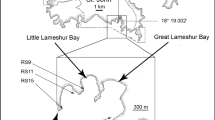
Abstract
Coastal lagoons are subjected to complex abiotic interactions which result to strong spatio-temporal variability of lagoon-inhabiting communities. Sponges are important components of coastal lagoons, yet their temporal variability is poorly understood. Here, we investigated the temporal variability in local distribution area (LDA), abundance (number of patches) and percentage cover of three lagoon-inhabiting sponge species: Neopetrosia chaliniformis, Amphimedon navalis and Spheciospongia vagabunda from Mauritius (Western Indian Ocean), over a 6- to 8-year period (depending on species). The correlations between two known drivers of sponge temporal patterns, sea surface temperature (SST) and chlorophyll a (Chl a) concentration, were also explored. After the survey, the total LDA and percentage cover of N. chaliniformis decreased by 40.2% and 14.6%, respectively, whereas total LDA and percentage cover of S. vagabunda increased by 135.1% and 23.3%, respectively. No significant changes were seen in the total LDA and percentage cover of A. navalis. While the abundance of N. chaliniformis and A. navalis declined over the study period, the abundance of S. vagabunda increased. SST and Chl a concentration were significantly correlated with temporal changes in abundance of all species, although correlations were species-specific. However, no significant correlations were seen between the environmental parameters and changes in sponge percentage cover, except between SST and N. chaliniformis percentage cover. Our results demonstrate that lagoon sponges show species-specific patterns in temporal variability and, while some species may potentially become locally extinct over the next few decades, if trends continue other species may become more abundant or may remain stable.





Similar content being viewed by others
References
Adjeroud M, Poisson E, Peignon C, Penin L, Kayal M (2019) Spatial patterns and short-term changes of coral assemblages along a cross-shelf gradient in the southwestern lagoon of New Caledonia. Diversity 11:21. https://doi.org/10.3390/d11020021
Aerts LAM (1998) Sponge/coral interactions in Caribbean reefs: analysis of overgrowth patterns in relation to species identity and cover. Mar Ecol Prog Ser 175:241–249. https://doi.org/10.3354/meps175241
Aerts LAM, Van Soest RWM (1997) Quantification of sponge/coral interactions in a physically stressed reef community, NE Colombia. Mar Ecol Prog Ser 148:125–134. https://doi.org/10.3354/meps148125
Anthony A, Atwood J, August P, Byron C, Cobb S, Foster C, Fry C, Gold A, Hagos K, Heffner L, Kellogg D, Lellis-Dibble K, Opaluch J, Oviatt C (2009) Coastal lagoons and climate change: ecological and social ramifications in U.S. Atlantic and Gulf Coast ecosystems. Ecol Soc 14. https://doi.org/10.5751/ES-02719-140108
Appadoo C, Beepat SS, Marie DEP (2011) Study of physico-chemical parameters affecting the distribution of sponge Xestospongia exigua (phylum Porifera, class Demospongiae) in a northern lagoon of Mauritius. J Environ Res Develop 5:741–748
Ashok AM, Schönberg CHL, Laju RL, Edward JKP (2019) Coral-killing sponge Terpios hoshinota in Southeast India—bested by Acropora muricata? Mar Biodivers 49:1069–1070. https://doi.org/10.1007/s12526-019-00966-8
Ashok AM, Calcinai B, Edward JKP (2020) The coral-killing red sponge Clathria (Microciona) aceratoobtusa (Porifera: Demosponigiae) invades various coral communities of Gulf of Mannar Marine National Park, southeast India. Eur Zool J 87:1–11. https://doi.org/10.1080/24750263.2019.1708486
