Abstract
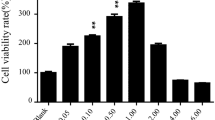
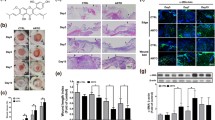
Enhanced cell migration in the course of wound healing is required to repair damaged skin. We investigated the effects of rice callus extracts on the migration of skin keratinocytes. Rice callus extracts were obtained by using three different methods: pressurized hot water, crude ethanol, and liquid-liquid extractions. The extract obtained by using crude ethanol extraction was more effective in the migration of skin cells than that obtained by pressurized hot water extraction (PHWE). The crude ethanol extract (CEE) was further partitioned by performing liquid-liquid extraction. As phenolics are inhibitory compounds affecting cell migration, we analyzed the total phenolic content of the rice callus extracts. The level of phenolics in the n-hexane partitioned extract (n-HPE) of CEE was lower than that in all other partitioned extracts. The n-HPE was most effective in enhancing cell migration. We analyzed wound healingrelated factors including platelet derived growth factor B (PDGF-B), transforming growth factor beta 1 (TGF-β1), heparin-binding epidermal growth factor (HB-EGF), and fibroblast growth factor 2 (FGF-2) after the treatment of n-HPE. Most of the expressions of cell migration-related growth factors increased, but HB-EGF dramatically increased (6.5-fold) in keratinocytes treated with n-HPE. The results indicate that n-HPE contains more stimulating growth factors or proteins and less cell migration inhibiting factors than the other tested extracts; thus, n-HPE treatment produced the greatest enhancement of cell migration.
Similar content being viewed by others
References
Proksch, E., J. M. Brandner, and J. M. Jensen (2008) The skin: An indispensable barrier. Exp. Dermatol. 17: 1063–1072.
Singer, A. J. (1999) Cutaneous wound healing. N. Engl. J. Med. 341: 738–746.
Brusotti, G., F. Andreola, G. Sferrazza, P. Grisoli, A. Merelli, F. S. R. Cuna, E. Calleri, G. Nicotera, P. Pierimarchi, and A. Serafino (2015) In vitro evaluation of the wound healing activity of Drypetes klainei stem bark extracts. J. Ethnopharmacol. 175: 412–421.
Eruygur, N., G. Yilmaz, O. Kutsal, G. Yücel, and O. Üstün (2016) Bioassay-guided isolation of wound healing active compounds from Echium species growing in Turkey. J. Ethnopharmacol. 185: 370–376.
Pinto, N., P. Cassini-Vieira, E. Souza-Fagundes, L. S. Barcelos, M. C. Castañon Marques Nogueira, and E. Scio (2016) Pereskia aculeata Miller leaves accelerate excisional wound healing in mice. J. Ethnopharmacol. 194: 131–136.
Hiei, Y. and T. Komari (2008) Agrobacterium-mediated transformation of rice using immature embryos or calli induced from mature seed. Nat. Protoc. 3: 824–834.
Coccia, A., D. Bastianelli, L. Mosca, R. Monticolo, I. Panuccio, A. Carbone, A. Calogero, and E. Lendaro (2014) Extra virgin olive oil phenols suppress migration and invasion of T24 human bladder cancer cells through modulation of matrix Metalloproteinase-2. Nutr. Cancer 66: 946–954.
Ham, S. L., S. Nasrollahi, K. N. Shah, A. Soltisz, S. Paruchuri, Y. H. Yun, G. D. Luker, A. Bishayee, and H. Tavana (2015) Phytochemicals potently inhibit migration of metastatic breast cancer cells. Integr. Biol. 7: 792–800.
Sang, J. L., W. L. Ki, J. H. Haeng, Y. C. Ji, Y. K. Seo, and J. L. Hyong (2007) Phenolic phytochemicals derived from red pine (Pinus densiflora) inhibit the invasion and migration of SK-Hep-1 human hepatocellular carcinoma cells. Ann. N. Y. Acad. Sci. 1095: 536–544.
Gurtner, G. C., S. Werner, Y. Barrando, and M. T. Longaker (2008) Wound repair and regeneration. Nature 453: 314–321.
Dai, J. and R. J. Mumper (2010) Plant phenolics: Extraction, analysis and their antioxidant and anticancer properties. Molecules 15: 7313–7352.
Huang, C., K. Jacobson, and M. D. Schaller (2004) MAP kinases and cell migration. J. Cell. Sci. 117: 4619–4628.
Klemke, R. L., S. Cai, A. L. Giannini, P. J. Gallagher, P. De Lanerolle, and D. A. Cheresh (1997) Regulation of cell motility by mitogen-activated protein kinase. J. Cell Biol. 137: 481–492.
Sharova, L. V., A. A. Sharov, T. Nedorezov, Y. Piao, N. Shaik, and M. S. H. Ko (2009) Database for mRNA half-life of 19 977 genes obtained by DNA microarray analysis of pluripotent and differentiating mouse embryonic stem cells. DNA Res. 16: 45–58.
Barrientos, S., O. Stojadinovic, M. S. Golinko, H. Brem, and M. Tomic-Canic (2008) Growth factors and cytokines in wound healing. Wound Repair Regen. 16: 585–601.
Shirakata, Y., R. Kimura, D. Nanba, R. Iwamoto, S. Tokumaru, C. Morimoto, K. Yokota, M. Nakamura, K. Sayama, E. Mekada, S. Higashiyama, and K. Hashimoto (2005) Heparin-binding EGF-like growth factor accelerates keratinocyte migration and skin wound healing. J. Cell. Sci. 118: 2363–2370.
Tokumaru, S., S. Higashiyama, T. Endo, T. Nakagawa, J. I. Miyagawa, K. Yamamori, Y. Hanakawa, H. Ohmoto, K. Yoshino, Y. Shirakata, Y. Matsuzawa, K. Hashimoto, and N. Taniguchi (2000) Ectodomain shedding of epidermal growth factor receptor ligands is required for keratinocyte migration in cutaneous wound healing. J. Cell Biol. 151: 209–220.
Goldman, R. (2004) Growth factors and chronic wound healing: Past, present, and future. Adv. Skin Wound Care. 17: 24–35.
Kawada, K., G. Upadhyay, S. Ferandon, S. Janarthanan, M. Hall, J. Vilardaga, and V. Yajnik (2009) Cell migration is regulated by platelet-derived growth factor receptor endocytosis. Mol. Cell. Biol. 29: 4508–4518.
Sabine Werner, R. G. (2003) Regulation of wound healing by growth factors and cytokines. Physiol. Rev. 83: 835–870.
Pastar, I., O. Stojadinovic, N. C. Yin, H. Ramirez, A. G. Nusbaum, A. Sawaya, S. B. Patel, L. Khalid, R. R. Isseroff, and M. Tomic-Canic (2014) Epithelialization in wound healing: A comprehensive review. Adv. Wound Care 3: 445–464.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, H., Kim, D., Lim, SM. et al. Rice callus extracts for enhancing skin wound healing. Biotechnol Bioproc E 22, 352–358 (2017). https://doi.org/10.1007/s12257-017-0137-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-017-0137-9