Abstract
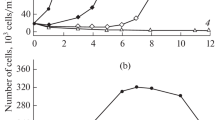
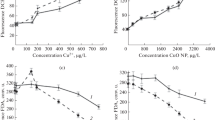
In the present study, changes were determined in morphological, structural–functional, and fluorescent parameters of Prorocentrum cordatum with the addition of CuO nanoparticles (NPs) and copper ions (CuSO4). A stimulating effect of low Cu2+ concentrations (30 μg L−1) on algal growth characteristics was observed. Higher Cu2+ concentration of 60–600 μg L−1 and CuO NPs concentration of 100–520 μg L−1 inhibited algal growth. Ionic copper is more toxic to P. cordatum than NPs. After 72 h of algae cultivation in the medium supplemented with CuSO4 and CuO NPs, EC50 values (calculated based on cell abundance) were of 60 and 300 μg L−1 (in terms of copper ions), respectively. Reduction in algal growth rate is due to disruption in cell cycle, changes in nuclear morphology, chromatin dispersion, and DNA damage. The studied pollutants slightly affected the efficiency of P. cordatum photosynthetic apparatus. Addition of the pollutants resulted in an increased production of reactive oxygen species (ROS). At a concentration of Cu2+ of 120 μg L−1 and a concentration of CuO NPs 0–300 μg L−1 of CuO NPs increase in ROS production is short-term with a decrease at later stages of the experiment. This is probably due to the activation of antioxidant mechanisms in cells and an increase in the concentration of carotenoids (peridinin) in cells. The high values of ROS production persisted throughout the experiment at sublethal copper concentrations (400–600 μg L−1 of CuSO4 and 520 μg L−1 of CuO NPs). Sublethal concentrations of pollutants caused restructuring of cell membranes in P. cordatum. Shedding of cell membranes (ecdysis) and formation of immobile stages (temporary or resting cysts) were recorded. The pronounced mechanical impact of NPs on the cell surface was observed such as—deformation and damage of a cell wall, its “wrinkling” and shrinkage, and adsorption of NP aggregates.






Similar content being viewed by others
Data availability
The datasets generated and/or analyzed during the present study are available from the corresponding author on reasonable request.
References
Adeleye AS, Keller AA (2016) Interactions between algal extracellular polymeric substances and commercial TiO2 nanoparticles in aqueous media. Environ Sci Technol 50(22):12258–12265. https://doi.org/10.1021/acs.est.6b03684
Adeleye AS, Conway JR, Garner K, Huang Y, Su Y, Keller AA (2016) Engineered nanomaterials for water treatment and remediation: Costs, benefits, and applicability. Chem Eng J 286:640–662. https://doi.org/10.1016/j.cej.2015.10.105
Ahner BA, Kong S, Morel FMM (1995) Phytochelatin production in marine algae. 1. An interspecies comparison. Limnol Oceanogr 40(4):649–657. https://doi.org/10.4319/lo.1995.40.4.0649
Andersen RA (2005) Algal culturing techniques. Elsevier Academic Press, New York
Barreto DM, Lombardi AT (2016) Environmentally relevant concentrations of TiO2 nanoparticles affected cell viability and photosynthetic yield in the Chlorophyceae Scenedesmus bijugus. Water Air Soil Pollut 227:1–11. https://doi.org/10.1007/s11270-016-3139-x
Barreto A, Carvalho A, Campos A, Osório H, Pinto E, Almeida T et al (2020) Effects of gold nanoparticles in gilthead seabream—a proteomic approach. Aquat Toxicol 221:105445. https://doi.org/10.1016/j.aquatox.2020.105445
Bravo I, Figueroa RI (2014) Towards an ecological understanding of dinoflagellate cyst functions. Microorganisms 2(1):11–32. https://doi.org/10.3390/microorganisms2010011
Bryantseva Yu, Lyakh A, Sergeeva A (2005) Calculation of volumes and surface areas of unicellular algae of the Black Sea. NAS of Ukraine, Institute of Biology of the Southern Seas, Sevastopol
Butz SV, Pinckney JL, Apte SC, Lead JR (2019) Uptake and impact of silver nanoparticles on the growth of an estuarine dinoflagellate, Prorocentrum minimum. NanoImpact 15:100181. https://doi.org/10.1016/j.impact.2019.100181
