Abstract
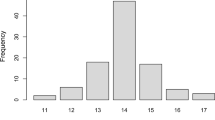
Spinocerebellar ataxia subtypes 1, 3, and 6 (SCA1, MJD/SCA3, and SCA6) are among the most prevalent autosomal dominant cerebellar ataxias worldwide, but their relative frequencies in Peru are low. Frequency of large normal (LN) alleles at spinocerebellar ataxia-causative genes has been proposed to be associated with disease prevalence. To investigate the allelic distribution of the CAG repeat in ATXN1, ATXN3, and CACNA1A genes in a Peruvian mestizo population and examine their association with the relative frequency of SCA1, MJD/SCA3, and SCA6 across populations. We genotyped 213 healthy mestizo individuals from Northern Lima, Peru, for ATXN1, ATXN3, and CACNA1A using polymerase chain reaction (PCR) and polyacrylamide gel electrophoresis (PAGE). We compared the frequency of LN alleles and relative disease frequency between populations. We also tested 40 samples for CAT repeat interruptions within the CAG tract of ATXN1. We found no association between disease frequency and population frequency of LN alleles at ATXN1 and ATXN3. All 40 ATXN1 samples tested for CAT interruptions were positive. Frequency of LN alleles at CACNA1A correlates with SCA6 frequency across several populations, but this effect was largely driven by data from a single population. Low frequency of SCA1 and MJD/SCA3 in Peru is not explained by frequency of LN alleles at ATXN1 and ATXN3, respectively. The observed correlation between CACNA1A LN alleles and SCA6 frequency requires further assessment.


Similar content being viewed by others
References
López-Flores I, Garrido-Ramos MA. The repetitive DNA content of eukaryotic genomes. Genome Dyn. 2010;7:1–28.
Durr A. Autosomal dominant cerebellar ataxias: polyglutamine expansions and beyond. Lancet Neurol. 2010;9:885–94.
Schöls L, Bauer P, Schmidt T, Schulte T, Riess O. Autosomal dominant cerebellar ataxias: clinical features, genetics, and pathogenesis. Lancet Neurol. 2004;3:291–304.
Jin DK, Oh MR, Song SM, Koh SW, Lee M, Kim GM, et al. Frequency of spinocerebellar ataxia types 1,2,3,6,7 and dentatorubral pallidoluysian atrophy mutations in Korean patients with spinocerebellar ataxia. J Neurol. 1999;246:207–10.
Magaña JJ, Tapia-Guerrero YS, Velázquez-Pérez L, Cerecedo-Zapata CM, Maldonado-Rodríguez M, Jano-Ito JS, et al. Analysis of CAG repeats in five SCA loci in Mexican population: epidemiological evidence of a SCA7 founder effect. Clin Genet. 2014;85:159–65.
Puzyrev VP, Maximova NP. Hereditary diseases among Yakuts. Russ J Genet. 2008;44:1141–7.
Maruyama H, Izumi Y, Morino H, Oda M, Toji H, Nakamura S, et al. Difference in disease-free survival curve and regional distribution according to subtype of spinocerebellar ataxia: a study of 1,286 Japanese patients. Am J Med Genet. 2002;114:578–83.
Soong B, Lu Y, Choo K, Lee H. Frequency analysis of autosomal dominant cerebellar ataxias in Taiwanese patients and clinical and molecular characterization of spinocerebellar ataxia type 6. Arch Neurol. 2001;58:1105–9.
Silveira I, Miranda C, Guimaraes L, Moreira M-C, Alonso I, Mendonca P, et al. Trinucleotide repeats in 202 families with ataxia: a small expanded (CAG) n allele at the SCA17 locus. Arch Neurol. 2002;59:623–9.
Bauer PO, Zumrova A, Matoska V, Marikova T, Krilova S, Boday A, et al. Absence of spinocerebellar ataxia type 3/Machado–Joseph disease within ataxic patients in the Czech population. Eur J Neurol. 2005;12:851–7.
Juvonen V, Hietala M, Kairisto V, Savontaus M-L. The occurrence of dominant spinocerebellar ataxias among 251 Finnish ataxia patients and the role of predisposing large normal alleles in a genetically isolated population. Acta Neurol Scand. 2005;111:154–62.
