Abstract
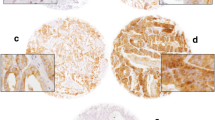
Cancer stem cells (CSCs) are the main players of prostate tumorigenesis thus; characterization of CSCs can pave the way for understanding the early detection, drug resistance, metastasis and relapse. The current study was conducted to evaluate the expression level and clinical significance of the potential CSC markers CD44 and CD133 in a series of prostate tissues. One hundred and forty eight prostate tissues composed of prostate cancer (PCa), high-grade prostatic intraepithelial neoplasia (HGPIN), and benign prostate hyperplasia (BPH) were immunostained for the putative CSC markers CD44 and CD133. Subsequently, the correlation between the expression of these markers and the clinicopathological variables was examined. A higher level of CD44 expression was observed in 42% of PCa, 57% of HGPIN, and 42% BPH tissues. In the case of CD133 expression PCa, HGPIN, and BPH samples demonstrated high immunoreactivity in 46%, 43%, and 42% of cells, respectively. Statistical analysis showed an inverse significant correlation between CD44 expression with Gleason score of PCa (P = 0.02), while no significant correlation was observed between CD133 expression and clinicopathological parameters. A significant reciprocal correlation was observed between the expression of two putative CSC markers CD44 and CD133 in PCa specimens while not indicating clinical significance. Further clinical investigation is required to consider these markers as targets of new therapeutic strategies for PCa





Similar content being viewed by others
References
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A (2015) Global cancer statistics, 2012. CA Cancer J Clin 65:87–108
Jemal A, Siegel R, Xu J, Ward E (2010) Cancer statistics, 2010. CA Cancer J Clin 60:277–300
Hurt EM, Kawasaki BT, Klarmann GJ, Thomas SB, Farrar WL (2008) CD44+ CD24− prostate cells are early cancer progenitor/stem cells that provide a model for patients with poor prognosis. Br J Cancer 98:756–765
van den Hoogen C, van der Horst G, Cheung H, Buijs JT, Lippitt JM, Guzmán-Ramírez N, Hamdy FC, Eaton CL, Thalmann GN, Cecchini MG (2010) High aldehyde dehydrogenase activity identifies tumor-initiating and metastasis-initiating cells in human prostate cancer. Cancer Res 70:5163–5173
Dubrovska A, Elliott J, Salamone RJ, Kim S, Aimone LJ, Walker JR, Watson J, Sauveur-Michel M, Garcia-Echeverria C, Cho CY (2010) Combination therapy targeting both tumor-initiating and differentiated cell populations in prostate carcinoma. Clin Cancer Res 16:5692–5702
Rangwala F, Omenetti A, Diehl AM (2010) Cancer stem cells: repair gone awry? J Oncol 2011:465343
Reyes EE, Kunovac SK, Duggan R, Kregel S, Griend DJV (2013) Growth kinetics of CD133-positive prostate cancer cells. Prostate 73:724–733
Ugolkov AV, Eisengart LJ, Luan C, Yang XJ (2011) Expression analysis of putative stem cell markers in human benign and malignant prostate. Prostate 71:18–25
AM DM, WG N, AK M, DS C (1998) Stem cell features of benign and malignant prostate epithelial cells. J Urol 160:2381–2392
Tirino V, Desiderio V, Paino F, De Rosa A, Papaccio F, La Noce M, Laino L, De Francesco F, Papaccio G (2013) Cancer stem cells in solid tumors: an overview and new approaches for their isolation and characterization. FASEB J 27:13–24
Marhaba R, Zöller M (2004) CD44 in cancer progression: adhesion, migration and growth regulation. J Mol Histol 35:211–231
Patrawala L, Calhoun-Davis T, Schneider-Broussard R, Tang DG (2007) Hierarchical organization of prostate cancer cells in xenograft tumors: the CD44+ α2β1+ cell population is enriched in tumor-initiating cells. Cancer Res 67:6796–6805
