Abstract
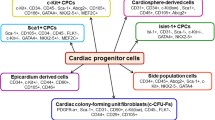
Experimental and clinical evidence suggests that a heterogeneous group of bone-marrow-derived circulating progenitor cells, with variations in phenotype and function, provide an endogenous repair mechanism, contributing to vascular healing and remodeling under physiological and pathological conditions, such as cancer, atherosclerosis, myocardial infarction, and end-stage heart failure. Implantation of ventricular assist devices (VADs) for circulatory support is indicated in selected patients with end-stage heart failure as a bridge to heart transplantation, however seldom; improvement of ventricular contractility has been well documented with prolonged cardiac unloading. The current review summarizes recent findings from in vitro and in vivo studies, focusing on the biological features and the possible role of progenitor cells in the transient myocardial recovery, occasionally seen after VAD implantation, and speculates on their clinical utilities for the treatment of the failing human heart.

Similar content being viewed by others
References
Aicher, A., Heeschen, C., Mildner-Rihm, C., Urbich, C., Ihling, C., Technau-Ihling, K., et al. (2003). Essential role of endothelial nitric oxide synthase for mobilization of stem and progenitor cells. Nature Medicine, 9, 1370–1376.
Asahara, T., Murohara, T., Sullivan, A., Silver, M., van der Zee, R., Li, T., et al. (1997). Isolation of putative progenitor endothelial cells for angiogenesis. Science, 275, 964–967.
Asahara, T., Takahashi, T., Masuda, H., Kalka, C., Chen, D., Iwaguro, H., et al. (1999). VEGF contributes to postnatal neovascularization by mobilizing bone marrow-derived endothelial progenitor cells. EMBO Journal, 18, 3964–3972.
Assmus, B., Schächinger, V., Teupe, C., Britten, M., Lehmann, R., Döbert, N., et al. (2002). Transplantation of progenitor cells and regeneration enhancement in acute myocardial infarction (TOPCARE-AMI). Circulation, 106, 3009–3017.
Bahlmann, F. H., de Groot, K., Mueller, O., Hertel, B., Haller, H., & Fliser, D. (2005). Stimulation of endothelial progenitor cells: A new putative therapeutic effect of angiotensin II receptor antagonists. Hypertension, 45, 526–529.
Behfar, A., Zingman, L. V., Hodgson, D. M., Rauzier, J. M., Kane, G. C., Terzic, A., et al. (2002). Stem cell differentiation requires a paracrine pathway in the heart. FASEB Journal, 16, 1558–1566.
Dimmeler, S., Aicher, A., Vasa, M., Mildner-Rihm, C., Adler, K., Tiemann, M., et al. (2001). HMG-CoA reductase inhibitors (statins) increase endothelial progenitor cells via the PI 3-kinase/Akt pathway. Journal of Clinical Investigation, 108, 391–397.
Fadini, G. P., Coracina, A., Baesso, I., Agostini, C., Tiengo, A., Avogaro, A., et al. (2006). Peripheral blood CD34+ KDR+ endothelial progenitor cells are determinants of subclinical atherosclerosis in a middle-aged general population. Stroke, 37, 2277–2282.
Fadini, G. P., de Kreutzenberg, S. V., Coracina, A., Baesso, I., Agostini, C., Tiengo, A., et al. (2006). Circulating CD34+ cells, metabolic syndrome, and cardiovascular risk. European Heart Journal, 27, 2247–2255.
Foresta, C., Lana, A., Cabrelle, A., Ferigo, M., Caretta, N., Garolla, A., et al. (2005). PDE-5 inhibitor, vardenafil, increases circulating progenitor cells in humans. International Journal of Impotence Research, 17, 377–380.
Friedrich, E. B., Walenta, K., Scharlau, J., Nickenig, G., & Werner, N. (2006). CD34−/CD133+/VEGFR-2+ endothelial progenitor cell subpopulation with potent vasoregenerative capacities. Circulation Research, 98, e20–e25.
George, J., Goldstein, E., Abashidze, A., Wexler, D., Hamed, S., Shmilovich, H., et al. (2005). Erythropoietin promotes endothelial progenitor cell proliferative and adhesive properties in a PI 3-kinase-dependent manner. Cardiovascular Research, 68, 299–306.
Gill, M., Dias, S., Hattori, K., Rivera, M. L., Hicklin, D., Witte, L., et al. (2001). Vascular trauma induces rapid but transient mobilization of VEGFR2(+)AC133(+) endothelial precursor cells. Circulation Research, 88, 167–164.
Gnecchi, M., Zhang, Z., Ni, A., & Dzau, V. J. (2008). Paracrine mechanisms in adult stem cell signaling and therapy. Circulation Research, 103, 1204–1219.
