Abstract
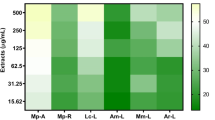
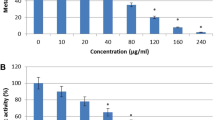
This study examined the effect of host extract in the culture medium on anti-candidal activity of Phomopsis sp. ED2, previously isolated from the medicinal herb Orthosiphon stamineus Benth. Interestingly, upon addition of aqueous host extract to the culture medium, the ethyl acetate extract prepared from fermentative broth exhibited moderate anti-candidal activity in a disc diffusion assay. The minimal inhibitory concentration of this extract was 62.5 μg/ml and it only exhibited fungistatic activity against C. albicans. In the time-kill study, a 50% growth reduction of C. albicans was observed at 31.4 h for extract from the culture incorporating host extract. In the bioautography assay, only one single spot (Rf 0.59) developed from the extract exhibited anti-candidal activity. A spot with the a similar Rf was not detected for the crude extract from YES broth without host extract. This indicated that the terpenoid anti-candidal compound was only produced when the host extract was introduced into the medium. The study concluded that the incorporation of aqueous extract of the host plant into the culture medium significantly enhanced the anti-candidal activity of Phomopsis sp. ED2.
Similar content being viewed by others
References
Carrado, M. and Rodrigues, K.F. 2004. Antimicrobial evaluation of fungal extracts produced by endophytic strains of Phomopsis sp. J. Basic Microbiol. 44, 157–160.
Castillo, U.F., Strobel, G.A., Ford, E.J., Hess, W.M., Porter, H., Jensen, J.B., Albert, H., Robinson, R., Condron, M.A.M., Teplow, D.B., and et al. 2002. Munumbicins, wide spectrum antibiotics produced by Streptomyces NRRL 30562, endophytic on Kennedia nigriscans. Microbiology 148, 2675–2685.
Ding, G., Liu, S., Guo, L., Zhou, Y., and Che, Y. 2008. Antifungal metabolites from the plant endophytic fungus Pestalotiopsis foedan. J. Nat. Prod. 71, 615–618.
Espinel-Ingroff, A., Kish, J.C.W., Kerkering, T.M., Fromtling, R.A., Bartizal, K., Galgiani, J.N., Villareal, K., Pfaller, M.A., Gerarden, T., and Rinaldi, M.G. 1992. Collaborative comparison of broth macrodilution and microdilution antifungal susceptibility tests. J. Clin. Microbiol. 30, 3138–3145.
Farr, D.F., Castlebury, L.A., and Rossman, A.Y. 2002. Morphological and molecular characterization of Phomopsis vaccinii and additional isolates of Phomopsis from blueberry and cranberry in the eastern United States. Mycologia 94, 494–504.
Fostel, J.M. and Lartey, P.A. 2000. Emerging novel antifungal agents. Ther. Focus 5, 25–32.
Gond, S.K., Verma, V.C., Kumar, A., Kumar, V., and Kharwar, R.N. 2007. Study of endophytic fungal community from different parts of Aegle marmelos Correae (Rutaceae) from Varanasi (India). World J. Microb. Biot. 23, 1371–1375.
Guo, Z., Hua, R., Bai, Y., Wu, X., Cao, H., Li, X., Wu, Y., and Tang, J. 2011. Screening and evaluation of antiphylopathogenic activity of endophytic fungi from live foliages of Ginkgo biloba L. Afr. J. Microbiol. Res. 5, 1686–1690.
Gupte, M., Kulkarni, P., and Ganguli, B.M. 2002. Antifungal antibiotics. Appl. Microbiol. Biotechnol. 58, 46–57.
Ho, W.H., To, P.C., and Hyde, K.D. 2003. Induction of antibiotic production of freshwater fungi using mix-culture fermentation. Fungal Divers. 12, 45–51.
Horn, W.S., Simmonds, M.S.J., Schwartz, R.E., and Blaney, W.M. 1995. Phomopsichalasin, a novel antimicrobial agent from an endophytic Phomopsis sp. Tetrahedron 51, 3969–3978.
Huang, Z., Cai, X., Shao, C., She, Z., Xia, X., Chen, Y., Yang, J., Zhou, S., and Lin, Y. 2008. Chemistry and weak antimicrobial activities of phomopsins produced by mangrove endophytic fungus Phomopsis sp. ZSU-H76. Phytochem. 69, 1604–1608.
