Abstract
Cardiac hypertrophy, in its aspects of localized thickening of the interventricular septum and concentric increase of the left ventricle, constitutes a risk factor of heart failure. Myocardial hypertrophy, in the presence of different degree of myocardial fibrosis, is paralleled by significant molecular, cellular, and histological changes inducing alteration of cardiac extracellular matrix composition as well as sarcomeres and cytoskeleton remodeling. Previous studies indicate osteopontin (OPN) and more recently survivin (SURV) overexpression as the hallmarks of heart failure although SURV function in the heart is not completely clarified. In this study, we investigated the involvement of SURV in intracellular signaling of hypertrophic cardiomyocytes and the impact of its transcriptional silencing, laying the foundation for novel target gene therapy in cardiac hypertrophy. Oligonucleotide-based molecules, like theranostic optical nanosensors (molecular beacons) and siRNAs, targeting SURV and OPN mRNAs, were developed. Their diagnostic and therapeutic potential was evaluated in vitro in hypertrophic FGF23-induced human cardiomyocytes and in vivo in transverse aortic constriction hypertrophic mouse model. Engineered erythrocyte was used as shuttle to selectively target and transfer siRNA molecules into unhealthy cardiac cells in vivo. The results highlight how the SURV knockdown could negatively influence the expression of genes involved in myocardial fibrosis in vitro and restores structural, functional, and morphometric features in vivo. Together, these data suggested that SURV is a key factor in inducing cardiomyocytes hypertrophy, and its shutdown is crucial in slowing disease progression as well as reversing cardiac hypertrophy. In the perspective, targeted delivery of siRNAs through engineered erythrocytes can represent a promising therapeutic strategy to treat cardiac hypertrophy.
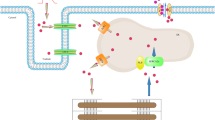
Graphical abstract
Theranostic SURV molecular beacon (MB-SURV), transfected into FGF23-induced hypertrophic human cardiomyocytes, significantly dampened SURV overexpression. SURV down–regulation determines the tuning down of MMP9, TIMP1 and TIMP4 extracellular matrix remodeling factors while induces the overexpression of the cardioprotective MCAD factor, which counterbalance the absence of pro-survival and anti-apoptotic SURV activity to protect cardiomyocytes from death. In transverse aortic constriction (TAC) mouse model, the SURV silencing restores the LV mass levels to values not different from the sham group and counteracts the progressive decline of EF, maintaining its values always higher with respect to TAC group. These data demonstrate the central role of SURV in the cardiac reverse remodeling and its therapeutic potential to reverse cardiac hypertrophy.









Similar content being viewed by others
Abbreviations
- ANP:
-
Atrial natriuretic peptide
- BBQ:
-
Blackberry Quencher 650
- β-MHC:
-
Beta- myosin heavy chain
- BNP:
-
Brain natriuretic peptide
- CLSM:
-
Confocal laser scanning microscopy
- CSA:
-
Cross-sectional area
- EF:
-
Ejection fraction
- EGR1:
-
Early growth response 1
- EMHV:
-
Erythro-Magneto-Ha virosome
- ERK:
-
Extracellular signal-regulated kinases
- FGF23:
-
Fibroblast growth factor 23
- FHA:
-
Filamentous hemagglutinin
- FS:
-
Fractional shortening
- GADPH:
-
Glyceraldehyde-3-phosphate dehydrogenase
- HCM:
-
Human cardiomyocyte
- HF:
-
Heart failure
- HRP:
-
Horseradish peroxidase
- HW:
-
Heart weight
- IHC:
-
Immunohistochemistry
- IVSTd :
-
Interventricular septum thickness, diastolic
- LC:
-
Left carotid
- LV:
-
Left ventricle
- LVAD:
-
Left ventricular assist device