Goette, A., Jentsch-Ullrich, K., Lendeckel, U., Röcken, C., Agbaria, M., Auricchio, A., et al. (2003). Effect of atrial fibrillation on hematopoietic progenitor cells: A novel pathophysiological role of the atrial natriuretic peptide? Circulation, 108, 2446–2449.
Heissig, B., Hattori, K., Dias, S., Friedrich, M., Ferris, B., Hackett, N. R., et al. (2002). Recruitment of stem and progenitor cells from the bone marrow niche requires MMP-9 mediated release of kit-ligand. Cell, 109, 625–637.
Hill, J. M., Zalos, G., Halcox, J. P., Schenke, W. H., Waclawiw, M. A., Quyyumi, A. A., et al. (2003). Circulating endothelial progenitor cells, vascular function, and cardiovascular risk. New England Journal of Medicine, 348, 593–600.
Hristov, M., Erl, W., & Weber, P. (2003). Endothelial progenitor cells. Mobilization, differentiation, and homing. Arteriosclerosis, Thrombosis, and Vascular Biology, 23, 1185–1189.
Ingram, D. A., Caplice, N. M., & Yoder, M. C. (2005). Unresolved questions, changing definitions, and novel paradigms for defining endothelial progenitor cells. Blood, 106, 1525–1531.
Iversen, P. O., Woldbaek, P. R., Tønnessen, T., & Christensen, G. (2002). Decreased hematopoiesis in bone marrow of mice with CHF. American Journal of Physiology: Regulatory, Integrative and Comparative Physiology, 282, R166–R172.
Jahanyar, J., Youker, K. A., Torre-Amione, G., Koerner, M. M., Bruckner, B., Noon, G. P., et al. (2008). Increased expression of stem cell factor and its receptor after left ventricular assist device support: A potential novel target for therapeutic interventions in heart failure. Journal of Heart and Lung Transplantation, 27, 701–709.
Kirshenbaum, A. S., Goff, J. P., Semere, T., Foster, B., Scott, L. M., & Metcalfe, D. D. (1999). Demonstration that human mast cells arise from a progenitor cell population that is CD34(+), c-kit(+), and expresses aminopeptidase N (CD13). Blood, 94, 2333–2342.
Kirshenbaum, A. S., Kessler, S. W., Goff, J. P., & Metcalfe, D. D. (1991). Demonstration of the origin of human mast cells from CD34+ bone marrow progenitor cells. Journal of Immunology, 146, 1410–1415.
Kissel, C. K., Lehmann, R., Assmus, B., Aicher, A., Honold, J., Fischer-Rasokat, U., et al. (2007). Selective functional exhaustion of hematopoietic progenitor cells in the bone marrow of patients with postinfarction heart failure. Journal of the American College of Cardiology, 49, 2341–2349.
Laufs, U., Werner, N., Link, A., Endres, M., Wassmann, S., Jürgens, K., et al. (2004). Physical training increases endothelial progenitor cells, inhibits neointima formation, and enhances angiogenesis. Circulation, 109, 220–226.
Leone, A. M., Rutella, S., Bonanno, G., Abbate, A., Rebuzzi, A. G., Giovannini, S., et al. (2005). Mobilization of bone marrow-derived stem cells after myocardial infarction and left ventricular function. European Heart Journal, 26, 1196–1204.
Leone, A. M., Rutella, S., Bonanno, G., Contemi, A. M., de Ritis, D. G., Giannico, M. B., et al. (2006). Endogenous G-CSF and CD34+ cell mobilization after acute myocardial infarction. International Journal of Cardiology, 111, 202–208.
Manginas, A., Goussetis, E., Koutelou, M., Karatasakis, G., Peristeri, I., Theodorakos, A., et al. (2007). Pilot study to evaluate the safety and feasibility of intracoronary CD133(+) and CD133(−) CD34(+) cell therapy in patients with nonviable anterior myocardial infarction. Catheterization and Cardiovascular Interventions, 69, 773–781.
Manginas, A., Tsiavou, A., Sfyrakis, P., Giamouzis, G., Tsourelis, L., Leontiadis, E., et al. (2009). Increased number of circulating progenitor cells after implantation of ventricular assist devices. Journal of Heart and Lung Transplantation, 28, 710–717.
Maybaum, S., Mancini, D., Xydas, S., Starling, R. C., Aaronson, K., Pagani, F. D., et al. (2007). Cardiac improvement during mechanical circulatory support: A prospective multicenter study of the LVAD Working Group. Circulation, 115, 2497–2505.
Murohara, T., Ikeda, H., Duan, J., Shintani, S., Sasaki, K., Eguchi, H., et al. (2000). Transplanted cord blood-derived endothelial precursor cells augment postnatal neovascularization. Journal of Clinical Investigation, 105, 1527–1536.