Ávila E, Ávila-García AK, Cruz-Barraza JA (2015) Temporal and small-scale spatial variations in abundance and biomass of seagrass-dwelling sponges in a tropical estuarine system. Mar Ecol 36:623–636. https://doi.org/10.1111/maec.12171
Bates D, Mächler M, Bolker B, Walker S (2014) Package Lme4: linear mixed-effects models using Eigen and S4. J Stat Softw 67:1–99
Beedessee G, Ramanjooloo A, Aubert G, Eloy L, Surnam-Boodhun R, Van Soest RW, Cresteil T, Marie DE (2012) Cytotoxic activities of hexane, ethyl acetate and butanol extracts of marine sponges from Mauritian waters on human cancer cell lines. Environ Toxicol Pharmacol 34:397–408. https://doi.org/10.1016/j.etap.2012.05.013
Beepat SS (2015) Ecology, diversity and symbiotic associates (with special reference to Actinobacteria) of marine sponges in the coastal lagoons of Mauritius. University of Mauritius, Dissertation
Beepat SS, Appadoo C, Marie DE, Paula J, Sivakumar K (2013) Distribution, abundance and ecology of the sponge Spheciospongia vagabunda (phylum: Porifera, class: Demospongiae) in a shallow lagoon of Mauritius. West Indian Ocean J Mar Sci 12:15–23
Beepat SS, Appadoo C, Marie DEP, Paula J, Çinar M, Sivakumar K (2014) Macrofauna associated with the sponge Neopetrosia exigua (Kirkpatrick, 1900) in Mauritius. West Indian Ocean J Mar Sci 13:133–142
Beepat SS, Appadoo C, Marie D, Sadally S, Paula J, Sivakumar K, Rao R, Salah M (2016) First records of sponge-associated Actinomycetes from two coastal sponges from Mauritius. West Indian Ocean J Mar Sci 15:31–38
Bell JJ (2008) The functional roles of marine sponges. Estuar Coast Shelf Sci 79:341–353. https://doi.org/10.1016/j.ecss.2008.05.002
Bell JJ, Davy SK, Jones T, Taylor MW, Webster NS (2013) Could some coral reefs become sponge reefs as our climate changes? Glob Chang Biol 19:2613–2624. https://doi.org/10.1111/gcb.12212
Bell JJ, Bennett HM, Rovellini A, Webster NS (2018) Sponges to be winners under near-future climate scenarios. Bioscience 68:955–968. https://doi.org/10.1093/biosci/biy142
Biggerstaff A, Jompa J, Bell JJ (2017) Increasing benthic dominance of the phototrophic sponge Lamellodysidea herbacea on a sedimented reef within the coral triangle. Mar Biol 164:220. https://doi.org/10.1007/s00227-017-3253-3
Brito A, Newton A, Tett P, Fernandes T (2011) How will shallow coastal lagoons respond to climate change? A modelling investigation. Estuar Coast Shelf Sci 112:98–104. https://doi.org/10.1016/j.ecss.2011.09.002
Butler MJ, Hunt JH, Herrnkind WF, Childress M, Bertelsen R, Wc S, Matthews T, Field JM, Marshall H (1995) Cascading disturbances in Florida Bay, USA: cyanobacteria blooms, sponge mortality, and implications for juvenile spiny lobsters Panulirus argus. Mar Ecol Prog Ser 129:119–125. https://doi.org/10.3354/meps129119
Carballo JL, Vega C, Cruz-Barraza JA, Yáñez B, Nava H, Ávila E, Wilson M (2008) Short- and long-term patterns of sponge diversity on a rocky tropical coast: evidence of large-scale structuring factors. Mar Ecol 29:216–236. https://doi.org/10.1111/j.1439-0485.2008.00228.x
Casareto B, Bhagooli R, Fujimura H, Yoshimi S (2017) Chemical and biological characteristics of Albion reef in the south-west of Mauritius Island with special reference to primary production and N2 fixation of benthic substrata. West Indian Ocean J Mar Sci Special issue 1:85–93