- LVPWTd :
-
Left ventricle posterior wall thickness, diastolic
- MB:
-
Molecular beacon
- MCAD:
-
Medium-chain acyl-CoA dehydrogenase
- MMP:
-
Matrix metalloproteinase
- NO:
-
Nitric oxide
- NOX4:
-
NADPH oxidase 4
- OPN:
-
Osteopontin
- PFS:
-
Pixel fluorescence signal
- RC:
-
Right carotid
- sk-α-actin:
-
Skeletal-alpha-actin
- SPION:
-
Super-paramagnetic iron oxide nanoparticles
- SURV:
-
Survivin
- TAC:
-
Transverse aortic constriction
- TIMP:
-
Tissue inhibitor of metalloproteinase
- TL:
-
Tibia length
- UHFUS:
-
Ultra high-frequency ultrasound
References
Brilla, C. G., Janicki, J. S., & Weber, K. T. (1991). Impaired diastolic function and coronary reserve in genetic hypertension. Role of interstitial fibrosis and medial thickening of intramyocardial coronary arteries. Circulation Research, 69, 107–115. https://doi.org/10.1161/01.res.69.1.107
Ho, C. Y., López, B., Coelho-Filho, O. R., Lakdawala, N. K., Cirino, A. L., Jarolim, P., Kwong, R., González, A., Colan, S. D., Seidman, J. G., Díez, J., & Seidman, C. E. (2010). Myocardial fibrosis as an early manifestation of hypertrophic cardiomyopathy. New England Journal of Medicine, 363, 552–563. https://doi.org/10.1056/NEJMoa1002659
Sadoshima, J., & Izumo, S. (1997). The cellular and molecular response of cardiac myocytes to mechanical stress. Annual Review of Physiology, 59, 551–571. https://doi.org/10.1146/annurev.physiol.59.1.551
Kim, H. J., Park, M., Park, H. C., Jeong, J. C., Kim, D. K., Joo, K. W., Hwang, Y.-H., Yang, J., Ahn, C., & Oh, K.-H. (2016). Baseline Fgf23 is associated with cardiovascular outcome in incident Pd patients. Peritoneal Dialysis International, 36, 26–32. https://doi.org/10.3747/pdi.2013.00343
Saito, A., Onuki, T., Echida, Y., Otsubo, S., & Nitta, K. (2014). Fibroblast growth factor 23 and left ventricular hypertrophy in hemodialysis patients. International Journal of Clinical Medicine, 05, 1102–1110. https://doi.org/10.4236/ijcm.2014.517141
Silswal, N., Touchberry, C. D., Daniel, D. R., McCarthy, D. L., Zhang, S., Andresen, J., Stubbs, J. R., & Wacker, M. J. (2014). FGF23 directly impairs endothelium-dependent vasorelaxation by increasing superoxide levels and reducing nitric oxide bioavailability. American Journal of Physiology Endocrinology and Metabolism, 307, E426–E436. https://doi.org/10.1152/ajpendo.00264.2014
Faul, C., Amaral, A. P., Oskouei, B., Hu, M.-C., Sloan, A., Isakova, T., Gutiérrez, O. M., Aguillon-Prada, R., Lincoln, J., Hare, J. M., Mundel, P., Morales, A., Scialla, J., Fischer, M., Soliman, E. Z., Chen, J., Go, A. S., Rosas, S. E., Nessel, L., … Wolf, M. (2011). FGF23 induces left ventricular hypertrophy. The Journal of Clinical Investigation, 121, 4393–4408. https://doi.org/10.1172/JCI46122
Touchberry, C. D., Green, T. M., Tchikrizov, V., Mannix, J. E., Mao, T. F., Carney, B. W., Girgis, M., Vincent, R. J., Wetmore, L. A., Dawn, B., Bonewald, L. F., Stubbs, J. R., & Wacker, M. J. (2013). FGF23 is a novel regulator of intracellular calcium and cardiac contractility in addition to cardiac hypertrophy. American Journal of Physiology Endocrinology and Metabolism, 304, E863–E873. https://doi.org/10.1152/ajpendo.00596.2012
Datta, R., Bansal, T., Rana, S., Datta, K., Datta Chaudhuri, R., Chawla-Sarkar, M., & Sarkar, S. (2017). Myocyte-derived Hsp90 modulates collagen upregulation via biphasic activation of STAT-3 in fibroblasts during cardiac hypertrophy. Molecular and Cellular Biology, 37, e00611-e616. https://doi.org/10.1128/MCB.00611-16
Bendall, J. K., Heymes, C., Ratajczak, P., & Samuel, J. L. (2002). Extracellular matrix and cardiac remodelling. Archives des Maladies du Coeur et des Vaisseaux, 95, 1226–1229.
