Abstract
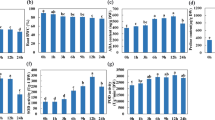
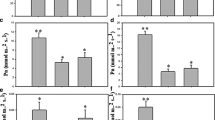
Climate change increases precipitation variability, particularly in savanna environments. We have used integrative strategies to understand the molecular mechanisms of drought tolerance, which will be crucial for developing improved genotypes. The current study compares the molecular and physiological parameters between the drought-tolerant Embrapa 48 and the sensitive BR16 genotypes. We integrated the root–shoot system’s transcriptome, proteome, and metabolome to understand drought tolerance. The results indicated that Embrapa 48 had a greater capacity for water absorption due to alterations in length and volume. Drought tolerance appears to be ABA-independent, and IAA levels in the leaves partially explain the higher root growth. Proteomic profiles revealed up-regulated proteins involved in glutamine biosynthesis and proteolysis, suggesting osmoprotection and explaining the larger root volume. Dysregulated proteins in the roots belong to the phenylpropanoid pathways. Additionally, PR-like proteins involved in the biosynthesis of phenolics may act to prevent oxidative stress and as a substrate for modifying cell walls. Thus, we concluded that alterations in the root–shoot conductive vessel system are critical in promoting drought tolerance. Moreover, photosynthetic parameters from reciprocal grafting experiments indicated that the root system is more essential than the shoots in the drought tolerance mechanism. Finally, we provided a comprehensive overview of the genetic, molecular, and physiological traits involved in drought tolerance mechanisms.










Similar content being viewed by others
Data availability
The data that support this study will be shared upon reasonable request to the corresponding author.
References
Agrawal GK, Thelen JJ (2009) A high-resolution two dimensional Gel-and Pro-Q DPS-based proteomics workflow for phosphoprotein identification and quantitative profiling. In: Phospho-Proteomics Humana Press, pp 3–19. https://doi.org/10.1007/978-1-60327-834-8_1
Akbudak MA, Yildiz S, Filiz E (2020) Pathogenesis related protein-1 (PR-1) genes in tomato (Solanum lycopersicum L.): bioinformatics analyses and expression profiles in response to drought stress. Genomics 112(6):4089–4099. https://doi.org/10.1016/j.ygeno.2020.07.004
Beis A, Patakas A (2015) Differential physiological and biochemical responses to drought in grapevines subjected to partial root drying and deficit irrigation. Eur J Agron 62:90–97. https://doi.org/10.1016/j.eja.2014.10.001
Bian S, Li R, Xia S, Liu Y, Jin D, Xie X, Dhaubhadel S, Zhai L, Wang J, Li X (2018) Soybean CCA1-like MYB transcription factor GmMYB133 modulates isoflavonoid biosynthesis. Biochem Biophys Res Commun 507(1–4):324–329. https://doi.org/10.1016/j.bbrc.2018.11.033
Carvalho JFC, Crusiol LGT, Perini LJ, Sibaldelli RNL, Ferreira LC, Guimarães FCM, Nepomuceno AL, Neumaier N, Farias JRB (2015) Phenotyping soybeans for drought responses using remot esensing techniques and nondestructive physiological analysis. Glob Sci Technol 8:1–16. https://doi.org/10.14688/1984-3801/gst.v8n2p1-16
Chen JY, Dai XF (2010) Cloning and characterization of the Gossypium hirutum major latex protein gene and functional analysis in Arabidopsis thaliana. Planta 231:861–873. https://doi.org/10.1007/s00425-009-1092-2