Collins AT, Berry PA, Hyde C, Stower MJ, Maitland NJ (2005) Prospective identification of tumorigenic prostate cancer stem cells. Cancer Res 65:10946–10951
Marker PC 2007 Highly purified CD44+ prostate cancer cells from xenograft human tumors are enriched in tumorigenic and metastatic progenitor cells: Patrawala L, Calhoun T, Schneider-Broussard R, Li H, Bhatia B, Tang S, Reilly JG, Chandra D, Zhou J, Claypool K, Coghlan L, Tang DG, Department of Carcinogenesis, The University of Texas MD Anderson Cancer Center, Science Park-Research Division, Smithville, TX. In: Urologic Oncology: Seminars and Original Investigations,. vol 3. Elsevier, pp 277–278
Nagabhushan M, Pretlow TG, Guo Y-J, Amini SB, Pretlow TP, Sy M-S (1996) Altered expression of CD44 in human prostate cancer during progression. Am J Clin Pathol 106:647–651
Kallakury BV, Yang F, Figge J, Smith KE, Kausik SJ, Tacy NJ, Fisher HA, Kaufman R, Figge H, Ross JS (1996) Decreased levels of CD44 protein and mRNA in prostate carcinoma: correlation with tumor grade and ploidy. Cancer 78:1461–1469
Simon RA, di Sant’Agnese PA, Huang L-S, Xu H, Yao JL, Yang Q, Liang S, Liu J, Yu R, Cheng L (2009) CD44 expression is a feature of prostatic small cell carcinoma and distinguishes it from its mimickers. Hum Pathol 40:252–258
Tirino V, Desiderio V, d’Aquino R, De Francesco F, Pirozzi G, Graziano A, Galderisi U, Cavaliere C, De Rosa A, Papaccio G (2008) Detection and characterization of CD133+ cancer stem cells in human solid tumours. PLoS One 3:e3469
Tirino V, Camerlingo R, Franco R, Malanga D, La Rocca A, Viglietto G, Rocco G, Pirozzi G (2009) The role of CD133 in the identification and characterisation of tumour-initiating cells in non-small-cell lung cancer. Eur J Cardiothorac Surg 36:446–453
Sabet MN, Rakhshan A, Erfani E, Madjd Z (2014) Co-expression of putative cancer stem cell markers, CD133 and nestin, in skin tumors. Asian Pac J Cancer Prev 15:8161–8169
Mehrazma M, Madjd Z, Kalantari E, Panahi M, Hendi A, Shariftabrizi A (2013) Expression of stem cell markers, CD133 and CD44, in pediatric solid tumors: a study using tissue microarray. Fetal Pediatr Pathol 32:192–204
Wognum AW, Eaves AC, Thomas TE (2003) Identification and isolation of hematopoietic stem cells. Arch Med Res 34:461–475
Richardson GD, Robson CN, Lang SH, Neal DE, Maitland NJ, Collins AT (2004) CD133, a novel marker for human prostatic epithelial stem cells. J Cell Sci 117:3539–3545
Miki J, Furusato B, Li H, Gu Y, Takahashi H, Egawa S, Sesterhenn IA, McLeod DG, Srivastava S, Rhim JS (2007) Identification of pPutative sStem cCell mMarkers, CD133 and CXCR4, in hTERT–Immortalized Primary Nonmalignant and Malignant Tumor-Derived Human Prostate Epithelial Cell Lines and in Prostate Cancer Specimens. Cancer Res 67:3153–3161
Epstein JI, Allsbrook WC Jr, Amin MB, Egevad LL (2005) The 2005 International Society of Urological Pathology (ISUP) consensus conference on Gleason grading of prostatic carcinoma. Am J Surg Pathol 29:1228–1242
Cheng L, Montironi R, Bostwick DG, Lopez-Beltran A, Berney DM (2012) Staging of prostate cancer. Histopathology 60:87–117. doi:10.1111/j.1365-2559.2011.04025.x
Madjd Z, Akbari ME, Zarnani AH, Khayamzadeh M, Kalantari E, Mojtabavi N (2013) Expression of EMSY, a novel BRCA2-link protein, is associated with lymph node metastasis and increased tumor size in breast carcinomas. Asian Pac J Cancer Prev 15:1783–1789