Carreño, J. E., Apablaza, F., Ocaranza, M. P., & Jalil, J. E. (2006). Cardiac hypertrophy: Molecular and cellular events. Rev Española Cardiol English Ed, 59, 473–486. https://doi.org/10.1016/S1885-5857(06)60796-2
Gerdes, A., & Capasso, J. (1995). Structural remodeling and mechanical dysfunction of cardiac myocytes in heart failure. Journal of Molecular and Cellular Cardiology, 27, 849–856. https://doi.org/10.1016/0022-2828(95)90000-4
Kuo, P.-L., Lee, H., Bray, M.-A., Geisse, N. A., Huang, Y.-T., Adams, W. J., Sheehy, S. P., & Parker, K. K. (2012). Myocyte shape regulates lateral registry of sarcomeres and contractility. American Journal of Pathology, 181, 2030–2037. https://doi.org/10.1016/j.ajpath.2012.08.045
Sawada, K., & Kawamura, K. (1991). Architecture of myocardial cells in human cardiac ventricles with concentric and eccentric hypertrophy as demonstrated by quantitative scanning electron microscopy. Heart and Vessels, 6, 129–142. https://doi.org/10.1007/BF02058278
Bernardo, B. C., Weeks, K. L., Pretorius, L., & McMullen, J. R. (2010). Molecular distinction between physiological and pathological cardiac hypertrophy: Experimental findings and therapeutic strategies. Pharmacology & Therapeutics, 128, 191–227. https://doi.org/10.1016/j.pharmthera.2010.04.005
Frey, N., Katus, H. A., Olson, E. N., & Hill, J. A. (2004). Hypertrophy of the heart: A new therapeutic target? Circulation, 109, 1580–1589. https://doi.org/10.1161/01.CIR.0000120390.68287.BB
Li, Y. Y., McTiernan, C. F., & Feldman, A. M. (2000). Interplay of matrix metalloproteinases, tissue inhibitors of metalloproteinases and their regulators in cardiac matrix remodeling. Cardiovascular Research, 46, 214–224. https://doi.org/10.1016/s0008-6363(00)00003-1
Parthasarathy, A., Gopi, V., Umadevi, S., Simna, A., Sheik, M. J. Y., Divya, H., & Vellaichamy, E. (2013). Suppression of atrial natriuretic peptide/natriuretic peptide receptor-A-mediated signaling upregulates angiotensin-II-induced collagen synthesis in adult cardiac fibroblasts. Molecular and Cellular Biochemistry, 378, 217–228. https://doi.org/10.1007/s11010-013-1612-z
Singh, M., Dalal, S., & Singh, K. (2014). Osteopontin: At the cross-roads of myocyte survival and myocardial function. Life Sciences, 118, 1–6. https://doi.org/10.1016/j.lfs.2014.09.014
Singh, M., Foster, C. R., Dalal, S., & Singh, K. (2010). Role of osteopontin in heart failure associated with aging. Heart Failure Reviews, 15, 487–494. https://doi.org/10.1007/s10741-010-9158-6
Dalal, S., Zha, Q., Singh, M., & Singh, K. (2016). Osteopontin-stimulated apoptosis in cardiac myocytes involves oxidative stress and mitochondrial death pathway: Role of a pro-apoptotic protein BIK. Molecular and Cellular Biochemistry, 418, 1–11. https://doi.org/10.1007/s11010-016-2725-y
Li, Y., Li, X., Guo, S., Chu, S., Gao, P., Zhu, D., Niu, W., & Jia, N. (2013). Apocynin attenuates oxidative stress and cardiac fibrosis in angiotensin II-induced cardiac diastolic dysfunction in mice. Acta Pharmacologica Sinica, 34, 352–359. https://doi.org/10.1038/aps.2012.164
Stawowy, P., Blaschke, F., Pfautsch, P., Goetze, S., Lippek, F., Wollert-Wulf, B., Fleck, E., & Graf, K. (2002). Increased myocardial expression of osteopontin in patients with advanced heart failure. European Journal of Heart Failure, 4, 139–146. https://doi.org/10.1016/s1388-9842(01)00237-9
