Abstract
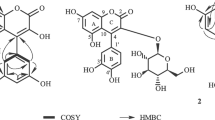
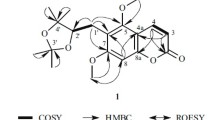
A new coumarin, 7-[(E)-3′,7′-Dimethyl-6′-oxo-2′,7′-octadienyl]oxy coumarin (1), together with three known compounds, schinilenol (2), schinindiol (3) and 7-[(E)-7′-hydroxy-3′,7′-dimethylocta-2′,5′-dienyloxy]-coumarin (4) were isolated from the methylene chloride fraction of Z. schinifolium by normal and reverse phase column chromatographies. Their structures were determined on the basis of physical and spectroscopic evidences. Compound 1 (IC50 8.10 μM) showed potent cytotoxicity compared to auraptene (IC50 55.36 μM) against Jurkat T cells. The other isolated compounds 2 and 4 exhibited weak cytotoxicities.
Similar content being viewed by others
References
Chang, C.-T., Doong, S.-L., Tsai, I.-L., and Chen, I.-S., Coumarins and anti-HBV constituents from Zanthoxylum schinifolium. Phytochemistry, 45, 1419–1422 (1997).
Chen, I. S., Lin, Y. C., Tsai, I. L., Teng, C. M., Ko, F. N., Ishikawa, T., and Ishii, H., Coumarins and anti-platelet aggregation constituents from Zanthoxylum schinifolium. Phytochemistry, 39, 1091–1097 (1995).
Coates, R. M. and Melvin, L. S. Jr., Total synthesis of (±)- marmin and related coumarin monoterpenes. Tetrahedron, 26, 5699–5706 (1970).
Han, M.-K., Kim, S.-I., and Ahn, Y.-J., Insecticidal and antifeedant activities of medicinal plant extracts against Attagenus unicolor japonicus. J. Stored Prod. Res., 42, 15–22 (2006).
Jo, Y. S., Huong, D. T. L., Bae, K., Lee, M. K., and Kim, Y. H., Monoamine oxidase inhibitory coumarin from Zanthoxylum schinifolium. Planta Med., 68, 84–85 (2002).
Jun, D. Y., Kim, J. S., Park, H. S., Han, C. R., Fang, Z., Woo, M. H., Rhee, I. K., and Kim, Y. H., Apoptogenic activity of auraptene of Zanthoxylum schinifolium toward human acute leukemia Jurkat T cells is associated with ER stress-mediated caspase-8 activation that stimulates mitochondria-dependent or -independent caspase cascade. Carcinogenesis, 28, 1303–1313 (2007).
Kim, D. S., Bang, W., and Yeum, D. M., Degradation of carcinogenic nitrosamine formation factor by natural food components. 1. Nitrite-scavenging of vegetable extracts. Bull. Korean Fish Soc., 20, 463–468 (1987).
Kim, J.-S., Jun, D. Y., Woo, M.-H., Rhee, I.-K., and Kim, Y. H., Chemical composition and antitumor apoptogenic activity of methylene chloride extracts from the leaves of Zanthoxylum schinifolium. J. Life Sci., 16, 546–554 (2006).
Liu, S. L., Wei, L. X., Wang, D., and Gao, C. Y., Studies on the chemical constituents from the peel of Zanthoxylum schinifolium Sieb et Zucc. Yao Xue Xue Bao, 26, 836–840 (1991).
Liu, Z. L., Chu, S. S., and Jiang, G. H., Feeding deterrents from Zanthoxylum schinifolium against two storedproduct insects. J. Agric. Food Chem., 57, 10130–10133 (2009).
Mun, S.-I., Ryu, H.-S., Lee, H.-J., and Choi, J.-S., Further screening for antioxidant activity of vegetable plants and its active principles from Zanthoxylum schinifolium. J. Korean Soc. Food Nutr., 23, 466–471 (1994).
Park, E.-J., Nan, J.-X., Kim, J.-Y., Kang, H.-C., Choi, J. H., Lee, S. J., Lee, B. H., Kim, S. J., Lee, J. H., Kim, Y. C., and Sohn, D. H., The ethanol soluble part of a hot water extract from Artemisia iwayomogi inhibits liver fibrosis induced by carbon tetrachloride in rats. J. Pharm. Pharmacol., 52, 875–881 (2000).
Quader, M. A., El-Turbi, J. A., Armstrong, J. A., Gray, A. I., and Waterman, P. G., Coumarins and their taxonomic value in the genus Phebalium. Phytochemistry, 31, 3083–3089 (1992).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Min, B.K., Hyun, D.G., Jeong, S.Y. et al. A new cytotoxic coumarin, 7-[(E)-3′,7′-dimethyl-6′-oxo-2′,7′-octadienyl] oxy Coumarin, from the leaves of Zanthoxylum schinifolium . Arch. Pharm. Res. 34, 723–726 (2011). https://doi.org/10.1007/s12272-011-0504-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-011-0504-6