Coutinho FS, Santos DS, Lima LL, Vital CE, Santos LA, Pimenta MR, Silva JC, Ramos JRLS, Mehta A, Fontes EPB, Ramos HJO (2019) Mechanism of the drought tolerance of a transgenic soybean overexpressing the molecular chaperone BiP. Physiol Mol Biol Plants 25:457–472. https://doi.org/10.1007/s12298-019-00643-x
Coutinho FS, Lima LL, Mesquita RO, Carpinetti PA, Machado JP, Vital CE, Vidigal PM, Maximiano MR, Mehta A, Fontes EPB, Ramos HJO (2021) Remodeling of the cell wall as a drought tolerance mechanism of a soybean genotype revealed by global gene expression analysis. Mol Biol Rep 2(1):14–31. https://doi.org/10.1007/s42994-021-00043-4
Dai A (2013) Increasing drought under global warming in observations and models. Nat Clim Change 3:52–58. https://doi.org/10.1038/nclimate1633
Dodd IC (2005) Root-to-shoot signalling: assessing the roles of ‘up’ in the up and down world of long-distance signalling in planta. Plant Soil 274:251–270. https://doi.org/10.1007/s11104-004-0966-0
Du Y, Zhao Q, Chen L, Yao X, Zhang W, Zhang B (2020) Effect of drought stress on sugar metabolism in leaves and roots of soybean seedlings. Plant Physiol Biochem 146:1–12. https://doi.org/10.1016/j.plaphy.2019.11.003
Gomez JD, Pinheiro VJM, Silva JC, Romero JV, Meriño-Cabrera Y, Coutinho FS, Lourenção AL, Serrão JE, Vital CE, Fontes EPB, Oliveira MGA, Ramos HJO (2020) Leaf metabolic profiles of two soybean genotypes differentially affect the survival and the digestibility of Anticarsia gemmatalis caterpillars. Plant Physiol Biochem 155:196–212. https://doi.org/10.1016/j.plaphy.2020.07.010
Gouveia A, Lima LL, Coutinho FS, Rodrigues J, Pinheiro V, Ramos MES, Vital CE, Pontes CS, Pinheiro DP, Vidigal P, Barros E, Ramos HJO (2019) Metabolic pathway analysis by liquid chromatography (UHPLC) coupled to high resolution mass spectrometry (LC/MS). https://doi.org/10.17504/protocols.io.8svhwe6
Gulen H, Eris A (2003) Some physiological changes in strawberry cv. ‘Camarosa’ plants under heat stress. J Hort Sci Biotech 78:894–989. https://doi.org/10.1080/14620316.2003.11511715
Ha J, Kang YG, Lee T, Kim M, Yoon MY, Lee E et al (2019) Comprehensive RNA sequencing and co-expression network analysis to complete the biosynthetic pathway of coumestrol, a phytoestrogen. Sci Rep 9:1–11. https://doi.org/10.1038/s41598-018-38219-6
Iqbal N, Hussain S, Raza MA, Yang CQ, Safdar ME, Brestic M, Aziz A, Hayyat MS, Asghar MA, Wang XC, Zhang J, Yang W, Liu J (2019) Drought tolerance of soybean (Glycine max L. Merr.) by improved photosynthetic characteristics and an efficient antioxidant enzyme activities under a split-root system. Front Physiol 10:786. https://doi.org/10.3389/fphys.2019.00786
Kell DB (2011) Breeding crop plants with deep roots: their role in sustainable carbon, nutrient and water sequestration. Ann Bot 108:407–418. https://doi.org/10.1093/aob/mcr175
Kumar S, Bhushan B, Wakchaure GC, Meena KK, Kumar M, Meena NL, Rane J (2020) Plant phenolics under water-deficit conditions: biosynthesis, accumulation, and physiological roles in water stress alleviation. Plant Phenol Sustain Agric. https://doi.org/10.1007/978-981-15-4890-1_19
Li Z, Jing W, Peng Y, Zhang XQ, Ma X, Huang LK, Yan YH (2015) Spermine alleviates drought stress in white clover with different resistance by influencing carbohydrate metabolism and dehydrins synthesis. PLoS ONE 10(4):e0120708. https://doi.org/10.1371/journal.pone.0120708
Li X, Feng H, Wen J, Dong J, Wang T (2018) MtCAS31 aids symbiotic nitrogen fixation by protecting the leghemoglobin MtLb120-1 under drought stress in Medicago truncatula. Front Plant Sci 9:633. https://doi.org/10.3389/fpls.2018.00633