Li, J., Yousefi, K., Ding, W., Singh, J., & Shehadeh, L. A. (2017). Osteopontin RNA aptamer can prevent and reverse pressure overload-induced heart failure. Cardiovascular Research, 113, 633–643. https://doi.org/10.1093/cvr/cvx016
Tsang, T.-J., Hsueh, Y.-C., Wei, E. I., Lundy, D. J., Cheng, B., Chen, Y.-T., Wang, S.-S., & Hsieh, P. C. H. (2017). Subcellular localization of survivin determines its function in cardiomyocytes. Theranostics, 7, 4577–4590. https://doi.org/10.7150/thno.20005
Bo, L., Zhu, X.-S., Zheng, Z., Hu, X.-P., & Chen, P.-Y. (2017). Research on the function and mechanism of survivin in heart failure mice model. European Review for Medical and Pharmacological Sciences, 21, 3699–3704. https://doi.org/10.26355/eurrev_201708_13287
Si, R., Tao, L., Zhang, H. F., Yu, Q. J., Zhang, R., Lv, A. L., Zhou, N., Cao, F., Guo, W. Y., Ren, J., Wang, H. C., & Gao, F. (2011). Survivin: A novel player in insulin cardioprotection against myocardial ischemia/reperfusion injury. Journal of Molecular and Cellular Cardiology, 50, 16–24. https://doi.org/10.1016/j.yjmcc.2010.08.017
Levkau, B., Schäfers, M., Wohlschlaeger, J., von Wnuck, L. K., Keul, P., Hermann, S., Kawaguchi, N., Kirchhof, P., Fabritz, L., Stypmann, J., Stegger, L., Flögel, U., Schrader, J., Fischer, J. W., Hsieh, P., Ou, Y.-L., Mehrhof, F., Tiemann, K., Ghanem, A., … Baba, H. A. (2008). Survivin determines cardiac function by controlling total cardiomyocyte number. Circulation, 117, 1583–1593. https://doi.org/10.1161/CIRCULATIONAHA.107.734160
Wohlschlaeger, J., Meier, B., Schmitz, K. J., Takeda, A., Takeda, N., Vahlhaus, C., Levkau, B., Stypmann, J., Schmid, C., Schmid, K. W., & Baba, H. A. (2010). Cardiomyocyte survivin protein expression is associated with cell size and DNA content in the failing human heart and is reversibly regulated after ventricular unloading. J Heart Lung Transplant, 29, 1286–1292. https://doi.org/10.1016/j.healun.2010.06.015
Wohlschlaeger, J., Schmitz, K. J., Schmid, C., Schmid, K. W., Keul, P., Takeda, A., Weis, S., Levkau, B., & Baba, H. A. (2005). Reverse remodeling following insertion of left ventricular assist devices (LVAD): A review of the morphological and molecular changes. Cardiovascular Research, 68, 376–386. https://doi.org/10.1016/j.cardiores.2005.06.030
Cinti, C., Taranta, M., Naldi, I., & Grimaldi, S. (2011). Newly engineered magnetic erythrocytes for sustained and targeted delivery of anti-cancer therapeutic compounds. PLoS ONE, 6, e17132. https://doi.org/10.1371/journal.pone.0017132
Grifantini, R., Taranta, M., Gherardini, L., Naldi, I., Parri, M., Grandi, A., Giannetti, A., Tombelli, S., Lucarini, G., Ricotti, L., Campagnoli, S., De Camilli, E., Pelosi, G., Baldini, F., Menciassi, A., Viale, G., Pileri, P., & Cinti, C. (2018). Magnetically driven drug delivery systems improving targeted immunotherapy for colon-rectal cancer. Journal of Controlled Release, 280, 76–86. https://doi.org/10.1016/j.jconrel.2018.04.052
Lande, C., Cecchettini, A., Tedeschi, L., Taranta, M., Naldi, I., Citti, L., Trivella, M. G., Grimaldi, S., & Cinti, C. (2012). Innovative erythrocyte-based carriers for gene delivery in porcine vascular smooth muscle cells: Basis for local therapy to prevent restenosis. Cardiovascular & Hematological Disorders: Drug Targets, 12, 68–75. https://doi.org/10.2174/187152912801823101