Nissen, N. N., Polverini, P. J., Koch, A. E., Volin, M. V., Gamelli, R. L., & DiPietro, L. A. (1998). Vascular endothelial growth factor mediates angiogenic activity during the proliferative phase of wound healing. American Journal of Pathology, 152, 1445–1452.
Nonaka-Sarukawa, M., Yamamoto, K., Aoki, H., Nishimura, Y., Tomizawa, H., Ichida, M., et al. (2007). Circulating endothelial progenitor cells in CHF. International Journal of Cardiology, 119, 344–348.
Olthof, S. G., Fatrai, S., Drayer, A. L., Tyl, M. R., Vellenga, E., & Schuringa, J. J. (2008). Downregulation of signal transducer and activator of transcription 5 (STAT5) in CD34+ cells promotes megakaryocytic development, whereas activation of STAT5 drives erythropoiesis. Stem Cells, 26, 1732–1742.
Orlandi, A., Pagani, F., Avitabile, D., Bonanno, G., Scambia, G., Vigna, E., et al. (2008). Functional properties of cells obtained from human cord blood CD34+ stem cells and mouse cardiac myocytes in coculture. American Journal of Physiology. Heart and Circulatory Physiology, 294, 1541–1549.
Papayannopoulou, T. (2003). Mechanisms of stem-/progenitor-cell mobilization: The anti-VLA-4 paradigm. Seminars in Hematology, 37(1 Suppl 2), 11–18.
Peichev, M., Naiyer, A. J., Pereira, D., Zhu, Z., Lane, W. J., Williams, M., et al. (2000). Expression of VEGFR-2 and AC133 by circulating human CD34(+) cells identifies a population of functional endothelial precursors. Blood, 95, 952–958.
Perin, E. C., Dohmann, H. F., Borojevic, R., Silva, S. A., Sousa, A. L., Mesquita, C. T., et al. (2003). Transendocardial, autologous bone marrow cell transplantation for severe, chronic ischemic heart failure. Circulation, 107, 2294–2302.
Pistrosch, F., Herbrig, K., Oelschlaegel, U., Richter, S., Passauer, J., Fischer, S., et al. (2005). PPARgamma-agonist rosiglitazone increases number and migratory activity of cultured endothelial progenitor cells. Atherosclerosis, 183, 163–167.
Popa, E. R., Harmsen, M. C., Tio, R. A., van der Strate, B. W., Brouwer, L. A., Schipper, M., et al. (2006). Circulating CD34+ progenitor cells modulate host angiogenesis and inflammation in vivo. Journal of Molecular and Cellular Cardiology, 41, 86–96.
Powell, T. M., Paul, J. D., Hill, J. M., Thompson, M., Benjamin, M., Rodrigo, M., et al. (2005). Granulocyte colony-stimulating factor mobilizes functional endothelial progenitor cells in patients with coronary artery disease. Arteriosclerosis, Thrombosis, and Vascular Biology, 25, 296–301.
Rafii, S., & Lyden, D. (2003). Therapeutic stem and progenitor cell transplantation for organ vascularization and regeneration. Nature Medicine, 9, 702–712.
Rafii, S., Oz, M. C., Seldomridge, J. A., Ferris, B., Asch, A. S., Nachman, R. L., et al. (1995). Characterization of hematopoietic cells arising on the textured surface of left ventricular assist devices. Annals of Thoracic Surgery, 60, 1627–1632.
Rose, E. A., Gelijns, A. C., Moskowitz, A. J., Heitjan, D. F., Stevenson, L. W., Dembitsky, W., et al. (2001). Long-term mechanical left ventricular assistance for end-stage heart failure. New England Journal of Medicine, 345, 1435–1443.
Scheubel, R. J., Zorn, H., Silber, R. E., Kuss, O., Morawietz, H., Holtz, J., et al. (2003). Age-dependent depression in circulating endothelial progenitor cells in patients undergoing coronary artery bypass grafting. Journal of the American College of Cardiology, 42, 2073–2080.
Schmidt-Lucke, C., Rössig, L., Fichtlscherer, S., Vasa, M., Britten, M., Kämper, U., et al. (2005). Reduced number of circulating endothelial progenitor cells predicts future cardiovascular events: Proof of concept for the clinical importance of endogenous vascular repair. Circulation, 111, 2981–2987.
Shi, Q., Rafii, S., Wu, M. H., Wijelath, E. S., Yu, C., Ishida, A., et al. (1998). Evidence for circulating bone marrow-derived endothelial cells. Blood, 92, 362–367.
Shintani, S., Murohama, T., Ikeda, H., Ueno, T., Honma, T., Katoh, A., et al. (2001). Mobilization of endothelial progenitor cells in patients with acute myocardial infarction. Circulation, 103, 2776–2779.