Alluri RV, Komandur S, Wagheray A, Chaudhuri JR, Null S, Meena AK, et al. Molecular analysis of CAG repeats at five different spinocerebellar ataxia loci: correlation and alternative explanations for disease pathogenesis. Mol Cells. 2007;24:338–42.
Gomez CM. Spinocerebellar Ataxia Type 6. In: Adam MP, Ardinger HH, Pagon RA, Wallace SE, Bean LJ, Stephens K, et al., editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993 [cited 2018 Oct 26]. Available from: http://www.ncbi.nlm.nih.gov/books/NBK1140/.
Storey E, du Sart D, Shaw JH, Lorentzos P, Kelly L, McKinley Gardner RJ, et al. Frequency of spinocerebellar ataxia types 1, 2, 3, 6, and 7 in Australian patients with spinocerebellar ataxia. Am J Med Genet. 2000;95:351–8.
Schöls L, Amoiridis G, Büttner T, Przuntek H, Epplen JT, Riess O. Autosomal dominant cerebellar ataxia: phenotypic differences in genetically defined subtypes? Ann Neurol. 1997;42:924–32.
Saleem Q, Choudhry S, Mukerji M, Bashyam L, Padma MV, Chakravarthy A, et al. Molecular analysis of autosomal dominant hereditary ataxias in the Indian population: high frequency of SCA2 and evidence for a common founder mutation. Hum Genet. 2000;106:179–87.
Chakraborty R, Kimmel M, Stivers DN, Davison LJ, Deka R. Relative mutation rates at di-, tri-, and tetranucleotide microsatellite loci. Proc Natl Acad Sci. 1997;94:1041–6.
Gacy AM, Goellner G, Juranić N, Macura S, McMurray CT. Trinucleotide repeats that expand in human disease form hairpin structures in vitro. Cell. 1995;81:533–40.
Guo J, Gu L, Leffak M, Li G-M. MutSβ promotes trinucleotide repeat expansion by recruiting DNA polymerase β to nascent (CAG)n or (CTG)n hairpins for error-prone DNA synthesis. Cell Res. 2016;26:775–86.
Ellegren H. Microsatellite mutations in the germline: implications for evolutionary inference. Trends Genet. 2000;16:551–8.
Fu Y-H, Kuhl DP, Pizzuti A, Pieretti M, Sutcliffe JS, Richards S, et al. Variation of the CGG repeat at the fragile X site results in genetic instability: resolution of the Sherman paradox. Cell. 1991;67:1047–58.
Falush D, Iwasa Y. Size-dependent mutability and microsatellite constraints. Mol Biol Evol. 1999;16:960.
Kay C, Collins JA, Wright GEB, Baine F, Miedzybrodzka Z, Aminkeng F, et al. The molecular epidemiology of Huntington disease is related to intermediate allele frequency and haplotype in the general population. Am J Med Genet Part B Neuropsychiatr Genet. 2018;177:346–57.
Squitieri F, Andrew SE, Goldberg YP, Kremer B, Spence N, Zelsler J, et al. DNA haplotype analysis of Huntington disease reveals clues to the origins and mechanisms of CAG expansion and reasons for geographic variations of prevalence. Hum Mol Genet. 1994;3:2103–14.
Laffita-Mesa JM, Velázquez-Pérez LC, Santos Falcón N, Cruz-Mariño T, González Zaldívar Y, Vázquez Mojena Y, et al. Unexpanded and intermediate CAG polymorphisms at the SCA2 locus (ATXN2) in the Cuban population: evidence about the origin of expanded SCA2 alleles. Eur J Hum Genet. 2012;20:41–9.
Pearson CE, Eichler EE, Lorenzetti D, Kramer SF, Zoghbi HY, Nelson DL, et al. Interruptions in the triplet repeats of SCA1 and FRAXA reduce the propensity and complexity of slipped strand DNA (S-DNA) formation. Biochemistry. 1998;37:2701–8.
Chung MY, Ranum LP, Duvick LA, Servadio A, Zoghbi HY, Orr HT. Evidence for a mechanism predisposing to intergenerational CAG repeat instability in spinocerebellar ataxia type I. Nat Genet. 1993;5:254–8.