Cebrian E, Uriz MJ, Garrabou J, Ballesteros E (2011) Sponge mass mortalities in a warming Mediterranean Sea: are cyanobacteria-harboring species worse off? PLoS One 6:e20211. https://doi.org/10.1371/journal.pone.0020211
Cerrano C, Bavestrello G, Bianchi CN, Cattaneo-vietti R, Bava S, Morganti C, Morri C, Picco P, Sara G, Schiaparelli S, Siccardi A, Sponga F (2000) A catastrophic mass-mortality episode of gorgonians and other organisms in the Ligurian Sea (north-western Mediterranean), summer 1999. Ecol Lett 3:284–293. https://doi.org/10.1046/j.1461-0248.2000.00152.x
Cerrano C, Bavestrello G, Boyer M, Calcinai B, Lalamentik LTX, Pansini M (2002) Psammobiontic sponges from the Bunaken Marine Park (North Sulawesi, Indonesia): interactions with sediments. Proc 9th Int Coral Reef Symp:279–283
Cerrano C, Pansini M, Valisano L, Calcinai B, Sarà M, Bavestrello G (2004) Lagoon sponges from Carrie bow cay (Belize): ecological benefits of selective sediment incorporation. Boll Mus Ist Biol Univ Genova 68:239–252
Cheshire AC, Wilkinson CR (1991) Modelling the photosynthetic production by sponges on Davies Reef, Great Barrier Reef. Mar Biol 109:13–18. https://doi.org/10.1007/BF01320226
Christia C, Giordani G, Papastergiadou E (2018) Environmental variability and macrophyte assemblages in coastal lagoon types of Western Greece (Mediterranean Sea). Water 10:151. https://doi.org/10.3390/w10020151
Corriero G, Longo C, Mercurio M, Marchini A, Occhipinti-Ambrogi A (2007) Porifera and Bryozoa on artificial hard bottoms in the Venice lagoon: spatial distribution and temporal changes in the northern basin. Ital J Zool 74:21–29. https://doi.org/10.1080/11250000601084100
Daby D (2003) Some quantitative aspects of seagrass ecology in a coastal lagoon of Mauritius. Mar Biol 142:193–203. https://doi.org/10.1007/s00227-002-0924-4
Daby D (2006) Current patterns and the distribution of benthic habitats in a coastal lagoon of Mauritius. Hydrobiologia 556:47–60. https://doi.org/10.1007/s10750-005-0593-7
de Goeij JM, van den Berg H, van Oostveen MM, Epping EHG, van Duyl FC (2008) Major bulk dissolved organic carbon (DOC) removal by encrusting coral reef cavity sponges. Mar Ecol Prog Ser 357:139–151. https://doi.org/10.3354/meps07403
de Moraes FC, Cervi F, Karez CS, Salgado LT, Moura RL, Leal GA, Bastos AC, Amado-Filho GM (2019) Long-term temporal and spatial patterns in bioeroding sponge distribution at the Abrolhos Bank, Brazil, Southwestern Atlantic. Mar Ecol 40:e12531. https://doi.org/10.1111/maec.12531
Di Camillo CG, Coppari M, Bartolucci I, Bo M, Betti F, Bertolino M, Calcinai B, Cerrano C, De Grandis G, Bavestrello G (2012) Temporal variations in growth and reproduction of Tedania anhelans and Chondrosia reniformis in the North Adriatic Sea. Hydrobiologia 687:299–313. https://doi.org/10.1007/s10750-011-0877-z
Duckworth AR, Peterson BJ (2013) Effects of seawater temperature and pH on the boring rates of the sponge Cliona celata in scallop shells. Mar Biol 160:27–35. https://doi.org/10.1007/s00227-012-2053-z
Duckworth AR, Wolff CW (2011) Population dynamics and growth of two coral reef sponges on rock and rubble substrates. J Exp Mar Biol Ecol 402:49–55. https://doi.org/10.1016/j.jembe.2011.03.017
Duckworth A, West L, Vansach T, Stubler A, Hardt M (2012) Effects of water temperature and pH on growth and metabolite biosynthesis of coral reef sponges. Mar Ecol Prog Ser 462:67–77. https://doi.org/10.3354/meps09853