Lima LL, Balbi BP, Mesquita RO, Silva JCF, Coutinho FS, Carmo FMS, Vital CE, Mehta A, Fontes EPB, Barros EG, Ramos HJO (2019) Proteomic and metabolomic analysis of a drought tolerant soybean genotype from Brazilian savanna. Crop Breed Genet Genomics 1:e190022-32. https://doi.org/10.20900/cbgg20190022
Lopes MS, Araus JL, Van Heerden PDR, Foyer CH (2011) Enhancing drought tolerance in C4 crops. J Exp Bot 62:3135–3153. https://doi.org/10.1093/jxb/err105
Mesquita RO, Soares EA, Barros EG, Loureiro ME (2012) Method optimization for proteomic analysis of soybean leaf: improvements in identification of new and low-abundance proteins. Genet Mol Biol 35:353–361. https://doi.org/10.1590/S1415-47572012000200017
Mesquita RO, Coutinho SC, Vital CE, Nepomuceno AL, Williams TCR, Ramos HJO, Loureiro ME (2020) Physiological approaches to decipher the drought tolerance of a soybean genotype from Brazilian savana. Plant Physiol Biochem 151:132–143. https://doi.org/10.1016/j.plaphy.2020.03.004
Morris JS, Caldo KMP, Liang S, Facchini PJ (2021) PR10/Bet v1-like proteins as novel contributors to plant biochemical diversity. ChemBioChem 22(2):264–287. https://doi.org/10.1002/cbic.202000354
Oya T, Nepomuceno AL, Neumaier N, Farias JR, Tobita S, Ito O (2004) Drought tolerance characteristics of Brazilian soybean genotypes. Plant Prod Sci 7(2):129–137. https://doi.org/10.1626/pps.7.129
Pei ZM, Murata Y, Benning G, Thomine S, Klüsener B, Allen GJ et al (2000) Calcium channels activated by hydrogen peroxide mediate abscisic acid signaling in guard cells. Nature 406:731–734. https://doi.org/10.1038/35021067
Prince SJ, Murphy M, Mutava RN, Durnell LA, Valliyodan B, Shannon LG, Nguyen HT (2017) Root xylem plasticity to improve water use and yield in water-stressed soybean. J Exp Bot 88(8):2027–2036. https://doi.org/10.1093/jxb/erw472
Reddy KR, Brand D, Wijewardana C, Gao W (2017) Temperature effects on cotton seedling emergence, growth, and development. Agron J 109:1379–1387. https://doi.org/10.2134/agronj2016.07.0439
Reggiani R, Nebuloni M, Mattana M, Brambilla I (2000) Anaerobic accumulation of amino acids in rice roots: role of the glutamine synthetase/glutamate synthase cycle. Amino Acids 18:207–217. https://doi.org/10.1007/s007260050018
Rodrigues JM, Coutinho FS, Pinto IPA, Ferreira MA, Soares-Ramos JRL, Oliveira MGA, Fontes, EPB, Ramos HJO (2023) BiP-overexpressing soybean shows differential hypersensitivity response (HR) altering protein and metabolite profiles involved in the plant defense. BiP-overexpressing soybean shows differential hypersensitivity response (HR) altering protein and metabolite profiles involved in the plant defense Plant Physiology Reports. https://doi.org/10.1007/s40502-023-00717-9
Saab IN, Sharp RE, Pritchard J (1992) Effect of inhibition of abscisic acid accumulation on the spatial distribution of elongation in the primary root and mesocotyl of maize at low water potentials. Plant Physiol 99:26–33. https://doi.org/10.1104/pp.99.1.26
Scholander PE, Hammel HT, Bradstreet ED, Hemmingsen EA (1965) Sap pressure in vascular plants. Science 148:339–346. https://doi.org/10.1126/science.148.3668.339
Seo PJ, Lee A, Xiang F, Park C (2008) Molecular and functional profiling of arabidopsis pathogenesis-related genes: insights into their roles in salt response of seed germination. Plant Cell Physiol 49(3):334–344. https://doi.org/10.1093/pcp/pcn011
Sharp RE, Poroyko V, Hejlek LG, Spollen WG, Springer GK, Bohnert HJ, Nguyen HT (2004) Root growth maintenance during water deficits: physiology to functional genomics. J Exp Bot 55:2343–2351. https://doi.org/10.1093/jxb/erh276