Lucarini, G., Sbaraglia, F., Vizzoca, A., Cinti, C., Ricotti, L., & Menciassi, A. (2020). Design of an innovative platform for the treatment of cerebral tumors by means of erythro-magneto-HA-virosomes. Biomed Phys Eng Express, 6, 45005. https://doi.org/10.1088/2057-1976/ab89f1
Naldi, I., Taranta, M., Gherardini, L., Pelosi, G., Viglione, F., Grimaldi, S., Pani, L., & Cinti, C. (2014). Novel epigenetic target therapy for prostate cancer: A preclinical study. PLoS ONE, 9, e98101. https://doi.org/10.1371/journal.pone.0098101
Carpi, S., Fogli, S., Giannetti, A., Adinolfi, B., Tombelli, S., Da Pozzo, E., Vanni, A., Martinotti, E., Martini, C., Breschi, M. C., Pellegrino, M., Nieri, P., & Baldini, F. (2014). Theranostic properties of a survivin-directed molecular beacon in human melanoma cells. PLoS ONE, 9, e114588. https://doi.org/10.1371/journal.pone.0114588
Nitin, N., Santangelo, P. J., Kim, G., Nie, S., & Bao, G. (2004). Peptide-linked molecular beacons for efficient delivery and rapid mRNA detection in living cells. Nucleic Acids Research, 32, e58. https://doi.org/10.1093/nar/gnh063
Langford, D. J., Bailey, A. L., Chanda, M. L., Clarke, S. E., Drummond, T. E., Echols, S., Glick, S., Ingrao, J., Klassen-Ross, T., Lacroix-Fralish, M. L., Matsumiya, L., Sorge, R. E., Sotocinal, S. G., Tabaka, J. M., Wong, D., van den Maagdenberg, A. M. J. M., Ferrari, M. D., Craig, K. D., & Mogil, J. S. (2010). Coding of facial expressions of pain in the laboratory mouse. Nature Methods, 7, 447–449. https://doi.org/10.1038/nmeth.1455
Cinti C, Lisi A, Grimaldi S (2010) Patent N. WO 2010/070620
Long, C. S., Ordahl, C. P., & Simpson, P. C. (1989). Alpha 1-adrenergic receptor stimulation of sarcomeric actin isogene transcription in hypertrophy of cultured rat heart muscle cells. The Journal of Clinical Investigation, 83, 1078–1082. https://doi.org/10.1172/JCI113951
Adachi, S., Ito, H., Tamamori, M., Tanaka, M., Marumo, F., & Hiroe, M. (1998). Skeletal and smooth muscle alpha-actin mRNA in endomyocardial biopsy samples of dilated cardiomyopathy patients. Life Sciences, 63, 1779–1791. https://doi.org/10.1016/s0024-3205(98)00452-4
Heineke, J., & Molkentin, J. D. (2006). Regulation of cardiac hypertrophy by intracellular signalling pathways. Nature Reviews Molecular Cell Biology, 7, 589–600. https://doi.org/10.1038/nrm1983
Hewett, T. E., Grupp, I. L., Grupp, G., & Robbins, J. (1994). Alpha-skeletal actin is associated with increased contractility in the mouse heart. Circulation Research, 74, 740–746. https://doi.org/10.1161/01.res.74.4.740
Liew, C.-C., & Dzau, V. J. (2004). Molecular genetics and genomics of heart failure. Nature Reviews Genetics, 5, 811–825. https://doi.org/10.1038/nrg1470
Zhao, Q. D., Viswanadhapalli, S., Williams, P., Shi, Q., Tan, C., Yi, X., Bhandari, B., & Abboud, H. E. (2015). NADPH oxidase 4 induces cardiac fibrosis and hypertrophy through activating Akt/mTOR and NFκB signaling pathways. Circulation, 131, 643–655. https://doi.org/10.1161/CIRCULATIONAHA.114.011079
Barger, P. M., & Kelly, D. P. (1999). Fatty acid utilization in the hypertrophied and failing heart: Molecular regulatory mechanisms. American Journal of the Medical Sciences, 318, 36–42. https://doi.org/10.1097/00000441-199907000-00006
Schulz, R. (2007). Intracellular targets of matrix metalloproteinase-2 in cardiac disease: Rationale and therapeutic approaches. Annual Review of Pharmacology and Toxicology, 47, 211–242. https://doi.org/10.1146/annurev.pharmtox.47.120505.105230