Dunlap M, Pawlik JR (1996) Video-monitored predation by Caribbean reef fishes on an array of mangrove and reef sponges. Mar Biol 126:117–123. https://doi.org/10.1007/BF00571383
Elliott J, Patterson M, Summers N, Miternique C, Montocchio E, Vitry E (2016) How does the proliferation of the coral-killing sponge Terpios hoshinota affect benthic community structure on coral reefs? Coral Reefs 35:1083–1095. https://doi.org/10.1007/s00338-016-1434-z
Elliott JA, Patterson MR, Staub CG, Koonjul M, Elliott SM (2018) Decline in coral cover and flattening of the reefs around Mauritius (1998-2010). PeerJ 6:e6014. https://doi.org/10.7717/peerj.6014
Fagoonee I (1990) Coastal marine ecosystems of Mauritius. Int J Aquat Sci 208:55–62. https://doi.org/10.1007/BF00008443
Ferreira JG, Andersen JH, Borja A, Bricker SB, Camp J, Cardoso da Silva M, Garcés E, Heiskanen A-S, Humborg C, Ignatiades L, Lancelot C, Menesguen A, Tett P, Hoepffner N, Claussen U (2011) Overview of eutrophication indicators to assess environmental status within the European Marine Strategy Framework Directive. Estuar Coast Shelf Sci 93:117–131. https://doi.org/10.1016/j.ecss.2011.03.014
Holmes KE (2000) Effects of eutrophication on bioeroding sponge communities with the description of new West Indian sponges, Cliona spp. (Porifera: Hadromerida: Clionidae). Invertebr Biol 119:125–138. https://doi.org/10.1111/j.1744-7410.2000.tb00001.x
Illan M, Abelson A (1995) The life of a sponge in a sandy lagoon. Biol Bull 189:363–369. https://doi.org/10.2307/1542154
IPCC (2014) Climate change 2014: synthesis report. Contribution of working groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. IPCC, Geneva, Switzerland
Kelmo F, Bell JJ, Attrill MJ (2013) Tolerance of sponge assemblages to temperature anomalies: resilience and proliferation of sponges following the 1997–8 El-Niño southern oscillation. PLoS One 8:e76441. https://doi.org/10.1371/journal.pone.0076441
Kjerfve Br (1994) Coastal lagoon processes. New York : Elsevier, Amsterdam
Koopmans M, Wijffels RH (2008) Seasonal growth rate of the sponge Haliclona oculata (Demospongiae: Haplosclerida). Marine biotechnology (New York, NY) 10: 502-510. https://doi.org/10.1007/s10126-008-9086-9
Levi C, Laboute P, Bargibant G, Menou J (1998) Sponges from the new Caledonian lagoon, Paris
Longo C, Cardone F, Mercurio M, Nonnis Marzano C, Pierri C, Corriero G (2015) Spatial and temporal distributions of the sponge fauna in southern Italian lagoon systems. Mediterr Mar Sci 17:174–189. https://doi.org/10.12681/mms.1426
López-Victoria M, Zea S (2005) Current trends of space occupation by encrusting excavating sponges on Colombian coral reefs. Mar Ecol 26:33–41. https://doi.org/10.1111/j.1439-0485.2005.00036.x
Maldonado M, Zhang X, Cao X, Xue L, Cao H, Zhang W (2010) Selective feeding by sponges on pathogenic microbes: a reassessment of potential for abatement of microbial pollution. Mar Ecol Prog Ser 403:75–89. https://doi.org/10.3354/meps08411
McCain C, Szewczyk T, Bracy Knight K (2016) Population variability complicates the accurate detection of climate change responses. Glob Chang Biol 22:2081–2093. https://doi.org/10.1111/gcb.13211
McMurray S, Blum JE, Pawlik JR (2008) Redwood of the reef: growth and age of the giant barrel sponge Xestospongia muta in the Florida Keys. Mar Biol 155:159–171. https://doi.org/10.1007/s00227-008-1014-z
