Abstract
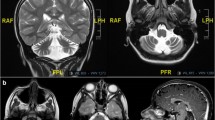
Spinocerebellar ataxia type15 (SCA15) is a pure ataxia characterized by very slow progression. Only seven families have been identified worldwide, in which partial deletions and a missense mutation of the inositol triphosphate receptor type I gene (ITPR1) have been reported. We examined a four-generation Italian family segregating an autosomal dominant cerebellar ataxia, in which linkage analysis was positive for the SCA15 locus. We performed a genomic real-time polymerase chain reaction to search for ITPR1 gene deletions in this family and in 60 SCA index cases negative for mutations in the SCA1–3, 6–8, 10, 12, and dentatorubral-pallidoluysian atrophy genes. The deleted segments were characterized using a custom array comparative genomic hybridization analysis. We have identified two families with an ITPR1 gene deletion: in one, the deletion involved ITPR1 only, while in the other both sulfatase-modifying factor 1 and ITPR1. Clinical data of ten patients and brain MRI (available for six) showed that the phenotype substantially overlapped known SCA15 cases, but we also noted buccolingual dyskinesias, facial myokymias, and pyramidal signs never reported in SCA15. ITPR1 expression analysis of two deleted cases showed a half dose. Our results further support ITPR1 gene as causative of SCA15. The families reported show that SCA15 is present in Italy and has a greater variability in the age at onset and clinical features than previously reported. We propose that the search for ITPR1 deletions is mandatory in the clinical hypothesis of SCA15 and that ITPR1-reduced expression in blood may be a useful marker to identify SCA15 patients harboring genomic deletions and possibly point mutations causing reduction of mRNA level.





Similar content being viewed by others
References
Duenas AM, Goold R, Giunti P (2006) Molecular pathogenesis of spinocerebellar ataxias. Brain 129:1357–1370
Matilla-Duenas A, Sanchez I, Corral-Juan M, Davalos A, Alvarez R, Latorre P (2009) Cellular and molecular pathways triggering neurodegeneration in the spinocerebellar ataxias. Cerebellum (in press)
Sato N, Amino T, Kobayashi K, Asakawa S, Ishiguro T, Tsunemi T et al (2009) Spinocerebellar ataxia type 31 is associated with “inserted” penta-nucleotide repeats containing (TGGAA)n. Am J Hum Genet 85:544–557
Harding AE (1993) Clinical features and classification of inherited ataxias. Raven, New York, pp 1–14
Gardner RJ, Knight MA, Hara K, Tsuji S, Forrest SM, Storey E (2005) Spinocerebellar ataxia type 15. Cerebellum 4:47–50
Knight MA, Kennerson ML, Anney RJ, Matsuura T, Nicholson GA, Salimi-Tari P et al (2003) Spinocerebellar ataxia type 15 (sca15) maps to 3p24.2-3pter: exclusion of the ITPR1 gene, the human orthologue of an ataxic mouse mutant. Neurobiol Dis 13:147–157
Storey E, Gardner RJ, Knight MA, Kennerson ML, Tuck RR, Forrest SM et al (2001) A new autosomal dominant pure cerebellar ataxia. Neurology 57:1913–1915
Hara K, Fukushima T, Suzuki T, Shimohata T, Oyake M, Ishiguro H et al (2004) Japanese SCA families with an unusual phenotype linked to a locus overlapping with SCA15 locus. Neurology 62:648–651
Iwaki A, Kawano Y, Miura S, Shibata H, Matsuse D, Li W et al (2008) Heterozygous deletion of ITPR1, but not SUMF1 in spinocerebellar ataxia type 16. J Med Genet 45:32–35
Ganesamoorthy D, Bruno DL, Schoumans J, Storey E, Delatycki MB, Zhu D et al (2009) Development of a multiplex ligation-dependent probe amplification assay for diagnosis and estimation of the frequency of spinocerebellar ataxia type 15. Clin Chem 55:1415–1418
van de Leemput J, Chandran J, Knight MA, Holtzclaw LA, Scholz S, Cookson MR et al (2007) Deletion at ITPR1 underlies ataxia in mice and spinocerebellar ataxia 15 in humans. PLoS Genet 3:e108
Hara K, Shiga A, Nozaki H, Mitsui J, Takahashi Y, Ishiguro H et al (2008) Total deletion and a missense mutation of ITPR1 in Japanese SCA15 families. Neurology 71:547–551
Brusco A, Gellera C, Cagnoli C, Saluto A, Castucci A, Michielotto C et al (2004) Molecular genetics of hereditary spinocerebellar ataxia: mutation analysis of spinocerebellar ataxia genes and CAG/CTG repeat expansion detection in 225 Italian families. Arch Neurol 61:727–733
Brussino A, Gellera C, Saluto A, Mariotti C, Arduino C, Castellotti B et al (2005) FMR1 gene premutation is a frequent genetic cause of late-onset sporadic cerebellar ataxia. Neurology 64:145–147
Cagnoli C, Mariotti C, Taroni F, Seri M, Brussino A, Michielotto C et al (2006) SCA28, a novel form of autosomal dominant cerebellar ataxia on chromosome 18p11.22-q11.2. Brain 129:235–242
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-delta delta C(T)) method. Methods 25:402–408
Street VA, Bosma MM, Demas VP, Regan MR, Lin DD, Robinson LC et al (1997) The type 1 inositol 1,4,5-trisphosphate receptor gene is altered in the opisthotonos mouse. J Neurosci 17:635–645
Dierks T, Schmidt B, Borissenko LV, Peng J, Preusser A, Mariappan M et al (2003) Multiple sulfatase deficiency is caused by mutations in the gene encoding the human C(alpha)-formylglycine generating enzyme. Cell 113:435–444
Hastings PJ, Ira G, Lupski JR (2009) A microhomology-mediated break-induced replication model for the origin of human copy number variation. PLoS Genet 5:e1000327
Acknowledgments
The authors are grateful to the family members for participating in this study. We thank Dr. Patrizia Pappi for technical help. Support for this work was provided by Regione Piemonte Ricerca Sanitaria Finalizzata (to AB), Telethon Italia grant no. GGP07110, and “Associazione Emma and Ernesto Rulfo per la Genetica Medica”.
Author information
Authors and Affiliations
Corresponding author
Additional information
Laura Orsi, Massimiliano Godani, and Giovanna Vaula equally contributed to the paper
Rights and permissions
About this article
Cite this article
Di Gregorio, E., Orsi, L., Godani, M. et al. Two Italian Families with ITPR1 Gene Deletion Presenting a Broader Phenotype of SCA15. Cerebellum 9, 115–123 (2010). https://doi.org/10.1007/s12311-009-0154-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12311-009-0154-0