Adinolfi, B., Pellegrino, M., Giannetti, A., Tombelli, S., Trono, C., Sotgiu, G., Varchi, G., Ballestri, M., Posati, T., Carpi, S., Nieri, P., & Baldini, F. (2017). Molecular beacon-decorated polymethylmethacrylate core-shell fluorescent nanoparticles for the detection of survivin mRNA in human cancer cells. Biosensors & Bioelectronics, 88, 15–24. https://doi.org/10.1016/j.bios.2016.05.102
Bernardo, B. C., Weeks, K. L., Pongsukwechkul, T., Gao, X., Kiriazis, H., Cemerlang, N., Boey, E. J. H., Tham, Y. K., Johnson, C. J., Qian, H., Du, X.-J., Gregorevic, P., & McMullen, J. R. (2018). Gene delivery of medium chain acyl-coenzyme A dehydrogenase induces physiological cardiac hypertrophy and protects against pathological remodelling. Clinical Science (London, England), 132, 381–397. https://doi.org/10.1042/CS20171269
Patten, R. D., & Hall-Porter, M. R. (2009). Small animal models of heart failure: Development of novel therapies, past and present. Circulation. Heart Failure, 2, 138–144. https://doi.org/10.1161/CIRCHEARTFAILURE.108.839761
Faul, C. (2017). Cardiac actions of fibroblast growth factor 23. Bone, 100, 69–79. https://doi.org/10.1016/j.bone.2016.10.001
Hammond, H. K., Penny, W. F., Traverse, J. H., Henry, T. D., Watkins, M. W., Yancy, C. W., Sweis, R. N., Adler, E. D., Patel, A. N., Murray, D. R., Ross, R. S., Bhargava, V., Maisel, A., Barnard, D. D., Lai, N. C., Dalton, N. D., Lee, M. L., Narayan, S. M., Blanchard, D. G., & Gao, M. H. (2016). Intracoronary gene transfer of adenylyl cyclase 6 in patients with heart failure: A randomized clinical trial. JAMA Cardiol, 1, 163–171. https://doi.org/10.1001/jamacardio.2016.0008
Miyamoto, M. I., del Monte, F., Schmidt, U., DiSalvo, T. S., Kang, Z. B., Matsui, T., Guerrero, J. L., Gwathmey, J. K., Rosenzweig, A., & Hajjar, R. J. (2000). Adenoviral gene transfer of SERCA2a improves left-ventricular function in aortic-banded rats in transition to heart failure. Proc Natl Acad Sci U S A, 97, 793–798. https://doi.org/10.1073/pnas.97.2.793
Tevaearai, H. T., Walton, G. B., Keys, J. R., Koch, W. J., & Eckhart, A. D. (2005). Acute ischemic cardiac dysfunction is attenuated via gene transfer of a peptide inhibitor of the beta-adrenergic receptor kinase (betaARK1). The Journal of Gene Medicine, 7, 1172–1177. https://doi.org/10.1002/jgm.770
Kieserman, J. M., Myers, V. D., Dubey, P., Cheung, J. Y., & Feldman, A. M. (2019). Current landscape of heart failure gene therapy. Journal of the American Heart Association, 8, e012239. https://doi.org/10.1161/JAHA.119.012239
Acknowledgements
The authors thank Dr. Silvia Burchielli for supporting animal protocol preparation and internal ethical committee.
Funding
This work was supported by National Flagship project NANOMAX-ENCODER of the Italian Ministry of Education, University and Research.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
No human studies were carried out by the authors for this article. All institutional and national guidelines for the care and use of laboratory animals were followed and approved by the appropriate institutional committees.
Additional information
Communicated by Associate Editor Junjie Xiao oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kusmic, C., Vizzoca, A., Taranta, M. et al. Silencing Survivin: a Key Therapeutic Strategy for Cardiac Hypertrophy. J. of Cardiovasc. Trans. Res. 15, 391–407 (2022). https://doi.org/10.1007/s12265-021-10165-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12265-021-10165-1