Abstract
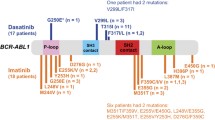
Tyrosine kinase inhibitors (TKI’s) are currently the drug of choice for management of chronic myeloid leukemia. Imatinib is the most commonly used first line TKI in India. Mutations leading to resistance to imatinib are the most common cause for imatinib failure. We studied pattern of kinase domain mutations in 40 patients of CML who either lost their response or did not achieve it in defined timepoints. Loss of molecular response was the most common indication for asking mutation analysis. Sixteen patients were found to have detectable mutations. M351T was the most common tyrosine kinase mutation followed by Y253H and H396R. Two patients had 2 mutations simultaneously. M351T is the most common mutation in our patient population.

Similar content being viewed by others
References
Bansal S, Prabhash K, Parikh P (2013) Chronic myeloid leukemia data from India. Indian J Med Paediatr Oncol 34(3):154–158
Lee SJ (2000) Chronic myelogenous leukaemia. Br J Haematol 111:993–1009
Druker B, Lydon N (2000) Lessons learned from the development of an Abl tyrosine kinase inhibitor for chronic myelogenous leukemia. J Clin Invest 105:3–7
Cohen MH, Williams G, Johnson JR et al (2002) Approval summary for imatinib mesylate capsules in the treatment of chronic myelogenous leukemia. Clin Cancer Res 8:935–942
Kantarjian H, O’Brien S, Jabbour E et al (2012) Improved survival in chronic myeloid leukemia since the introduction of imatinib therapy: a single institution historical experience. Blood 119(9):1981–1987
Kantarjian H, Hochhaus A, Saglio G, Souza C, Flinn I, Stenke L et al (2011) Nilotinib versus imatinib for the treatment of patients with newly diagnosed chronic phase, Philadelphia chromosome-positive, chronic myeloid leukaemia: 24-month minimum follow-up of the phase 3 randomised ENESTnd trial. Lancet Oncol 12:841–851
Hochhaus A, O’Brien S, Guilhot F, Druker B, Branford S, Foroni L et al (2009) Six-year follow-up of patients receiving imatinib for the first-line treatment of chronic myeloid leukemia. Leukemia 23:1054–1061
Bhamidipati PK, Kantarjian H, Cortes J et al (2013) Management of imatinib resistant patients with chronic myeloid leukemia. Ther Adv Hematol 4(2):103–117
Soverini S, Hochhaus A, Nicolini FE, Franz G, Lange T, Saglio G et al (2011) BCR–ABL kinase domain mutation analysis in chronic myeloid leukemia patients treated with tyrosine kinase inhibitors: recommendations from an expert panel on behalf of European leukemiaNet. Blood 118(5):1208–1215
Elias MH, Baba AA, Azian H et al (2014) BCR–ABL kinase domain mutations, including 2 novel mutations in imatinib resistant Malaysian chronic myeloid leukemia patients—frequency and clinical outcome. Leuk Res 38(4):454–549
Soverini S, Colarossi S, Gnani A, Rosti G, Castagnetti F, Poerio A et al (2006) Contribution of ABL kinase domain mutations in imatinib resistance in different subsets of Philadelphia positive patients: by the GIMEMA working party on Chronic Myeloid Leukemia. Clin Cancer Res 12:7374–7379
Branford S, Rudzki Z, Walsh S, Parkinson I, Grigg A, Szer J et al (2003) Detection of BCR–ABL mutations in patients with CML treated with imatinib is virtually always accompanied by clinical resistance, and mutations in the ATP phosphate binding loop(p-loop) are associated with poor prognosis. Blood 102:276–283
Babu G (2013) Report of patients with chronic myeloid leukemia Kidwai memorial institute of oncology Bangalore over 15 years. Indian J Med Paediatr Oncol 34(3):196–198
Prabhash K, Vora T, Chadyalpatil NS et al (2009) Patterns of imatinib resistance mutation analysis in chronic myeloid leukemia patients on imatinib at the time of loss of response to the drug in Asian Indian subjects. J Clin Oncol 27:7079
Srivastava S, Dutta S (2013) Imatinib mesylate resistance and mutations: an Indian experience. Indian J Med Paediatr Oncol 34(3):213–220
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declared no conflict of interest.
Ethical Approval
All procedures performed in this study were in accordance with the ethical standards of the institution and with the 1964 Helsinki’s Declaration.
Informed Consent Statement
Informed consent was obtained for all individuals participating in this study.
Rights and permissions
About this article
Cite this article
Tripathi, A.K., Verma, S.P. & Kumar, N. Mutation Analysis in Chronic Myeloid Leukemia Patient in Chronic Phase on Imatinib Having Delayed Achievement of Milestones or Loss of Response. Indian J Hematol Blood Transfus 33, 316–320 (2017). https://doi.org/10.1007/s12288-016-0755-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12288-016-0755-y