Zhou YX, Qiao WH, Gu WH, Xie H, Tang BS, Zhou LS, et al. Spinocerebellar ataxia type 1 in China: molecular analysis and genotype-phenotype correlation in 5 families. Arch Neurol. 2001;58:789–94.
Mittal U, Sharma S, Chopra R, Dheeraj K, Pal P, Srivastava A, et al. Insights into the mutational history and prevalence of SCA1 in the Indian population through anchored polymorphisms. Hum Genet. 2005;118:107–14.
Spinocerebellar Ataxia Type 1 - GeneReviews™ - NCBI Bookshelf [Internet]. [cited 2012 Sep 17]. Available from: http://www.ncbi.nlm.nih.gov/books/NBK1184/.
Gao R, Matsuura T, Coolbaugh M, Zühlke C, Nakamura K, Rasmussen A, et al. Instability of expanded CAG/CAA repeats in spinocerebellar ataxia type 17. Eur J Hum Genet. 2008;16:215–22.
Freund AA, Scola RH, Teive HAG, Arndt RC, da Costa-Ribeiro MCV, Alle LF, et al. Spinocerebellar ataxias: microsatellite and allele frequency in unaffected and affected individuals. Arq Neuropsiquiatr. 2009;67:1124–32.
Popova SN, Slominsky PA, Pocheshnova EA, Balanovskaya EV, Tarskaya LA, Bebyakova NA, et al. Polymorphism of trinucleotide repeats in loci DM, DRPLA and SCA1 in East European populations. Eur J Hum Genet. 2001;9:829.
Limprasert P, Nouri N, Heyman RA, Nopparatana C, Kamonsilp M, Deininger PL, et al. Analysis of CAG repeat of the Machado-Joseph gene in human, chimpanzee and monkey populations: a variant nucleotide is associated with the number of CAG repeats. Hum Mol Genet. 1996;5:207–13.
Limprasert P, Nouri N, Nopparatana C, Deininger PL, Keats BJ. Comparative studies of the CAG repeats in the spinocerebellar ataxia type 1 (SCA1) gene. Am J Med Genet. 1997;74:488–93.
Sobczak K, Krzyzosiak WJ. Patterns of CAG repeat interruptions in SCA1 and SCA2 genes in relation to repeat instability. Hum Mutat. 2004;24:236–47.
Details: IMBS [Internet]. [cited 2019 Mar 24]. Available from: https://www.imbs.uni-luebeck.de/forschung/software/details.html.
Harris DN, Song W, Shetty AC, Levano KS, Cáceres O, Padilla C, et al. Evolutionary genomic dynamics of Peruvians before, during, and after the Inca Empire. Proc Natl Acad Sci. 2018;115:E6526–35.
Miller SA, Dykes DD, Polesky HF. A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res. 1988;16:1215.
Orr HT, Chung MY, Banfi S, Kwiatkowski TJ Jr, Servadio A, Beaudet AL, et al. Expansion of an unstable trinucleotide CAG repeat in spinocerebellar ataxia type 1. Nat Genet. 1993;4:221–6.
Kawaguchi Y, Okamoto T, Taniwaki M, Aizawa M, Inoue M, Katayama S, et al. CAG expansions in a novel gene for Machado-Joseph disease at chromosome 14q32.1. Nat Genet. 1994;8:221–8.
Zhuchenko O, Bailey J, Bonnen P, Ashizawa T, Stockton DW, Amos C, et al. Autosomal dominant cerebellar ataxia (SCA6) associated with small polyglutamine expansions in the α1A-voltage-dependent calcium channel. Nat Genet. 1997;15:62–9.
Rousset F. genepop’007: a complete re-implementation of the genepop software for Windows and Linux. Mol Ecol Resour. 2008;8:103–6.
Takano H, Cancel G, Ikeuchi T, Lorenzetti D, Mawad R, Stevanin G, et al. Close associations between prevalences of dominantly inherited spinocerebellar ataxias with CAG-repeat expansions and frequencies of large normal CAG alleles in Japanese and Caucasian populations. Am J Hum Genet. 1998;63:1060–6.
Clopper CJ, Pearson ES. The use of confidence or fiducial limits illustrated in the case of the binomial. Biometrika. 1934;26:404–13.
