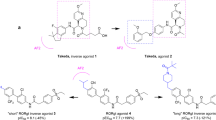
Abstract
Thirty two thiourea derivatives were prepared and their agonistic activities on the retinoic acid receptor-related orphan receptor α (RORα) were evaluated. The replacement of the 3-allyl-2-imino-thiazolidin-4-one moiety of the lead compound CGP52608 (1) with various functional group substituted aromatic rings, improved the agonistic activity of RORα. Among the prepared derivatives, 1-methyl-3-(4-phenoxy-benzyl)-thiourea (32) showed 2.6-fold higher agonistic activity than CGP52608 in the RORα-activation assay.
Similar content being viewed by others
References
Armstrong, J.D., III, Wolfe, C. N., Keller, J. L., Lynch, J., Bhupathy, M., Volante, R. P., and De Vita, R. J., A novel synthesis of disubstituted ureas using titanium (IV) isopropoxide and sodium borohydride. Tetrahedron Letts., 38, 1531–1532 (1997).
Becker-Andre, M., Andre, E., and DeLamarter, J. F., Identification of nuclear receptor mRNAs by RT-PCR amplification of conserved zinc-finger motif sequences. Biochem. Biophys. Res. Commun., 194, 1371–1379 (1984).
Carlberg, C., Hooft van Huijsduijnen, R., Staple, J. K., De-Lamarter, J. F., and Becker-Andre, M., RZRs, a new family of retinoid-related orphan receptors that function as both monomers and homodimers.Mol. Endocrinol., 8, 757–770 (1994).
Eberl, G. and Littman, D. R., The role of the nuclear hormone receptor RORgammat in the development of lymph nodes and Peyer’s patches. Immunol. Rev., 195, 81–90 (2003).
Giguere, V., Tini, M., Flock, G., Ong, E., Evans, R. M., and Otulakowski, G., Isoform-specific amino-terminal domains dictate DNA-binding properties of ROR alpha, a novel family of orphan hormone nuclear receptors. Genes Dev., 8, 538–553 (1994).
Giguere, V., Beatty, B., Squire, J., Copeland, N. G., and Jenkins, N. A., The orphan nuclear receptor ROR alpha (RORA) maps to a conserved region of homology on human chromosome 15q21-q22 and mouse chromosome 9. Genomics, 28, 596–598 (1995).
Gold, D.A., Baek, S. H., Schork, N. J., Rose, D. W., Larsen, D. D., Sachs, B. D., Rosenfeld, M. G., and Hamilton, B. A., RORalpha coordinates reciprocal signaling in cerebellar development through sonic hedgehog and calcium-dependent pathways. Neuron, 40, 1119–1131 (2003).
He, Y.W., Deftos, M. L., Ojala, E. W., and Bevan, M. J., RORgamma t, a novel isoform of an orphan receptor, negatively regulates Fas ligand expression and IL-2 production in T cells. Immunity, 9, 797–806 (1998).
He, Y.W., Orphan nuclear receptors in T lymphocyte development. J. Leukoc. Biol., 72, 440–452 (2002).
Hirose, T., Smith, R. J., and Jetten, A. M., ROR gamma: the third member of ROR/RZR orphan receptor subfamily that is highly expressed in skeletal muscle. Biochem. Biophys. Res. Commun., 205, 1976–1983 (1994).
Jetten, A.M., Kurebayashi, S., and Ueda, E., The ROR nuclear orphan receptor subfamily: critical regulators of multiple biological processes. Prog. Nucl. Acid Res., 69, 205–247 (2001).
Jetten, A.M. and Ueda, E., Retinoid-related orphan receptors (RORs): roles in cell survival, differentiation and disease. Cell Death Differ., 9, 1167–1177 (2002).
Jetten, A.M., Recent advances in the mechanisms of action and physiological functions of the retinoid-related orphan receptors (RORs). Curr. Drug Targets Inflamm. Allergy, 3, 395–412 (2004).
Kallen, J.A., Schlaeppi, J. M., Bitsch, F., Geisse, S., Geiser, M., Delhon, I., and Fournier, B., X-ray structure of the hRORalpha LBD at 1.63 A: structural and functional data that cholesterol or a cholesterol derivative is the natural ligand of RORalpha. Structure, 10, 1697–1707 (2002).