Sun X, Luo X, Sun M, Chen C, Ding X, Wang X, Yang S, Yu Q, Jia B, Ji W, Cai H, Zhu Y (2014) A Glycine soja 14–3-3 protein GsGF14o participates in stomatal and root hair development and drought tolerance in Arabidopsis thaliana. Plant Cell Physiol 55(1):99–118. https://doi.org/10.1093/pcp/pct161
Tanaka N, Kato M, Tomioka R, Kurata R, Fukao Y, Aoyama T et al (2014) Characteristics of a root hair-less line of Arabidopsis thaliana under physiological stresses. J Exp Bot 65:1497–1512. https://doi.org/10.1093/jxb/eru014
Tardieu F (2016) Too many partners in root–shoot signals. Does hydraulics qualify as the only signal that feeds back over time for reliable stomatal control? New Phytol 212:802–804. https://doi.org/10.1111/nph.14292
Tenhaken R (2015) Cell wall remodeling under abiotic stress. Front Plant Sci 5:771. https://doi.org/10.3389/fpls.2014.00771
Thitisaksakul MS, Dong DM, Beckles, (2017) How rice glycogen synthase kinase-like 5 (OsGSK5) integrates salinity stress response to source-sink adaptation: a proposed model? Plant signal. Beha 12:1403708. https://doi.org/10.1080/15592324.2017.1403708
Trachsel S, Stamp P, Hind A (2010) Effect of high temperatures, drought and aluminum toxicity on root growth of tropical maize (Zea mays L.) seedlings. Maydica 55:249–260
Vadez V (2014) Root hydraulics: the forgotten side of roots in drought adaptation. Field Crops Res 165:15–24. https://doi.org/10.1016/j.fcr.2014.03.017
Valente MA, Faria JA, Soares-Ramos JR, Reis PA, Pinheiro GL, Piovesan ND, Morais AT, Menezes CC, Cano MA, Fietto LG, Loureiro ME, Aragão FJ, Fontes EP (2009) The ER luminal binding protein (BiP) mediates an increase in drought tolerance in soybean and delays drought-induced leaf senescence in soybean and tobacco. J Exp Bot 60(2):533–546. https://doi.org/10.1093/jxb/ern296
Vital CE, Gómez JD, Vidigal PM, Barros E, Pontes CSL, Vieira NM, Ramos HJO (2018) Phytohormone profiling by liquid chromatography coupled to mass spectrometry (LC/MS). Protocols.io. https://doi.org/10.17504/protocols.io.zgff3tn
Wang Y, Yang L, Chen X, Ye T, Zhong B, Liu R, Wu Y, Chan Z (2016) Major latex protein-like protein 43 (MLP43) functions as a positive regulator during abscisic acid responses and confers drought tolerance in Arabidopsis thaliana. J Exp Bot 67(1):421–434. https://doi.org/10.1093/jxb/erv477
Wijewardana C, Alsajri FA, Irby JT, Krutz LJ, Golden BR, Henry WB, Reddy KR (2019) Water deficit effects on soybean root morphology and early-season vigor. Agronomy 9:836. https://doi.org/10.3390/agronomy9120836
Wu Y, Gong W, Wang Y, Yong T, Yang F, Liu W et al (2018) Leaf area and photosynthesis of newly emerged trifoliolate leaves are regulated by mature leaves in soybean. J Plant Res 131:1–10. https://doi.org/10.1007/s10265-018-1027-8
Yang ZB, Rao IM, Horst WJ (2013) Interaction of aluminum and drought stress on root growth and crop yield on acid soils. Plant Soil 372:3–25. https://doi.org/10.1007/s11104-012-1580-1
Acknowledgements
The authors would like to thank the Núcleo de Análise de Biomoléculas (NuBioMol), the Universidade Federal de Viçosa, MG, Brazil, the Instituto de Biotecnologia Aplicada a Agropecuária (BIOAGRO), the Instituto Nacional de Ciência e Tecnologia em Interações Planta-Praga (INC-TIPP) and the Brazilian Soybean Genome Consortium (GENOSOJA). This work was funded by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Fundação de Amparo a Pesquisa de Minas Gerais (FAPEMIG) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq).