Cid A, Herrero C, Torres E, Abalde J (1995) Copper toxicity on the marine microalga Phaeodactylum tricornutum: effects on photosynthesis and related parameters. Aquat Toxicol 31(2):165–174. https://doi.org/10.1016/0166-445X(94)00071-W
Couet D, Pringault O, Bancon-Montigny C, Briant N, Poulichet FE, Delpoux S et al (2018) Effects of copper and butyltin compounds on the growth, photosynthetic activity and toxin production of two HAB dinoflagellates: the planktonic Alexandrium catenella and the benthic Ostreopsis cf. ovata. Aquat Toxicol 196:154–167. https://doi.org/10.1016/j.aquatox.2018.01.005
Davies AG (1974) The growth kinetics of Isochrysis galbana in cultures containing sublethal concentrations of mercuric chloride. J Mar Biol Assoc UK 54(1):157–169. https://doi.org/10.1017/S002531540002213X
Deeds JR, Landsberg JH, Etheridge SM, Pitcher GC, Longan SW (2008) Non-traditional vectors for paralytic shellfish poisoning. Mar Drugs 6(2):308–348. https://doi.org/10.3390/md6020308
Ebenezer V, Ki JS (2013) Quantification of toxic effects of the herbicide metolachlor on marine microalgae Ditylum brightwellii (Bacillariophyceae), Prorocentrum minimum (Dinophyceae), and Tetraselmis suecica (Chlorophyceae). J Microbiol 51(1):136–139. https://doi.org/10.1007/s12275-013-2114-0
Esperanza M, Cid Á, Herrero C, Rioboo C (2015) Acute effects of a prooxidant herbicide on the microalga Chlamydomonas reinhardtii: screening cytotoxicity and genotoxicity endpoints. Aquat Toxicol 165:210–221. https://doi.org/10.1016/j.aquatox.2015.06.004
Falkowski PG, Raven JA (2013) Aquatic photosynthesis. Princeton University Press. https://doi.org/10.1515/9781400849727
Ferradás Y, López M, Rey M, González MV (2014) Programmed cell death in kiwifruit stigmatic arms and its relationship to the effective pollination period and the progamic phase. Ann Bot 114(1):35–45. https://doi.org/10.1093/aob/mcu073
Fisher NS, Jones GJ, Nelson DM (1981) Effects of copper and zinc on growth, morphology, and metabolism of Asterionella japonica (Cleve) 1. J Exp Mar Biol Ecol 51(1):37–56. https://doi.org/10.1016/0022-0981(81)90153-2
Giri BR, Roy B, Babu SPS (2013) Evidence of apoptosis in Raillietina echinobothrida induced by methanolic extracts of three traditional medicinal plants of Northeast India. Exp Parasitol 134(4):466–473. https://doi.org/10.1016/j.exppara.2013.05.003
Grzebyk D, Berland B (1996) Influences of temperature, salinity and irradiance on growth of Prorocentrum minimum (Dinophyceae) from the Mediterranean Sea. J Plankton Res 18(10):1837–1849. https://doi.org/10.1093/plankt/18.10.1837
Grzebyk D, Denardou A, Berland B, Pouchus YF (1997) Evidence of a new toxin in the red-tide dinoflagellate Prorocentrum minimum. J Plankton Res 19(8):1111–1124. https://doi.org/10.1093/plankt/19.8.1111
Guo R, Lim WA, Ki JS (2016) Genome-wide analysis of transcription and photosynthesis inhibition in the harmful dinoflagellate Prorocentrum minimum in response to the biocide copper sulfate. Harmful Algae 57:27–38. https://doi.org/10.1016/j.hal.2016.05.004
Heil CA, Glibert PM, Fan C (2005) Prorocentrum minimum (Pavillard) Schiller: a review of a harmful algal bloom species of growing worldwide importance. Harmful Algae 4(3):449–470. https://doi.org/10.1016/j.hal.2004.08.003
Herzi F, Jean N, Zhao H, Mounier S, Mabrouk HH, Hlaili AS (2013) Copper and cadmium effects on growth and extracellular exudation of the marine toxic dinoflagellate Alexandrium catenella: 3D-fluorescence spectroscopy approach. Chemosphere 93(6):1230–1239. https://doi.org/10.1016/j.chemosphere.2013.06.084
Hofmann E, Wrench PM, Sharples FP, Hiller RG, Welte W, Diederichs K (1996) Structural basis of light harvesting by carotenoids: peridinin-chlorophyll-protein from Amphidinium carterae. Science 272(5269):1788–1791. https://doi.org/10.1126/science.272.5269.1788