Jorgensen, J.H. and Turnidge, J.D. 2007. Susceptibility test methods: dilution and disk diffusion methods, pp. 1152–1172. In Murray, P.R., Baron, E.J., Jorgensen, J.H., Landry, M.L., and Pfaller, M.A. (eds.), Manual of Clinical Microbiology. ASM Press, Washington, D.C., USA.
Kim, J. and Sudbery, P. 2011. Candida albicans, a major human fungal pathogen. J. Microbiol. 49, 171–177.
Liu, C.H., Liu, T.T., Yuan, F.F., and Gu, Y.C. 2010. Isolating endophytic fungi from evergreen plants and determining their antifungal activities. Afr. J. Microbiol. Res. 21, 2243–2248.
Liu, J.Y., Song, Y.C., Zhang, Z., Wang, L., Guo, Z.J., Zou, W.X., and Tan, R.X. 2004. Aspergillus fumigatus CY018, an endophytic fungus in Cynodon dactylon as a versatile producer of new and bioactive metabolites. J. Biot. 114, 279–287.
Mourad, M.H.E.S. 2010. New bioactive compounds from Verticillium alboatrum and Verticillium leptobactrum. Aust. J. Basic Appl. Sci. 4, 2166–2175.
Ninthya, K. and Muthumary, J. 2011. Bioactive metabolites produced by Phomopsis sp. an endophytic fungus in Allamanda cathartica Linn. Recent Res. Sci. Technol. 3, 44–48.
Oyama, M. and Kubota, K. 1993. Induction of antibiotic production by protease in Bacillus brevis (ATCC8185). J. Biochem. 113, 637–641.
Qiu, Z.Q., Cao, L.X., Tan, H.M., and Zhou, S.M. 2006. Isolation and characterization of endophytic Stretomyces sp. S5 with herbicidal activity from tomato roots. Chin. J. Agr. Biotechnol. 3, 7–12.
Rukachaisirikul, V., Sommart, U., Phongpaichit, S., Sakayaroj, J., and Kirtikara, K. 2007. Metabolites from endophytic fungus Phomopsis sp. PSU-D15. Phytochem. 69, 783–787.
Schulz, B., Boyle, C., Draeger, S., Rommert, A.K., and Krohn, K. 2002. Endophytic fungi: a source of novel biologically active secondary metabolites. Mycol. Res. 106, 996–1004.
Sreekanth, D., Sushim, G.K., Syed, A., Khan, B.M., and Ahmad, A. 2011. Molecular and morphological characterization of a taxol-producing endophytic fungus, Gliocladium sp. from Taxus baccata. Mycobiology 39, 151–157.
Strobel, G.A. 2003. Endophytes as sources of bioactive products. Microb. Infect. 5, 535–544.
Tan, R.X. and Zou, W.X. 2001. Endophytes: a rich source of functional metabolites. Nat. Prod. Rep. 18, 448–459.
Tansuwan, S., Pornpakakul, S., Roengsumran, S., Petsom, A., Muangsin, N., Sihanomta, P., and Chaichit, N. 2007. Antimalarial benzoquinone from an endophytic fungus Xylaria sp. J. Nat. Prod. 70, 1620–1623.
Tong, W.Y., Darah, I., and Latiffah, Z. 2011. Antimicrobial activities of endophytic fungal isolates from medicinal herb Orthosiphon stamineus Benth. J. Med. Plants Res. 5, 831–836.
Valgas, C., Souza, S.M., Smania, E.F.A., and Smania, A.J. 2007. Screening methods to determine antibacterial activity of natural products. Braz. J. Microbiol. 38, 369–380.
Weber, R.W., Kappe, R., Paululat, T., Moskev, E., and Anke, H. 2007. Anti-candida metabolites from endophytic fungi. Phytochem. 68, 886–892.
Yu, H., Zhang, L., Li, L., Zheng, C., Guo, L., Li, W., Sun, P., and Qin, L. 2010. Recent developments and future prospects of antimicrobial metabolites produced by endophytes. Microbiol. Res. 165, 437–449.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yenn, T.W., Lee, C.C., Ibrahim, D. et al. Enhancement of anti-candidal activity of endophytic fungus Phomopsis sp. ED2, isolated from Orthosiphon stamineus Benth, by incorporation of host plant extract in culture medium. J Microbiol. 50, 581–585 (2012). https://doi.org/10.1007/s12275-012-2083-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-012-2083-8