Abstract
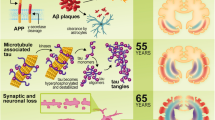
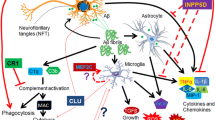
Inflammation has been shown to play an important role in the progression of Alzheimer’s disease (AD). Recent epidemical study indicates that the incidence of AD in some populations is substantially influenced by the gene polymorphisms of the inflammation mediators. Meanwhile, an ensured risk factor, the ApoE ɛ4 allele is also reported to directly promote inflammation. Accordingly, it appears that an individual genetic background has partly determined his predisposition for AD by the extent of the inflammation response to the chronic stimulus by β-amyloid peptide (Aβ) deposits and other antigen stressor in the elderly. Hence we present a hypothesis that the inflammation genotypes may contribute to AD susceptibility. This may provide a new orientation both for future identification of individuals at risk and for personalized medication.
摘要
炎症反应在阿尔茨海默病的进展中发挥着重要的作用。 近来流行病学研究显示炎症介质的基因多态性可以影响阿尔茨海默病在人群中的发病率。 另有报道, ApoE ɛ4, 阿尔茨海默病的危险因子, 可直接增强炎症反应。 因此, 在老龄化过程中, 个体是否容易罹患阿尔茨海默病, 可能与对Aβ斑块和其他免疫原的长期刺激引发的炎症反应强度有关, 其反应强度取决于其遗传基因。 炎症相关的基因可能影响个体对阿尔茨海默病的易感性, 并可能为将来的阿尔茨海默病危险人群的筛选和个体化治疗提供新的研究方向。
Similar content being viewed by others
References
Wang XP, Ding HL. Alzheimer’s disease: epidemiology, genetics, and beyond. Neurosci Bull 2008, 24: 105–109.
Streit WJ. Micorglia and Alzheimer’s disease pathogenesis. J Neurosci Res 2004, 77: 1–8.
Hoozemans JJ, O’Banion MK. The role of COX-1 and COX-2 in Alzheimer’s disease pathology and the therapeutic potentials of non-steroidal anti-inflammatory drugs. Curr Drug Targets CNS Neurol Disord 2005, 4: 307–315.
Davis S, Laroche S. What can rodent models tell us about cognitive decline in Alzheimer’s disease? Mol Neurobiol 2003, 27: 249–276.
Tuppo EE, Arias HR. The role of inflammation in Alzheimer’s disease. J Biochem Cell Biol 2005, 37: 289–305.
Finch CE, Morgan TE. Systemic inflammation, infection, ApoE alleles, and Alzheimer disease: a position paper. Curr Alzheimer Res 2007, 4: 185–189.
Lindberg C, Hjorth E, Post C, Winblad B, Schultzberg M. Cytokine production by a human microglia cell line: effects of beta-amyloid and alpha-melanocyte-stimulating hormone. J Neurotox Res 2005, 8: 367–376.
Nee LE, Lippa CF. Alzheimer’s disease in 22 twin pairs-13-year follow-up: hormonal, infectious and traumatic factors. Dement J Geriatr Cogn Disord 1999, 10: 148–151.
Lu KT, Wang YW, Yang JT, Yang YL, Chen HI. Effect of interleukin-1 on traumatic brain injury-induced damage to hippocampal neurons. J Neurotrauma 2005, 22: 885–895.
Tong L, Balazs R, Soiampornkul R, Thangnipon W, Cotman CW. Interleukin-1β impairs brain derived neurotrophic factor-induced signal transduction. Neurobiol Aging 2008, 29: 1380–1393.
Ma G, Chen S, Wang X, Ba M, Yang H, Lu G. Short-term interleukin-1β increases the release of secreted APP(alpha) via MEK1/2-dependent and JNK-dependent alpha-secretase cleavage in neuroglioma U251 cells. J Neurosci Res 2005, 80: 683–692.
