Abstract
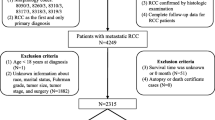
The aim of this study was to compare the Memorial Sloan-Kettering Cancer Center (MSKCC) and the Cleveland Clinic Foundation (CCF) models of classification of aRCC patients. In addition, the model developed from the pivotal trial of temsirolimus and those proposed by Motzer et al. in 2004, Escudier et al., Heng et al., Choueiri et al. and Bamias et al. were examined. An observational, retrospective study of patients starting first-line systemic therapy was conducted between 2008 and 2011. The variables used to evaluate the classification models were median overall survival (mOS) and median progression-free survival (mPFS). The comparison of different classification models was performed by comparing the area under the ROC (Receiver Operating Characteristic) curve (AUC) for time-dependent variables proposed by Heagerty. Eighty-eight patients were included. When the different models were compared, it was found that although based on the mOS, the Escudier model had better short-term (1-year) prognostic value, followed by the Heng model; in the long term, the models that presented a higher prognosis capacity were the Hudes and CCF models, closely followed by the Heng model. In addition, the Heng model had a slightly higher predictive ability than the other models. Based on the results, and in line with the European society for medical oncology (ESMO) guidelines, it appears that the model of Heng could be the best model to classify patients with aRCC and combines good short- and long-term prognostics while possessing better predictive ability and a more equal distribution of patients.


Similar content being viewed by others
References
Ljungberg B, Hanbury DC, Kuczyk MA et al (2007) Renal cell carcinoma guideline. Eur Urol 51:1502–1510
Sánchez Folgueras MV, Palacio Vázquez IP (2009) Registro hospitalario de tumores del Servicio de Salud del Principado de Asturias. Año 2009. Astursalud website. http://www.hca.es/huca/web/contenidos/servicios/rt/rt2012/rt2012.pdf. Accessed 05 Oct 2015
Chow W, Gridley G, Fraumeni J et al (2000) Obesity, hypertension and the risk of kidney cancer in men. N Engl J Med 343:1305–1311
Escudier B, Eisen T, Porta C et al (2014) Clinical practice guidelines renal cell carcinoma: ESMO clinical practice guidelines for diagnosis, treatment and follow-up clinical practice guidelines. Ann Oncol 25(3):49–56
NCNN Clinical practice guidelines in oncology. Version 1.2013. Kidney cancer. The National Comprehensive Cancer Network website http://www.nccn.org/professionals/physician_gls/f_guidelines_nojava.asp#site. Accessed 05 Oct 2015
Motzer RJ, Bacik J, Murphy BA et al (2002) Interferon-alfa as a comparative treatment for clinical trials of new therapies against advanced renal cell carcinoma. J Clin Oncol 20(1):289–296
Motzer RJ, Bacik J, Schwartz LH et al (2004) Prognostic factors for survival in previously treated patients with metastatic renal cell carcinoma. J Clin Oncol 22(3):454–463
Mekhail TM, Abou-Jawde RM, Boumerhi G et al (2005) Validation and extension of the memorial Sloan- Kettering prognostic factors model for survival in patients with previously untreated metastatic renal cell carcinoma. J Clin Oncol 23(4):832–841
Hudes G, Carducci M, Tomczak P et al (2007) Temsirolimus, interferon alfa, or both for advanced renal-cell carcinoma. N Engl J Med 356:2271–2281
Escudier B, Choueiri TK, Oudard S et al (2007) Prognostic factors of metastatic renal cell carcinoma after failure of immunotherapy: new paradigm from a large phase III trial with shark cartilage extract AE 941. J Urol 178(5):1901–1905
Choueiri TK, Garcia JA, Elson P et al (2007) Clinical factors associated with outcome in patients with metastatic clear-cell renal cell carcinoma treated with vascular endothelial growth factor-targeted therapy. Cancer 110(3):543–550
Heng DY, Xie W, Regan MM et al (2009) Prognostic factors for overall survival in patients with metastatic renal cell carcinoma treated with vascular endothelial growth factor-targeted agents: results from a large, multicenter study. J Clin Oncol 27(34):5794–5799
Bamias A, Karadimou A, Lampaki S et al (2010) Prognostic stratification of patients with advanced renal cell carcinoma treated with sunitinib: comparison with the memorial Sloan-Kettering prognostic factors model. BMC Cancer 2(10):45
Patil S, Figlin RA, Hutson TE et al (2011) Prognostic factors for progression-free and overall survival with sunitinib targeted therapy and with cytokine as first-line therapy in patients with metastatic renal cell carcinoma. Ann Oncol 22(2):295–300
Heagerty PJ, Zheng Y (2005) Survival model predictive accuracy and ROC curves. Biometrics 61(1):92–105
Sachin S (2010) Pharmacotherapy self-assessment program 6th edition. American College of Clinical Pharmacy website. http://www.accp.com/bookstore/p6_se.aspx. Accessed 10 Oct 2015
Aguiló M, Alba F, Barrio J et al (2007) Renal cell cancer. Pharm Lett 9(10):8592 (eLetter) http://www.dicaf.es/pharmletter.php#.VgQutn1kbGQ. Accessed 10 Oct 2015
Karakiewcz P, Sun M, Bellmunt J et al (2011) Prediction of progression-free survival rates after bevacizumab plus interferon alone in patients with metastatic renal cell carcinoma: comparion of a normogram to the Motzer criteria. Eur Urol 60:48–56
Sun M, Shariat SF, Cheng C et al (2011) Prognostic factors and predictive models in renal cell carcinoma: a contemporary review. Eur Urol 60:644–661
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Ethical Approval
The Ethics Committee of Central University Hospital of Asturias (Spain) approved the study.
Informed Consent
Consent was obtained for the use of patient data.
Rights and permissions
About this article
Cite this article
Sastre-Heres, A.J., Iglesias, I., Alaguero-Calero, M. et al. Comparative Study of Different Classification Models in Renal-Cell Carcinoma. Pathol. Oncol. Res. 25, 1357–1362 (2019). https://doi.org/10.1007/s12253-018-0385-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12253-018-0385-9