Jiang W, Mashayekhi H, Xing B (2009) Bacterial toxicity comparison between nano- and micro-scaled oxide particles. Environ Pollut 157(5):1619–1625. https://doi.org/10.1016/j.envpol.2008.12.025
Jiménez C, Capasso JM, Edelstein CL, Rivard CJ, Lucia S, Breusegem S et al (2009) Different ways to die: cell death modes of the unicellular chlorophyte Dunaliella viridis exposed to various environmental stresses are mediated by the caspase-like activity DEVDase. J Exp Bot 60(3):815–828. https://doi.org/10.1093/jxb/ern330
Joonas E, Aruoja V, Olli K, Kahru A (2019) Environmental safety data on CuO and TiO2 nanoparticles for multiple algal species in natural water: filling the data gaps for risk assessment. Sci Total Environ 647:973–980. https://doi.org/10.1016/j.scitotenv.2018.07.446
Kajstura M, Halicka HD, Pryjma J, Darzynkiewicz Z (2007) Discontinuous fragmentation of nuclear DNA during apoptosis revealed by discrete “sub-G1” peaks on DNA content histograms. Cytometry A 71(3):125–131. https://doi.org/10.1002/cyto.a.20357
Khare P, Sonane M, Pandey R, Ali S, Gupta KC, Satish A (2011) Adverse effects of TiO2 and ZnO nanoparticles in soil nematode, Caenorhabditis elegans. J Biomed Nanotechnol 7(1):116–117. https://doi.org/10.1166/jbn.2011.1229
Klaine SJ, Alvarez PJ, Batley GE, Fernandes TF, Handy RD, Lyon DY et al (2008) Nanomaterials in the environment: behavior, fate, bioavailability, and effects. Environ Toxicol Chem Int J 27(9):1825–1851. https://doi.org/10.1897/08-090.1
Kreslavski VD, Los DA, Allakhverdiev SI, Kuznetsov VV (2012) Signaling role of reactive oxygen species in plants under stress. Russ J Plant Physiol 59(2):141–154. https://doi.org/10.1134/S1021443712020057
Lage OM, Sansonetty F, O’Connor JE, Parente AM (2001) Flow cytometric analysis of chronic and acute toxicity of copper (II) on the marine dinoflagellate Amphidinium carterae. Cytom Cytol 44(3):226–235. https://doi.org/10.1002/1097-0320(20010701)44:3%3c226::AID-CYTO1115%3e3.0.CO;2-9
Levy JL, Stauber JL, Jolley DF (2007) Sensitivity of marine microalgae to copper: the effect of biotic factors on copper adsorption and toxicity. Sci Total Environ 387(1–3):141–154. https://doi.org/10.1016/j.scitotenv.2007.07.016
Li M, Chen D, Liu Y, Chuang CY, Kong F, Harrison PJ, Zhu X, Jiang Y (2018) Exposure of engineered nanoparticles to Alexandrium tamarense (Dinophyceae): healthy impacts of nanoparticles via toxin-producing dinoflagellate. Sci Total Environ 610:356–366. https://doi.org/10.1016/j.scitotenv.2017.05.170
Li M, Jiang Y, Chuang CY, Zhou J, Zhu X, Chen D (2019) Recovery of Alexandrium tamarense under chronic exposure of TiO2 nanoparticles and possible mechanisms. Aquat Toxicol 208:98–108. https://doi.org/10.1016/j.aquatox.2019.01.007
Li S, Chu R, Hu D, Yin Z, Mo F, Hu T, Liu C, Zhu L (2020) Combined effects of 17β-estradiol and copper on growth, biochemical characteristics and pollutant removals of freshwater microalgae Scenedesmus dimorphus. Sci Total Environ 730:138597. https://doi.org/10.1016/j.scitotenv.2020.138597
Libralato G, Minetto D, Totaro S, Mičetić I, Pigozzo A, Sabbioni E et al (2013) Embryotoxicity of TiO2 nanoparticles to Mytilus galloprovincialis (Lmk). Mar Environ Res 92:71–78. https://doi.org/10.1016/j.marenvres.2013.08.015
Lim CY, Yoo YH, Sidharthan M, Ma CW, Bang IC, Kim JM et al (2006) Effects of copper (I) oxide on growth and biochemical compositions of two marine microalgae. J Environ Biol 27(3):461–466
Macedo RS, Lombardi AT, Omachi CY, Rörig LR (2008) Effects of the herbicide bentazon on growth and photosystem II maximum quantum yield of the marine diatom Skeletonema costatum. Toxicol in Vitro 22(3):716–722. https://doi.org/10.1016/j.tiv.2007.11.012
Machado MD, Soares EV (2014) Modification of cell volume and proliferative capacity of Pseudokirchneriella subcapitata cells exposed to metal stress. Aquat Toxicol 147:1–6. https://doi.org/10.1016/j.aquatox.2013.11.017