McMurray SE, Stubler AD, Erwin PM, Finelli CM, Pawlik JR (2018) A test of the sponge-loop hypothesis for emergent Caribbean reef sponges. Mar Ecol Prog Ser 588:1–14. https://doi.org/10.3354/meps12466
Moothien Pillay R, Terashima H, Venkatasami A, Uchida H (2002) Field guide to corals of Mauritius. Albion Fisheries Research Centre (AFRC), Ministry of Fisheries, Mauritius & Japan International Cooperation Agency (JICA)
Morganti T, Coma R, Yahel G, Ribes M (2017) Trophic niche separation that facilitates co-existence of high and low microbial abundance sponges is revealed by in situ study of carbon and nitrogen fluxes. Limnol Oceanogr 62:1963–1983. https://doi.org/10.1002/lno.10546
Muko S, Suzuki G, Saito M, Nakamura T, Nadaoka K (2019) Transitions in coral communities over 17 years in the Sekisei lagoon and adjacent reef areas in Okinawa, Japan. Ecol Res 34:524–534. https://doi.org/10.1111/1440-1703.12013
Nava H, Carballo JL (2008) Chemical and mechanical bioerosion of boring sponges from Mexican Pacific coral reefs. J Exp Biol 211:2827–2831. https://doi.org/10.1242/jeb.019216
Nicolaidou A, Reizopoulou S, Koutsoubas D, Orfranidis S, Kevrekidis T (2005) Biological components of Greek lagoonal ecosystems: an overview. Mediterr Mar Sci 6:31–50. https://doi.org/10.12681/mms.184
Nixon SW (1995) Coastal marine eutrophication: a definition, social causes, and future concerns. Ophelia 41:199–219. https://doi.org/10.1080/00785236.1995.10422044
Obura D, Gudka M, Rabi F, Gian S, Bijoux J, Freed S, Maharavo J, Mwaura J, Porter S, Sola E, Wickel J, Yahya S, Ahamada S (2017) Coral reef status report for the Western Indian Ocean (2017). Global Coral Reef Monitoring Network (GCRMN)/International Coral Reef Initiative (ICRI)
Pawlik JR (1998) Coral reef sponges: do predatory fishes affect their distribution? Limnol Oceanogr 43:1396–1399. https://doi.org/10.4319/lo.1998.43.6.1396
Pérez-Ruzafa A, Marcos C, Pérez-Ruzafa IM, Barcala E, Hegazi MI, Quispe J (2007) Detecting changes resulting from human pressure in a naturally quick-changing and heterogeneous environment: spatial and temporal scales of variability in coastal lagoons. Estuar Coast Shelf Sci 75:175–188. https://doi.org/10.1016/j.ecss.2007.04.030
Pérez-Ruzafa A, Hegazi MI, Perez-Ruzafa IM, Marcos C (2008) Differences in spatial and seasonal patterns of macrophyte assemblages between a coastal lagoon and the open sea. Mar Environ Res 65:291–314. https://doi.org/10.1016/j.marenvres.2007.11.008
Peterson CH (1979) Predation, competitive exclusion, and diversity in the soft-sediment benthic communities of estuaries and lagoons. In: Livingston RJ (ed) Ecological processes in coastal and marine systems. Springer US, Boston, pp 233–264
Povinec PP, Burnett WC, Beck A, Bokuniewicz H, Charette M, Gonneea ME, Groening M, Ishitobi T, Kontar E, Liong Wee Kwong L, Marie DEP, Moore WS, Oberdorfer JA, Peterson R, Ramessur R, Rapaglia J, Stieglitz T, Top Z (2012) Isotopic, geophysical and biogeochemical investigation of submarine groundwater discharge: IAEA-UNESCO intercomparison exercise at Mauritius Island. J Environ Radioact 104:24–45. https://doi.org/10.1016/j.jenvrad.2011.09.009
QGIS Development Team (2018) QGIS geographic information system. Open Source Geospatial Foundation Project. http://qgis.osgeo.org
R Core Team (2019) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Ramsby BD, Hoogenboom MO, Whalan S, Webster NS, Thompson A (2017) A decadal analysis of bioeroding sponge cover on the inshore Great Barrier Reef. Sci Rep 7:2706. https://doi.org/10.1038/s41598-017-02196-z