Funding
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
12298_2023_1307_MOESM2_ESM.docx
Temporal profile of leaf water potential in the morning (ψPLWP) in two soybean genotypes, one sensitive (BR16), and another tolerant (Embrapa 48) during the water deficit assay. Each point represents the mean + standard error (n = 3, where n represents the number of plants), IRR = irrigated, NI = non-irrigated (not irrigated after imposition of stress). (DOCX 49 KB)
12298_2023_1307_MOESM3_ESM.docx
Effect of drought stress on soluble sugar glucose, fructose, sucrose and starch contents. In (A), (C), (E) and (G) for leaves and in (B), (D), (F) and (H) for roots of the soybean genotypes under irrigation and drought (-1.0 MPa) conditions. Bars represent the mean ± standard error (n = 5, where n represents the number of plants). Different capital letters indicate significant differences between means of the same treatment in different genotypes. Lowercase letters show significant differences between means within the same genotype under different treatments (test-t, p <0.05). (DOCX 588 KB)
12298_2023_1307_MOESM4_ESM.docx
Differences in protein abundances of the roots from both genotypes under regular irrigation. Differences in the protein abundances expressed as fold change between the volume (%) of each protein spot of the drought-tolerant genotype Embrapa 48 relative to the sensitive BR16 genotype under normal irrigation conditions. Positive and negative fold change values indicate up- and down-regulation, respectively. Values of fold change for protein spots when presenting only in a genotype or treatment are shown as 10.00 or -10.00, respectively. (DOCX 20 KB)
12298_2023_1307_MOESM5_ESM.docx
Differences in the abundance of proteins in roots from drought-tolerant Embrapa 48 under drought conditions. Protein abundances were expressed as fold change between the volume (%) of each protein spot of the drought-tolerant genotype Embrapa 48 under drought related to normal irrigation conditions. Positive and negative fold change values indicate up- and down-regulation, respectively. Values of fold change for protein spots when presenting only in a genotype or treatment are shown as 10.00 or -10.00, respectively. (DOCX 18 KB)
12298_2023_1307_MOESM6_ESM.docx
Differences in the protein abundances of the roots from both genotypes under drought conditions. Protein abundances were described as fold change between the volume (%) from 2DE profiles of the drought-tolerant genotype Embrapa 48 related to sensitive BR16 under drought conditions. Positive and negative fold change values indicate up- and down-regulation, respectively. Values of fold change for protein spots when presenting only in a genotype or treatment are shown as 10.00 or -10.00, respectively. (DOCX 17 KB)
12298_2023_1307_MOESM7_ESM.docx
Differences in the abundance of the protein spots stained by Pro-Q® Diamond from the roots of both genotypes under regular irrigation. Differences in phosphoprotein abundances are described as fold changes between each phosphoprotein spot's volume (%). Comparations were performed between drought-tolerant genotype Embrapa 48 compared to sensitive BR16 under normal irrigation conditions. Positive and negative fold change values indicate up- and down-regulation, respectively. Values of fold change for protein spots when presenting only in a genotype or treatment are shown as 10.00 or -10.00, respectively. (DOCX 23 KB)
12298_2023_1307_MOESM8_ESM.docx
Differences in the abundance of the protein spots stained by Pro-Q® Diamond from root 2DE profiles of both genotypes under drought conditions. Differences in phosphoprotein abundances are indicated as fold changes between each phosphoprotein spot's volume (%). Differences in phosphoprotein abundances are described as fold changes between each phosphoprotein spot's volume (%). Comparations were performed between the drought-tolerant genotype Embrapa 48 related to sensitive BR16 drought conditions. Positive and negative fold change values indicate up- and down-regulation, respectively. Values of fold change for protein spots when presenting only in a genotype or treatment are shown as 10.00 or -10.00, respectively. (DOCX 21 KB)
12298_2023_1307_MOESM9_ESM.docx
Analysis of 2D Scores Plot by Partial Least Squares Discriminant Analysis (PLS-DA) of the metabolite profiles by LC/MS from soybean leaves. This plot shows the differences in the abundance of the metabolites analyzed by LC-MS in response to treatments. Blue color represents a decrease, and red color an increase. (DOCX 486 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Coutinho, F.S., Mesquita, R.O., Rodrigues, J.M. et al. Alterations in the root phenylpropanoid pathway and root–shoot vessel system as main determinants of the drought tolerance of a soybean genotype. Physiol Mol Biol Plants 29, 559–577 (2023). https://doi.org/10.1007/s12298-023-01307-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12298-023-01307-7