Kallen, J., Schlaeppi, J. M., Bitsch, F., Delhon, I., and Fournier, B., Crystal structure of the human RORalpha Ligand binding domain in complex with cholesterol sulfate at 2.2 A. J. Biol. Chem., 279, 14033–14038 (2004).
Kim, E.J., Yoo, Y. G., Yang, W. K., Lim, Y. S., Na, T. Y., Lee, I. K., and Lee, M.-O., Transcriptional activation of HIF-1 by RORalpha and its role in hypoxia signaling. Arterioscler. Thromb. Vasc. Biol., 28, 1796–1802 (2008).
Lau, P., Fitzsimmons, R. L., Raichur, S., Wang, S. C., and Lechtken, A., The orphan nuclear receptor, RORalpha, regulates gene expression that controls lipid metabolism: staggerer (SG/SG) mice are resistant to diet-induced obesity. J. Biol. Chem., 283, 18411–18421 (2008).
Medvedev, A., Yan, Z. H., Hirose, T., Giguere, V., and Jetten, A. M., Cloning of a cDNA encoding the murine orphan receptor RZR/ROR gamma and characterization of its response element. Gene, 181, 199–206 (1996).
Meyer, T., Kneissel, M., Mariani, J., and Fournier, B., In vitro and in vivo evidence for orphan nuclear receptor RORalpha function in bone metabolism. Proc. Natl. Acad. Sci. U. S. A., 97, 9197–9202 (2000).
Missbach, M., Jagher, B., Sigg, I., Nayeri, S., Carlberg, C., and Wiesenberg. I., Thiazolidine diones, specific ligands of the nuclear receptor retinoid Z receptor/retinoid acid receptor-related orphan receptor alpha with potent antiarthritic activity. J. Biol. Chem., 271, 13515–13522 (1996).
Ortiz, M.A., Piedrafita, F. J., Pfahl, M., and Maki, R., TOR: a new orphan receptor expressed in the thymus that can modulate retinoid and thyroid hormone signals. Mol. Endocrinol., 9, 1679–1691 (1995).
Raspe, E., Duez, H., Gervois, P., Fievet, C., Fruchart, J. C., Besnard, S., Mariani, J., Tedgui, A., and Staels, B., Transcriptional regulation of apolipoprotein C-III gene expression by the orphan nuclear receptor RORalpha. J. Biol. Chem., 276, 2865–2871 (2001).
Steinhilber, D., Brungs, M., Werz, O., Wiesenberg, I., Danielsson, C., Kahlen, J.-P., Nayeri, S., Schrader, M., and Carlberg, C., The nuclear receptor for melatonin represses 5-lipoxygenase gene expression in human B lymphocytes. J. Biol. Chem., 270, 7037–7040 (1995).
Vu-Dac, N., Gervois, P., Grötzinger, T., De Vos, P., Schoonjans, K., and Fruchart, J. C., Transcriptional regulation of apolipoprotein A-I gene expression by the nuclear receptor RORalpha. J. Biol. Chem., 272, 22401–22404 (1997).
Wada, T., Kang, H. S., Jetten, A. M., and Xie, W., The emerging role of nuclear receptor RORalpha and its crosstalk with LXR in xeno- and endobiotic gene regulation.Exp. Biol. Med., 233, 1191–1201 (2008).
Wang. Y., Solt, L. A., and Burris, T. P., Regulation of FGF21 expression and secretion by retinoic acid receptor-related orphan receptor alpha. J. Biol. Chem., 285, 15668–15673 (2010).
Wiesenberg, I., Missbach, M., Kahlen, J.-P., Schraeder, M., and Carlberg, C., Transcriptional activation of the nuclear receptor RZR alpha by the pineal gland hormone melatonin and identification of CGP 52608 as a synthetic ligand. Nucleic Acids Res., 23, 327–333 (1995).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Park, Y., Hong, S., Lee, M. et al. N-methylthioureas as new agonists of retinoic acid receptor-related orphan receptor. Arch. Pharm. Res. 35, 1393–1401 (2012). https://doi.org/10.1007/s12272-012-0809-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-012-0809-0