic forces inside amphoteric dye molecules.
Similar content being viewed by others
References
H. Kusic, N. Koprivanac, and A.L. Bozic, Environmental aspects on the photodegradation of reactive triazine dyes in aqueous media, J. Photochem. Photobiol. A, 252(2013), p. 131.
N. Junnarkar, D.S. Murty, N.S. Bhatt, and D. Madamwar, Decolorization of diazo dye Direct Red 81 by a novel bacterial consortium, World J. Microbiol. Biotechnol., 22(2006), No. 2, p. 163.
M. Asadullah, M. Asaduzzaman, M.S. Kabir, M.G. Mostofa, and T. Miyazawa, Chemical and structural evaluation of activated carbon prepared from jute sticks for Brilliant Green dye removal from aqueous solution, J. Hazard. Mater., 174(2010), No. 1-3, p. 437.
G. Mezohegyi, F.P. van der Zee, J. Font, A. Fortuny, and A. Fabregat, Towards advanced aqueous dye removal processes: a short review on the versatile role of activated carbon, J. Environ. Manage., 102(2012), p. 148.
A. Erto, R. Andreozzi, A. Lancia, and D. Musmarra, Factors affecting the adsorption of trichloroethylene onto activated carbons, Appl. Surf. Sci., 256(2010), No. 17, p. 5237.
A. Bhatnagar, W. Hogland, M. Marques, and M. Sillanpää, An overview of the modification methods of activated carbon for its water treatment applications, Chem. Eng. J., 219(2013), p. 499.
M.F.R. Pereira, S.F. Soares, J.J.M. Órfão, and J.L. Figueiredo, Adsorption of dyes on activated carbons: influence of surface chemical groups, Carbon, 41(2003), No. 4, p. 811.
S.B. Wang and Z.H. Zhu, Effects of acidic treatment of activated carbons on dye adsorption, Dyes Pigm., 75(2007), No. 2, p. 306.
C.Y. Yin, M.K. Aroua, and W.M.A.W. Daud, Review of modifications of activated carbon for enhancing contaminant uptakes from aqueous solutions, Sep. Purif. Technol., 52(2007), No. 3, p. 403.
F. López, F. Medina, M. Prodanov, and C. Güell, Oxidation of activated carbon: application to vinegar decolorization, J. Colloid Interface Sci., 257(2003), No. 2, p. 173.
H.P. Boehm, Some aspects of the surface chemistry of carbon blacks and other carbons, Carbon, 32(1994), No. 5, p. 759.
H.P. Boehm, Surface oxides on carbon and their analysis: a critical assessment, Carbon, 40(2002), No. 2, p. 145.
C. Sangwichien, G.L. Aranovich, and M.D. Donohue, Density functional theory predictions of adsorption isotherms with hysteresis loops, Colloids Surf. A, 206(2002), No. 1-3, p. 313.
G.L. Aranovich and M.D. Donohue, Adsorption isotherms for microporous adsorbents, Carbon, 33(1995), No. 10, p. 1369.
C. Moreno-Castilla, M.V. López-Ramón, and F. Carrasco-Marín, Changes in surface chemistry of activated carbon by wet oxidation, Carbon, 38(2000), No. 14, p. 1995.
C.A. Leon y Leon, J.M. Solar, V. Calemma, and L.R. Radovic, Evidence for the protonation of basal plane sites on carbon, Carbon, 30(1992), No. 5, p. 797.
K. Ebie, F.S. Li, Y. Azuma, A. Yuasa, and T. Hagishita, Pore distribution effect of activated carbon in adsorbing organic micropollutants from natural water, Water Res., 35(2001), No. 1, p. 167.
A. Rodríguez, J. García, G. Ovejero, and M. Mestanza, Adsorption of anionic and cationic dyes on activated carbon from aqueous solutions: Equilibrium and kinetics, J. Hazard. Mater., 172(2009), No. 2-3, p. 1311.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhou, Hl., Zhen, Wj., Zhu, Q. et al. Role of the surface chemistry of activated carbons in dye removal from aqueous solution. Int J Miner Metall Mater 22, 770–776 (2015). https://doi.org/10.1007/s12613-015-1133-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12613-015-1133-8