Ramsby BD, Hoogenboom MO, Smith HA, Whalan S, Webster NS (2018) The bioeroding sponge Cliona orientalis will not tolerate future projected ocean warming. Sci Rep 8:8302. https://doi.org/10.1038/s41598-018-26535-w
Reiswig H (1971a) In situ pumping activities of tropical Demospongiae. Mar Biol 9:38–50. https://doi.org/10.1007/BF00348816
Reiswig HM (1971b) Particle feeding in natural populations of three marine demosponges. Biol Bull 141:568–591. https://doi.org/10.2307/1540270
Rix L, Goeij J, Oevelen D, Struck U, Al-Horani FA, Wild C, Naumann MS (2018) Reef sponges facilitate the transfer of coral-derived organic matter to their associated fauna via the sponge loop. Mar Ecol Prog Ser 589:85–96. https://doi.org/10.3354/meps12443
Rose CS, Risk MJ (1985) Increase in Cliona delitrix infestation of Montastrea cavernosa heads on an organically polluted portion of the Grand Cayman Fringing Reef. Mar Ecol 6:345–363. https://doi.org/10.1111/j.1439-0485.1985.tb00142.x
Rossi G, Montori S, Cerrano C, Calcinai B (2015) The coral killing sponge Chalinula nematifera (Porifera: Haplosclerida) along the eastern coast of Sulawesi Island (Indonesia). Ital J Zool 82:143–148. https://doi.org/10.1080/11250003.2014.994046
Rovellini A, Dunn MR, Fulton EA, Webster NS, Smith DJ, Jompa J, Haris A, Berman J, Bell JJ (2019) Decadal variability in sponge abundance and biodiversity on an Indo-Pacific coral reef. Mar Ecol Prog Ser 620:63–76. https://doi.org/10.3354/meps12968
Rützler K (1975) The role of burrowing sponges in bioerosion. Oecologia 19:203–216. https://doi.org/10.1007/BF00345306
Rützler K (1988) Mangrove sponge disease induced by cyanobacterial symbionts: failure of a primitive immune system? Dis Aquat Org 5:143–149. https://doi.org/10.3354/dao005143
Schönberg CHL, Fang JK-H, Carballo JL (2017) Bioeroding sponges and the future of coral reefs. In: Carballo JL, Bell JJ (eds) Climate change, ocean acidification and sponges: impacts across multiple levels of organization. Springer International Publishing, Cham, pp 179–372
Souchu P, Bec B, Smith VH, Laugier T, Fiandrino A, Benau L, Orsoni V, Collos Y, Vaquer A (2010) Patterns in nutrient limitation and chlorophyll a along an anthropogenic eutrophication gradient in French Mediterranean coastal lagoons. Can J Fish Aquat Sci 67:743–753. https://doi.org/10.1139/F10-018
Stevely J, Sweat D, Bert T, Sim-Smith C, Kelly M (2010) Sponge mortality at marathon and long key, Florida: patterns of species response and population recovery. Proc Gulf Caribb Fish Inst 63:384–400
Stokes MD, Deane GB (2009) Automated processing of coral reef benthic images. Limnol Oceanogr Methods 7:157–168. https://doi.org/10.4319/lom.2009.7.157
Taylor D, Nixon S, Granger S, Buckley B (1995) Nutrient limitation and the eutrophication of coastal lagoons. Mar Ecol Prog Ser 127:235–244. https://doi.org/10.3354/meps127235
Thacker RW (2005) Impacts of shading on sponge-cyanobacteria symbioses: a comparison between host-specific and generalist associations. Integr Comp Biol 45:369–376. https://doi.org/10.1093/icb/45.2.369
Turicchia E, Hoeksema BW, Ponti M (2018) The coral-killing sponge Chalinula nematifera as a common substrate generalist in Komodo National Park, Indonesia. Mar Biol Res 14:827–833. https://doi.org/10.1080/17451000.2018.1544420
Valentine M, Butler MJ (2019) Sponges structure water column characteristics in shallow, tropical coastal ecosystems. Mar Ecol Prog Ser 608:133–147. https://doi.org/10.3354/meps12758