Marie D, Partensky F, Jacquet S, Vaulot D (1997) Enumeration and cell cycle analysis of natural populations of marine picoplankton by flow cytometry using the nucleic acid stain SYBR Green I. Appl Environ Microbiol 63(1):186–193. https://doi.org/10.1128/aem.63.1.186-193.1997
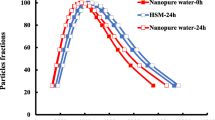
Matantseva O, Berdieva M, Kalinina V, Pozdnyakov I, Pechkovskaya S, Skarlato S (2020) Stressor-induced ecdysis and the cate cyst formation in the armoured dinoflagellates Prorocentrum cordatum. Sci Rep 10(1):1–17. https://doi.org/10.1038/s41598-020-75194-3
Melegari SP, Perreault F, Costa RHR, Popovic R, Matias WG (2013) Evaluation of toxicity and oxidative stress induced by copper oxide nanoparticles in the green alga Chlamydomonas reinhardtii. Aquat Toxicol 142:431–440. https://doi.org/10.1016/j.aquatox.2013.09.015
Miller RJ, Lenihan HS, Muller EB, Tseng N, Hanna SK, Keller AA (2010) Impacts of metal oxide nanoparticles on marine phytoplankton. Environ Sci Technol 44(19):7329–7334. https://doi.org/10.1021/es100247x
Morrill LC, Loeblich AR (1983) Formation and release of body scales in the dinoflagellate genus Heterocapsa. J Mar Biol Assoc UK 63(4):905–913. https://doi.org/10.1017/S0025315400071319
Motta AGC, do Amaral DF, Benvindo-Souza M, Rocha TL, Silva DDM (2020) Genotoxic and mutagenic effects of zinc oxide nanoparticles and zinc chloride on tadpoles of Lithobates catesbeianus (Anura: Ranidae). Environ Nanotechnol Monit Manag 14:100356. https://doi.org/10.1016/j.enmm.2020.100356
Murtey MD, Ramasamy P (2016) Sample preparations for scanning electron microscopy–life sciences. In: Janecek M, Kral R (eds) Modern electron microscopy in physical and life sciences. InTech, pp 161–185. https://doi.org/10.5772/61720
Navarro E, Baun A, Behra R, Hartmann NB, Filser J, Miao AJ, Sigg L (2008) Environmental behavior and ecotoxicity of engineered nanoparticles to algae, plants, and fungi. Ecotoxicology 17(5):372–386. https://doi.org/10.1007/s10646-008-0214-0
Novák Z, Harangi S, Baranyai E, Gonda S, B-Béres V, Bácsi I, (2020) Effects of metal quantity and quality to the removal of zinc and copper by two common green microalgae (Chlorophyceae) species. Psychol Res 68(3):227–235. https://doi.org/10.1111/pre.12422
Pakrashi S, Dalai S, Prathna TC, Trivedi S, Myneni R, Raichur AM et al (2013) Cytotoxicity of aluminium oxide nanoparticles towards fresh water algal isolate at low exposure concentrations. Aquat Toxicol 132:34–45. https://doi.org/10.1016/j.aquatox.2013.01.018
Perales-Vela HV, González-Moreno S, Montes-Horcasitas C, Cañizares-Villanueva RO (2007) Growth, photosynthetic and respiratory responses to sub-lethal copper concentrations in Scenedesmus incrassatulus (Chlorophyceae). Chemosphere 67(11):2274–2281. https://doi.org/10.1016/j.chemosphere.2006.11.036
Pogosyan SI, Matorin DN (2005) Variability in the condition of the photosynthetic system of the Black Sea phytoplankton. Oceanology 45:139–148
Pozdnyakov IA, Skarlato SO (2015) Analysis of the dinoflagellate Prorocentrum minimum transcriptome: identifying the members of the voltage-gated cation channel superfamily. Cell Tissue Biol 9(6):483–492. https://doi.org/10.1134/S1990519X15060085
Rioboo C, O’Connor JE, Prado R, Herrero C, Cid Á (2009) Cell proliferation alterations in Chlorella cells under stress conditions. Aquat Toxicol 94(3):229–237. https://doi.org/10.1016/j.aquatox.2009.07.009
Sekhon BS (2014) Nanotechnology in agri-food production: an overview. Nanotechnol Sci Appl 7:31–53. https://doi.org/10.2147/NSA.S39406
Sendra M, Moreno-Garrido I, Blasco J, Araújo CV (2018) Effect of erythromycin and modulating effect of CeO2 NPs on the toxicity exerted by the antibiotic on the microalgae Chlamydomonas reinhardtii and Phaeodactylum tricornutum. Environ Pollut 242:357–366. https://doi.org/10.1016/j.envpol.2018.07.009