Schlirbs R, Heidel K, Apelt J, Gniezdzinska M, Kirazov L, Szutowicz A. Interaction of interleukin-1β with muscarinic acetylchline receptor-mediated signaling cascade in cholinergically differentiated SH-SY5Y cells. Brain Res 2006, 1122: 78–85.
Hampel H, Haslinger A, Scheloske M, Padberg F, Fischer P, Unger J, et al. Pattern of interleukin-6 receptor complex immunoreactivity between cortical regions of rapid autopsy normal and Alzheimer’s disease brain. Eur Arch Psychiatry Clin Neurosci 2005, 255: 269–278.
Quintanilla RA, Orellana DI, González-Billault C, Maccini RB. Interleukin-6 induces Alzheimer-type phosphorylation of tau protein by deregulating the cdk5/p35 pathway. Exp Cell Res 2004, 295: 245–257.
Braida D, Sacerdote P, Panerai AE, Bianchi M, Aloisi AM, Iosuè S, et al. Cognitive function in young and adult IL-6 deficient mice. Behav Brain Res 2004, 153: 423–429.
Qiu Z, Gruol DL. Interleukin-6, β-amyloid peptide and NMDA interactions in rat cortical neurons. J Neuroimmunol 2003, 139: 51–57.
Alvarez A, Cacabelos R, Sanpedro C, Garciìa-Fantini M, Aleixandre M. Serum TNF-alpha levels are increased and correlate negatively with free IGF-I in Alzheimer disease. Neurobiol Aging 2007, 28: 533–536.
Takeuchi H, Jin S, Wang J, Zhang G, Kawanokuchi J, Kuno R, et al. Tumor necrosis factor-α induces neurotoxicity via glutamate release from hemichannels of activated microglia in an autocrine manner. J Biol Chem 2006, 281: 21362–21368.
Tweedie D, Sambamurti K, Greig NH. TNF-α inhibition as a treatment strategy for neurodegenerative disorders: new drug candidates and targets. Curr Alzheimer Res 2007, 4: 378–385.
Burton T, Liang B, Dibrov A, Amara F. Transcriptional activation and increase in expression of Alzheimer’s β-amyloid precursor protein gene is mediated by TGF-β in normal human astrocytes. Biochem Biophys Res Commun 2002, 295: 702–712.
Aigner L, Winkler J, Bogdahn U. Protection or reconstruction: Neuroprotective effects on transforming growth factor-β1 at the cost of reduced neurogenesis. J Neuroforum 2007, 13: 4–12.
Salins P, He Y, Olson K, Glazner G, Kashour T, Amara F. TGF-β1 is increased in a transgenic mouse model of familial Alzheimer’s disease and causes neuronal apoptosis. Neurosci Lett 2008, 430: 81–86.
Tesseur I, Zou K, Esposito L, Bard F, Berber E, Can JV, et al. Deficiency in neuronal TGF-β signaling promotes neurodegeneration and Alzheimer’s pathology. J Clin Invest 2006, 116: 3060–3069.
Motta M, Imbesi R, Di Rosa M, Stivala F, Malaguarnera L. Altered plasma cytokine levels in Alzheimer’s disease: correlation with the disease progression. Immunol Lett 2007, 114: 46–51.
Walter S, Letiembre M, Liu Y, Heine H, Penke B, Hao W, et al. Role of the toll-Like receptor 4 in neuroinflammation in Alzheimer’s disease. Cell Physiol Biochem 2007, 20: 974–956.
Minoretti P, Gazzaruso C, Vito CD, Emanuele E, Bianchi M, Coen E, et al. Effect of the functional toll-like receptor 4 Asp299Gly polymorphism on susceptibility to late-onset Alzheimer’s disease. Neurosci Lett 2006, 391: 147–149.
Wang WF, Liao YC, Wu SL, Tsai FJ, Lee CC, Hua CS. Association of interleukin-I beta and receptor antagonist gene polymorphisms with late onset Alzheimer’s disease in Taiwan Chinese. Eur J Neruol 2005, 12: 609–613.