Venables WN, Ripley BD (2013) Modern applied statistics with S-plus. Springer, New York
Vernberg F (1982) Environmental adaptation to lagoon systems. Oceanol Acta 5
Wall C, Rodgers B, Gobler C, Peterson B (2012) Responses of loggerhead sponges Spheciospongia vesparium during harmful cyanobacterial blooms in a sub-tropical lagoon. Mar Ecol Prog Ser 451:31–43. https://doi.org/10.3354/meps09537
Webster NS (2007) Sponge disease: a global threat? Environ Microbiol 9:1363–1375. https://doi.org/10.1111/j.1462-2920.2007.01303.x
Wilkinson C (1978) Microbial associations in sponges. I. Ecology, physiology and microbial populations of coral reef sponges. Mar Biol 49:161–167. https://doi.org/10.1007/BF00387115
Wilkinson CR (1983) Net primary productivity in coral reef sponges. Science 219:410. https://doi.org/10.1126/science.219.4583.410
Wulff JL (1995) Effects of a hurricane on survival and orientation of large erect coral reef sponges. Coral Reefs 14:55–61. https://doi.org/10.1007/BF00304073
Wulff JL (2006) Rapid diversity and abundance decline in a Caribbean coral reef sponge community. Biol Conserv 127:167–176. https://doi.org/10.1016/j.biocon.2005.08.007
Zea S (1994) Patterns of sponge and coral abundance in stressed coral reefs at Santa Marta, Colombian Caribbean. In: VanSoest (ed) Sponges in time and space biology, chemistry, Paleontology. Balkema, Rotterdam, pp 257–264
Zeileis A, Kleiber C, Jackman S (2008) Regression models for count data in R. 27:1–25. https://doi.org/10.18637/jss.v027.i08
Acknowledgements
We thank D. E. P. Marie and C. Samyan from the Mauritius Oceanography Institute (MOI) for initial sponge surveys in Mauritius. We also thank K. S. Beesoon, Akhilesh N. S. Beepat and Bhavna D. Ramlochun, for their assistance with logistics and fieldwork in Mauritius. We are also grateful to the three anonymous reviewers and Prof. M. Klautau who provided constructive comments to improve the quality of this manuscript.
Funding
This study was funded by the New Zealand Commonwealth Doctoral Scholarship from the Ministry of Foreign Affairs and Trade, New Zealand, awarded to S. S. Beepat (Grant Number 4036).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicate international, national, and/or institutional guidelines for the care use of animals were followed by the authors.
Sampling and field studies
All necessary permits for sampling and observational field studies have been obtained by the authors from the competent authorities and are mentioned in the acknowledgments.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Author contribution
Conception of ideas and methodology designs for this study was carried out by SSB, JJB and SKD. SSB conducted all field surveys and wrote the manuscript. LW contributed to the statistical analysis of the data. Satellite data was acquired and interpreted by KJ. All authors have contributed in reading and editing and have approved the manuscript.
Additional information
Communicated by M. Klautau
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 41 kb)
Rights and permissions
About this article
Cite this article
Beepat, S.S., Woods, L., Davy, S.K. et al. Temporal variability in tropical lagoon sponges from Mauritius (Western Indian Ocean). Mar. Biodivers. 50, 57 (2020). https://doi.org/10.1007/s12526-020-01079-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12526-020-01079-3