Singh SK, Clarke ID, Terasaki M, Bonn VE, Hawkins C, Squire J, Dirks PB (2003) Identification of a cancer stem cell in human brain tumors. Cancer Res 63:5821–5828
Kalantari E, Saadi FH, Asgari M, Shariftabrizi A, Roudi R, Madjd Z (2016) Increased expression of ALDH1A1 in prostate cancer is correlated with tumor aggressiveness: a tissue microarray study of Iranian patients. Appl Immunohistochem Mol Morphol AIMM Off Publ Soc Appl Immunohistochem. doi:10.1097/PAI.0000000000000343
Camp RL, Charette LA, Rimm DL (2000) Validation of tissue microarray technology in breast carcinoma. Lab Investig 80:1943–1949
Roudi R, Korourian A, Shariftabrizi A, Madjd Z (2015) Differential expression of cancer stem cell markers ALDH1 and CD133 in various lung cancer subtypes. Cancer Invest 33:294–302
Madjd Z, Ramezani B, Molanae S, Asadi-Lari M (2012) High expression of stem cell marker ALDH1 is associated with reduced BRCA1 in invasive breast carcinomas. Asian Pac J Cancer Prev 13:2973–2978
Walker R (2006) Quantification of immunohistochemistry—issues concerning methods, utility and semiquantitative assessment I. Histopathology 49:406–410
Nagel H, Laskawi R, Wahlers A, Hemmerlein B (2004) Expression of matrix metalloproteinases MMP-2, MMP-9 and their tissue inhibitors TIMP-1,-2, and-3 in benign and malignant tumours of the salivary gland. Histopathology 44:222–231
Madjd Z, Pinder S, Paish C, Ellis I, Carmichael J, Durrant L (2003) Loss of CD59 expression in breast tumours correlates with poor survival. J Pathol 200:633–639
Mohsenzadegan M, Madjd Z, Asgari M, Abolhasani M, Shekarabi M, Taeb J, Shariftabrizi A (2013) Reduced expression of NGEP is associated with high-grade prostate cancers: a tissue microarray analysis. Cancer Immunol Immunother 62:1609–1618
Taeb J, Asgari M, Abolhasani M, Farajollahi MM, Madjd Z (2014) Expression of prostate stem cell antigen (PSCA) in prostate cancer: a tissue microarray study of Iranian patients. Pathology-research and. Practice 210:18–23
Kerfoot C, Huang W, Rotenberg SA (2004) Immunohistochemical analysis of advanced human breast carcinomas reveals downregulation of protein kinase Cα. J Histochem Cytochem 52:419–422
McCarty K Jr, Miller L, Cox E, Konrath J, McCarty K Sr (1985) Estrogen receptor analyses. Correlation of biochemical and immunohistochemical methods using monoclonal antireceptor antibodies. Arch Pathol Lab Med 109:716–721
Sharma BK, Manglik V, Elias EG (2010) Immuno-expression of human melanoma stem cell markers in tissues at different stages of the disease. J Surg Res 163:e11–e15
Hemmati HD, Nakano I, Lazareff JA, Masterman-Smith M, Geschwind DH, Bronner-Fraser M, Kornblum HI (2003) Cancerous stem cells can arise from pediatric brain tumors. Proc Natl Acad Sci 100:15178–15183
Chang B, Liu G, Xue F, Rosen DG, Xiao L, Wang X, Liu J (2009) ALDH1 expression correlates with favorable prognosis in ovarian cancers. Mod Pathol 22:817–823
Nguyen LV, Vanner R, Dirks P, Eaves CJ (2012) Cancer stem cells: an evolving concept. Nat Rev Cancer 12:133–143
Sullivan JP, Spinola M, Dodge M, Raso MG, Behrens C, Gao B, Schuster K, Shao C, Larsen JE, Sullivan LA (2010) Aldehyde dehydrogenase activity selects for lung adenocarcinoma stem cells dependent on notch signaling. Cancer Res 70:9937–9948
Castellón EA, Valenzuela R, Lillo J, Castillo V, Contreras HR, Gallegos I, Mercado A, Huidobro C (2012) Molecular signature of cancer stem cells isolated from prostate carcinoma and expression of stem markers in different Gleason grades and metastasis. Biol Res 45:297–305
Jaworska D, Król W, Szliszka E (2015) Prostate cancer stem cells: research advances. Int J Mol Sci 16:27433–27449