Soppa, G. K., Barton, P. J., Terracciano, C. M., & Yacoub, M. H. (2008). Left ventricular assist device-induced molecular changes in the failing myocardium. Current Opinion in Cardiology, 23, 206–218.
Stamm, C., Westphal, B., Kleine, H. D., Petzsch, M., Kittner, C., Klinge, H., et al. (2003). Autologous bone-marrow stem-cell transplantation for myocardial regeneration. Lancet, 361, 45–46.
Strauer, B. E., Brehm, M., Zeus, T., Köstering, M., Hernandez, A., Sorg, R. V., et al. (2002). Repair of infarcted myocardium by autologous intracoronary mononuclear bone marrow cell transplantation in humans. Circulation, 106, 1913–1918.
Takahashi, T., Kalka, C., Masuda, H., Chen, D., Silver, M., Kearney, M., et al. (1999). Ischemia- and cytokine-induced mobilization of bone marrow-derived endothelial progenitor cells for neovascularization. Nature Medicine, 5, 434–438.
Tse, H. F., Kwong, Y. L., Chan, J. K., Lo, G., Ho, C. L., & Lau, C. P. (2003). Angiogenesis in ischaemic myocardium by intramyocardial autologous bone marrow mononuclear cell implantation. Lancet, 361, 47–49.
Urbich, C., Aicher, A., Heeschen, C., Dernbach, E., Hofmann, W. K., Zeiher, A. M., et al. (2005). Soluble factors released by endothelial progenitor cells promote migration of endothelial cells and cardiac resident progenitor cells. Journal of Molecular and Cellular Cardiology, 39, 733–742.
Urbich, C., & Dimmeler, S. (2004). Endothelial progenitor cells: Characterization and role in vascular biology. Circulation Research, 95, 343–353.
Valgimigli, M., Rigolin, G. M., Fucili, A., Porta, M. D., Soukhomovskaia, O., Malagutti, P., et al. (2004). CD34+ and endothelial progenitor cells in patients with various degrees of CHF. Circulation, 110, 1209–1212.
Vasa, M., Fichtlscherer, S., Aicher, A., Adler, K., Urbich, C., Martin, H., et al. (2001). Number and migratory activity of circulating endothelial progenitor cells inversely correlate with risk factors for coronary artery disease. Circulation Research, 89, e1–e7.
Vranken, I., De Visscher, G., Lebacq, A., Verbeken, E., & Flameng, W. (2008). The recruitment of primitive Lin(−) Sca-1(+), CD34(+), c-kit(+) and CD271(+) cells during the early intraperitoneal foreign body reaction. Biomaterials, 29, 797–808.
Wojciech, W., RataJczak, M., & Tendera, M. (2006). Intereukin-8: More on the mechanisms of progenitor cells mobilization in acute coronary syndromes. European Heart Journal, 27, 1013–1015.
Wollert, K. C., Meyer, G. P., Lotz, J., Ringes-Lichtenberg, S., Lippolt, P., Breidenbach, C., et al. (2004). Intracoronary autologous bone-marrow cell transfer after myocardial infarction: the BOOST randomised controlled clinical trial. Lancet, 364, 141–148.
Yamaguchi, J., Kusano, K. F., Masuo, O., Kawamoto, A., Silver, M., Murasawa, S., et al. (2003). Stromal cell-derived factor-1 effects on ex vivo expanded endothelial progenitor cell recruitment for ischemic neovascularization. Circulation, 107, 1322–1328.
Yeh, E. T., Zhang, S., Wu, H. D., Körbling, M., Willerson, J. T., & Estrov, Z. (2003). Transdifferentiation of human peripheral blood CD34+-enriched cell population into cardiomyocytes, endothelial cells, and smooth muscle cells in vivo. Circulation, 108, 2070–2073.
Yoshioka, T., Ageyama, N., Shibata, H., Yasu, T., Misawa, Y., Takeuchi, K., et al. (2005). Repair of infarcted myocardium mediated by transplanted bone marrow-derived CD34+ stem cells in a nonhuman primate model. Stem Cells, 23, 355–364.
Zhang, S., Wang, D., Estrov, Z., Raj, S., Willerson, J. T., & Yeh, E. T. (2004). Both cell fusion and transdifferentiation account for the transformation of human peripheral blood CD34-positive cells into cardiomyocytes in vivo. Circulation, 110, 3803–3807.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tsiavou, A., Manginas, A. Dynamics of Progenitor Cells and Ventricular Assist Device Intervention. J. of Cardiovasc. Trans. Res. 3, 147–152 (2010). https://doi.org/10.1007/s12265-009-9141-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12265-009-9141-6