Abstract
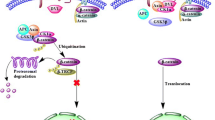
The X protein (HBx) of Human hepatitis B virus (HBV) acts as an indirect transcriptional transactivator to regulate the expression of many viral and cellular genes, as well as playing a critical role in pathogenesis and the development of Hepatocellular carcinoma (HCC). Here we described the biological effects of HBx in association with four cellular factors, including inflammatory factors (COX-2 and iNOS), oncoprotein (Ras), and a newly identified tumor suppressor (YueF). The characteristics of these effectors, which might be associated with hepatocellular carcinoma, are also discussed.
Article PDF
Similar content being viewed by others
References
Asare G A, Mossanda K S, Kew M C, et al. 2006. Hepatocellular carcinoma caused by iron overload: a possible mechanism of direct hepatocarcinogenicity. Toxicology, 219: 41–52.
Bertoletti A, Gehring A J. 2006. The immune response during hepatitis B virus infection. J Gen Virol, 87: 1439–1449.
Chen C N, Hsieh F J, Cheng Y M, et al. 2006. Expression of inducible nitric oxide synthase and cycloo-xygenase-2 in angiogenesis and clinical outcome of human gastric cancer. J Surg Oncol, 94: 226–233.
Cullis E R, Kalber T L, Ashton S E, et al. 2006. Tumour overexpression of inducible nitric oxide synthase (iNOS) increases angiogenesis and may modulate the anti-tumour effects of the vascular disrupting agent ZD6126. Microvasc Res, 71:76–84.
Dionne R. 1999. COX-2 inhibitors—IBC conference. 12–13 April 1999, Coronado, CA, USA. Idrugs, 2: 664–666.
Dooley A, Low S Y, Holmes A, et al. 2008. Nitric oxide synthase expression and activity in the tight-skin mouse model of fibrosis. Rheumatology (Oxford), 47: 272–280.
Fabregat I, Roncero C, Fernández M. 2007. Survival and apoptosis: a dysregulated balance in liver cancer. Liver Int, 27: 155–162.
He Y, Yang F, Wang F, et al. 2007. The upregulation of expressed proteins in HepG2 cells transfected by the recombinant plasmid-containing HBx gene. Scand J Immunol, 65: 249–256.
Huang J, Wu K, Zhang J, et al. 2008. Putative tumor suppressor YueF affects the functions of hepatitis B virus X protein in hepatoma cell apoptosis and p53 expression. Biotechnol Lett, 30: 235–242.
Kim Y C, Song K S, Yoon G, et al. 2001. Activated ras oncogene collaborates with HBx gene of hepatitis B virus to transform cells by suppressing HBx-mediated apoptosis. Oncogene, 20: 16–23.
Lara-Pezzi E, Gómez-Gaviro M V, Gálvez B G, et al. 2002. The hepatitis B virus X protein promotes tumor cell invasion by inducing membrane-type matrix metal-loproteinase-1 and cyclooxygenase-2 expression. J Clin Invest, 110: 1831–1838.
Liu M, Yang Y, Gu C, et al. 2007. Spike protein of SARS-CoV stimulates cyclooxygenase-2 expression via both calcium-dependent and calcium-independent protein kinase C pathways. FASEB J, 21: 1586–1596.
McClain SL, Clippinger A J, Lizzano R, et al. 2007. Hepatitis B virus replication is associated with an HBx-dependent mitochondrion-regulated increase in cytosolic calcium levels. J Virol, 81: 12061–12065.
McGinty A, Chang Y W, Sorokin A, et al. 2000. Cyclooxygenase-2 expression inhibits trophic withdrawal apoptosis in nerve growth factor-differentiated PC12 cells. J Biol Chem, 275: 12095–12101.
Miyamoto T, Saika S, Ueyama T, et al. 2006. Cyclooxygenase 2 expression in rat corneas after ethanol exposure. J Cataract Refract Surg, 32:1736–1740.
Oishi N, Shilagardi K, Nakamoto Y, et al. 2007. Hepatitis B virus X protein overcomes oncogenic RAS-induced senescence in human immortalized cells. Cancer Sci, 98: 1540–1548.
Peng G, Zhang F, Zhang Q, et al. 2007. Borna disease virus P protein inhibits nitric oxide synthase gene expression in astrocytes. Virology, 366: 446–452.
Pesce C, Grattarola M, Menini S, et al. 2006. Cyclooxygenase 2 expression in vessels and nerves in reversal reaction leprosy. Am J Trop Med Hyg, 74: 1076–1077.
Seeger C, Mason W S. 2000. Hepatitis B virus biology. Microbiol Mol Biol Rev, 64: 51–68.
Tsujii M, Kawano S, Tsuji S, et al. 1998. Cyclooxygenase regulates angiogenesis induced by colon cancer cells. Cell, 93: 705–716.
Wei W, Huang W, Pan Y, et al. 2006. Functional switch of viral protein HBx on cell apoptosis, transformation, and tumorigenesis in association with oncoprotein Ras. Cancer Lett, 244: 119–128.
Williams C S, Tsujii M, Reese J, et al. 2000. Host cyclooxygenase-2 modulates carcinoma growth. J Clin Investig, 105: 1589–1594.
Wullaert A, van Loo G, Heyninck K, et al. 2007. Hepatic tumor necrosis factor signaling and nuclear factor-kappaB: effects on liver homeostasis and beyond. Endocr Rev, 28: 365–386.
Yan X, Hao Q, Mu Y, et al. 2006. Nucleocapsid protein of SARS-CoV activates the expression of cyclooxygenase-2 by binding directly to regulatory elements for nuclear factor-kappa B and CCAAT/enhancer binding protein. Int J Biochem Cell Biol, 38: 1417–1428.
Yang Q, Ito S, Gonzalez F J. 2007. Hepatocyte-restricted constitutive activation of PPAR alpha induces hepatoproliferation but not hepatocarcinogenesis. Carcinogenesis, 28: 1171–1177.
Yun C, Cho H, Kim S J, et al. 2004. Mitotic aberration coupled with centrosome amplification is induced by hepatitis B virus X oncoprotein via the Ras-mitogen-activated protein/extracellular signal-regulated kinase-mitogen-activated protein pathway. Mol Cancer Res, 2: 159–2169.
Zhang J L, Zhao W G, Wu K L, et al. 2005. Human hepatitis B virus X protein promotes cell proliferation and inhibits cell apoptosis through interacting with a serine protease Hepsin. Arch Virol, 150: 721–741.
Author information
Authors and Affiliations
Corresponding author
Additional information
Foundation items: the Major State Basic Research Development Program of China (“973” project No. 2005CB5 22901 and No. 2005CB522905); The National Natural Science Foundation of China (No. 30470087 and No. 30730001), The Department of Science Technology of Hubei Province (No. 2005S2354), and Program for Changjiang Scholars and Innovative Research Team in University (PCSIRT) to J. Wu.
Rights and permissions
About this article
Cite this article
Wang, Sq., Wu, Jg. Biological effects of HBV X protein on hepatocellular carcinogenesis in association with cellular factors. Virol. Sin. 23, 146–151 (2008). https://doi.org/10.1007/s12250-008-2952-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12250-008-2952-9