Zhou YT, Zhang ZX, Zhang JW, He XM, Xu T. Association between interleukin-1 α-889 C/T polymorphism and Alzheimer’s disease in Chinese Han population. J Acta Academiae Medicinae Sinicae 2006, 28: 186–190. (Chinese, English abstract)
Licastro F, Grimaldi LM, Bonafè M, Martina C, Olivieri F, Cavallone L, et al. Interleukin-6 gene alleles affect the risk of Alzheimer’s disease and levels of the cytokine in blood and brain. Neurobiol Aging 2003, 24: 921–926.
Lio D, Annoni G, Licastro F, Crivello A, Forte GI, Colonna-Romano G, et al. Tumor necrosis factor-α-308A/G polymorphism is associated with age at onset of Alzheimer’s disease. Mech Ageing Dev 2006, 127: 567–571.
Ramos EM, Lin MT, Larson EB, Maezawa I, Tseng LH, Edwards KL, et al. Tumor necrosis factor α and interleukin 10 promoter region polymorphisms and risk of late-onset Alzheimer disease. Arch Neurol 2006, 63: 1165–1169.
Arosio B, Bergamaschini L, Galimberti L, La Porta C, Zanetti M, Calabresi C, et al. +10 T/C polymorphisms in the gene of transforming growth factor-β1 are associated with neurodegeneration and its clinical evolution. Mech Ageing Dev 2007, 128: 553–557.
O’Hara R, Sommer B, Way N, Kraemer HC, Taylor J, Murphy G. Slower speed-of-processing of cognitive tasks is associated with presence of the apolipoprotein ɛ4 allele. J Psychiatric Res 2007, 42: 199–204.
Espeseth T, Westlye LT, Fjell AM, Walhovd KB, Rootwelt H, Reinvang I. Accelerated age-related cortical thinning in healthy carriers of apolipoprotein E ɛ4. Neurol Aging 2008, 29: 329–340.
Laskowitz D, Lee DM, Schmechel D, Staats HF. Altered immune responses in apolipoprotein E-deficient mice. J Lipid Res 2000, 41: 613–620.
Jofre-Monseny L, de pascual-Teresa S, Plonka E, Huebbe P, Boesch-Saadatmandi C, Miniane AM, et al. Differential effects of apolipoprotein E3 and E4 on markers of oxidative status in macrophages. Br J Nutr 2007, 97: 864–871.
Colton CA, Needham LK, Brown C, Cook D, Rasheed K, Burke JR, et al. APOE genotype-specific differences in human and mouse macrophage nitric oxide production. J Neuroimmunol 2004, 147:62–67.
Vitek MP, Brown CM, Colton CA. APOE genotype-specific differences in the innate immune response. Neurobiol Aging 2007, Epub ahead of print. (doi:10.1016/j.neurobiolaging.2007.11.014)
Maezawa I, Nivison M, Montine KS, Maeda N, Montine TJ. Neurotoxicity from innate immune response is greatest with targeted replacement of E4 allele of apolipoprotein E gene and is mediated by microglial p38MAPK. FASEB J 2006, 20: 797–799.
Ophir G, Amariglio N, Jacob-Hirsch J, Elkon R, Rechavi G, Michaelson DM. Apolipoprotein E4 enhances brain inflammation by modulation of the NF-kappaB signaling cascade. Neurobiol Dis 2005, 20: 709–718.
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contributed equally to this work.
Rights and permissions
About this article
Cite this article
Wan, Y., Wang, G. & Chen, SD. Genetic predisposition to inflammation: a new risk factor of Alzheimer’s disease. Neurosci. Bull. 24, 314–322 (2008). https://doi.org/10.1007/s12264-008-0619-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12264-008-0619-z
Keywords
- Alzheimer’s disease
- inflammation
- predisposition
- polymorphism
- inflammation mediators
- ApoE ɛ4 allele
- identification
- personalized medication