Dorai-Raj S. binom: binomial confidence intervals for several parameterizations [Internet]. 2014 [cited 2019 May 8]. Available from: https://CRAN.R-project.org/package=binom.
R Development Core Team. R: a language and environment for statistical computing. Vienna: R Foundation for Statistical Computing; 2013. p. 2014.
Gan S-R, Shi S-S, Wu J-J, Wang N, Zhao G-X, Weng S-T, et al. High frequency of Machado-Joseph disease identified in southeastern Chinese kindreds with spinocerebellar ataxia. BMC Med Genet. 2010;11:47.
Cornejo-Olivas M, Inca-Martinez M, Castilhos R, Vasata Furtado G, Preusser Mattos E, Bavia Bampi G, et al. Hereditary ataxias in Peru: genetic analysis of familial and sporadic cases. The Cerebellum. 2019;In press.
Semaka A, Kay C, Doty C, Collins JA, Bijlsma EK, Richards F, et al. CAG size-specific risk estimates for intermediate allele repeat instability in Huntington disease. J Med Genet. 2013;50:696–703.
Andrés AM, Lao O, Soldevila M, Calafell F, Bertranpetit J. Dynamics of CAG repeat loci revealed by the analysis of their variability. Hum Mutat. 2003;21:61–70.
Lima M, Costa M d C, Montiel R, Ferro A, Santos C, Silva C, et al. Population genetics of wild-type CAG repeats in the Machado-Joseph disease gene in Portugal. Hum Hered. 2005;60:156–63.
Pujana MA, Corral J, Gratacòs M, Combarros O, Berciano J, Genís D, et al. Spinocerebellar ataxias in Spanish patients: genetic analysis of familial and sporadic cases. Hum Genet. 1999;104:516–22.
Jiang H, Tang B, Xia K, Zhou Y, Xu B, Zhao G, et al. Spinocerebellar ataxia type 6 in Mainland China: molecular and clinical features in four families. J Neurol Sci. 2005;236:25–9.
Templeton AR. Population genetics and microevolutionary theory: John Wiley & Sons; 2006.
Gaspar C, Lopes-Cendes I, Hayes S, Goto J, Arvidsson K, Dias A, et al. Ancestral origins of the Machado-Joseph disease mutation: a worldwide haplotype study. Am J Hum Genet. 2001;68:523–8.
Maciel P, Gaspar C, Guimarães L, Goto J, Lopes-Cendes I, Hayes S, et al. Study of three intragenic polymorphisms in the Machado-Joseph disease gene (MJD1) in relation to genetic instability of the (CAG)n tract. Eur J Hum Genet. 1999;7:147–56.
Acknowledgments
CGS and CCR contributed equally to this study, and are both to be considered first authors. Results of this study are derived from CGS and CCR Master’s thesis’ work.
We are very grateful to the participants of the community of Huaura, Santa María, Hualmay, and Huacho for contributing samples for this study. We kindly thank the Universidad de Huacho for the logistical support on the recruitment of participants. We thank Saul Lindo Samanamud for his cooperation in the recruitment of participants. We thank Kevin Boehnke, Miguel Inca Martínez, Hugo Sarapura Castro, and Erick Figueroa Ildefonso for their contribution on critical review and genotyping.
Funding
Research reported in this publication was supported by the Fogarty International Center (FIC) of the National Institutes of Health and the National Institute of Neurological Disorders and Stroke (NINDS) under grant #D43TW009345 awarded to the Northern Pacific Global Health Fellows Program and grant #D43TW009137 awarded to the Interdisciplinary Cerebrovascular Diseases Training Program in South America.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All procedures performed in this study involving human participants were in accordance with the standards of the Ethical Research Committee of the Instituto Nacional de Ciencias Neurológicas and with the 1964 Helsinki declaration and its later amendments.
Conflict of Interest
The authors declare that they have no conflict of interest.
Disclosure
The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gonzales-Sáenz, C., Cruz-Rodriguez, C., Espinoza-Huertas, K. et al. Distribution of the CAG Triplet Repeat in ATXN1, ATXN3, and CACNA1A Loci in Peruvian Population. Cerebellum 19, 527–535 (2020). https://doi.org/10.1007/s12311-020-01129-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12311-020-01129-3