Patrawala L, Calhoun T, Schneider-Broussard R, Li H, Bhatia B, Tang S, Reilly J, Chandra D, Zhou J, Claypool K (2006) Highly purified CD44+ prostate cancer cells from xenograft human tumors are enriched in tumorigenic and metastatic progenitor cells. Oncogene 25:1696–1708
Paradis V, Eschwege P, Loric S, Dumas F, Ba N, Benoit G, Jardin A, Bedossa P (1998) De novo expression of CD44 in prostate carcinoma is correlated with systemic dissemination of prostate cancer. J Clin Pathol 51:798–802
De Marzo AM, Bradshaw C, Sauvageot J, Epstein JI, Miller GJ (1998) CD44 and CD44v6 downregulation in clinical prostatic carcinoma: relation to Gleason grade and cytoarchitecture. Prostate 34:162–168
Corbeil D, Röper K, Fargeas CA, Joester A, Huttner WB (2001) Prominin: a story of cholesterol, plasma membrane protrusions and human pathology. Traffic 2:82–91
Wang C-C, De Marzo AM, Lotan TL, Epstein JI (2015) Overlap of CD44 expression between prostatic small cell carcinoma and acinar adenocarcinoma. Hum Pathol 46:554–557
Trerotola M, Rathore S, Goel HL, Li J, Alberti S, Piantelli M, Adams D, Jiang Z, Languino LR (2010) CD133, trop-2 and alpha2beta1 integrin surface receptors as markers of putative human prostate cancer stem cells. Am J Transl Res 2:135–144
Bussolati B, Bruno S, Grange C, Buttiglieri S, Deregibus MC, Cantino D, Camussi G (2005) Isolation of renal progenitor cells from adult human kidney. Am J Pathol 166:545–555
Eramo A, Lotti F, Sette G, Pilozzi E, Biffoni M, Di Virgilio A, Conticello C, Ruco L, Peschle C, De Maria R (2008) Identification and expansion of the tumorigenic lung cancer stem cell population. Cell Death Differ 15:504–514
O’Brien CA, Pollett A, Gallinger S, Dick JE (2007) A human colon cancer cell capable of initiating tumour growth in immunodeficient mice. Nature 445(7123):106–110
Ma S, Lee T, Zheng B, Chan K, Guan X (2008) CD133+ HCC cancer stem cells confer chemoresistance by preferential expression of the Akt/PKB survival pathway. Oncogene 27:1749–1758
Liu G, Yuan X, Zeng Z, Tunici P, Ng H, Abdulkadir IR, Lu L, Irvin D, Black KL, John SY (2006) Analysis of gene expression and chemoresistance of CD133+ cancer stem cells in glioblastoma. Mol Cancer 5:67
Vatansever H, Gumus B, Aydogdu O, Sivrikoz O, Türköz-Uluer E, Kivanç M, Ateşçi Y, Bugdayci H (2014) The role of stem/progenitor cells and Wnt/β-catenin signaling pathway in the patients with prostate cancer. Minerva urologica e nefrologica = The Italian journal of urology and nephrology 66:249–255
Fargeas C, Huttner W, Corbeil D (2007) Nomenclature of prominin-1 (CD133) splice variants–an update. Tissue Antigens 69:602–606
Irollo E, Pirozzi G (2013) CD133: to be or not to be, is this the real question. Am J Transl Res 5:563–581
Acknowledgments
This study was supported by Iran University of Medical Sciences [Project 94-1207 and 1491 « Immunohistochemical expression of Cancer Stem Cell markers CD44 and CD133 in prostate cancer »].
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Informed Consent
“Informed consent was obtained from all individual participants included in the study.”
Ethical Approval
“All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted.
Rights and permissions
About this article
Cite this article
Kalantari, E., Asgari, M., Nikpanah, S. et al. Co-Expression of Putative Cancer Stem Cell Markers CD44 and CD133 in Prostate Carcinomas. Pathol. Oncol. Res. 23, 793–802 (2017). https://doi.org/10.1007/s12253-016-0169-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12253-016-0169-z