Senicheva MI (2002) New and rare species of the diatoms and dinoflagellates in the Black Sea. Ekol Moray 62:25–29 (in Russian)
Shoman N, Solomonova E, Akimov A (2021) Application of structural, functional, fluorescent, and cytometric indicators for assessing physiological state of marine diatoms under different light growth conditions. Turk J Bot 45(5):511–521. https://doi.org/10.3906/bot-2102-39
Solomonova ES, Akimov AI, Shoman NYu (2018) Investigation of the applicability of the relative variable of chlorophyll fluorescence and fluorescein diacetate staining for the assessment and control of the state of algae culture on the example of Phaeodactylum tricornutum (Phaeodactylaceae). Botanicheski Jzhurnal 103(9):1177–1191. https://doi.org/10.7868/S0006813618090089 (in Russian)
Stauber JL, Florence TM (1987) Mechanism of toxicity of ionic copper and copper complexes to algae. Mar Biol 94(4):511–519. https://doi.org/10.1007/BF00431397
Terenko LM, Terenko GV (2008) Longterm dynamics of blooms microalgae in the coastal zone of the Odessa Bay (Black Sea). Morskoj Ekologicheskij Zhurnal 7(2):76–86 (in Russian)
Wan JK, Wan-Loy C, Kok YY, Cheong KW (2018) Assessing the toxicity of copper oxide nanoparticles and copper sulfate in a tropical Chlorella. J Appl Phycol 30(6):3153–3165. https://doi.org/10.1007/s10811-018-1408-3
Wang H, Joseph JA (1999) Quantifying cellular oxidative stress by dichlorofluorescein assay using microplate reader. Free Radic Biol Med 27:612–616. https://doi.org/10.1016/S0891-5849(99)00107-0
Wang J, Zhang X, Chen Y, Sommerfeld M, Hu Q (2008) Toxicity assessment of manufactured nanomaterials using the unicellular green alga Chlamydomonas reinhardtii. Chemosphere 73:1121–1128. https://doi.org/10.1016/j.chemosphere.2008.07.040
Wu T, Tang M (2018) Review of the effects of manufactured nanoparticles on mammalian target organs. J Appl Toxicol 38(1):25–40. https://doi.org/10.1002/jat.3499
Zhu Y, Xu J, Lu T, Zhang M, Ke M, Fu Z, Pan X, Qian H (2017) A comparison of the effects of copper nanoparticles and copper sulfate on Phaeodactylum tricornutum physiology and transcription. Environ Toxicol Pharmacol 56:43–49. https://doi.org/10.1016/j.etap.2017.08.029
Funding
This work was supported by A.O. Kovalevsky Institute of Biology of the Southern Seas of RAS (state projects No. 121041400077-1 “Functional, metabolic, and toxicological aspects of hydrobionts’ existence and their populations in habitats with different physical and chemical regime” and No. 121030300149-0 “Investigation of the mechanisms of controlling production processes in biotechnological complexes with the aim of developing the scientific foundations for the production of biologically active substances and technical products of marine genesis”), within the program “Prioritet – 2030” of Sevastopol State University (strategic project № 3, № 121121700318-1).
Author information
Authors and Affiliations
Contributions
NS—formal analysis, investigation, and writing—review and editing. ES—methodology, investigation, formal analysis, and writing—original draft. AA—investigation, formal analysis, and writing—review and editing. OR—methodology and investigation, YM—investigation.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest. The authors have no financial or proprietary interests in any material discussed in this article.
Ethical approval
The present study does not need an ethical approval as the studied organism is microalgae.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shoman, N., Solomonova, E., Akimov, A. et al. Responses of Prorocentrum cordatum (Ostenfeld) Dodge, 1975 (Dinoflagellata) to copper nanoparticles and copper ions effect. Physiol Mol Biol Plants 28, 1625–1637 (2022). https://doi.org/10.1007/s12298-022-01228-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12298-022-01228-x