Abstract
Purpose
Microemulsions (MEs) are homogeneous, optically clear and thermodynamically stable systems intended to increase the bioavaibility, solubility, and entrapment of drugs. In this study, a new system of oil-in-water MEs was formulated with Fusidic acid as a drug. MEs were composed of Isopropyl myristate, non-ionic surfactants tween 80, 1-propanol and water.
Methods
The optimum weight ratios of components and MEs areas were determined by pseudoternary phase diagram. The prepared microemulsions were evaluated regarding their stability, pH, refractive index, conductivity, viscosity, particle size, zeta potential, surface tension and partition coefficient.
Results
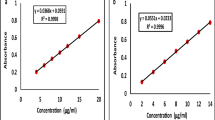
These parameters suggested that the optimized MEs were stable and optically clear.
Conclusion
This study reveals that the product can be used for ocular treatment.


Similar content being viewed by others
References
Jalali-Jivan M, Garavand F, Jafari SM. Microemulsions as nanoreactors for the solubilization, separation, purification and encapsulation of bioactive compounds. Adv Colloid Interface Sci 2020;283. https://doi.org/10.1016/j.cis.2020.102227.
Shinde UA, Modani SH, Singh KH. Design and development of repaglinide microemulsion gel for transdermal delivery. AAPS Pharm Sci Tech. 2018;19(1):315–325. https://doi.org/10.1208/s12249-017-0811-4
Mena-Hernández J, Jung-Cook H, Llaguno-Munive M, García-López P, Ganem-Rondero A, López-Ramírez S, Barragán-Aroche F, Rivera-Huerta M, Mayet-Cruz L. Preparation and evaluation of mebendazole microemulsion for intranasal delivery: an alternative approach for glioblastoma treatment. AAPS PharmSciTech. 2020;21(7):264. https://doi.org/10.1208/s12249-020-01805-x.
Vandamme TF. Microemulsions as ocular drug delivery systems: recent developments and future challenges. Prog Retin Eye Res 2002;21: 15–34. https://doi.org/10.1016/S1350-9462(01)00017-9.
Jayne ML, Rees GD (2012) Microemulsion-based media as novel drug delivery systems. Adv Drug Deliv Rev 64: 175–193. https://doi.org/10.1016/S0169-409X(00)00103-4.
Zhu, Y, Xu W, Zhang J, Liao Y, Firempong CK, Adu-Frimpong M, Deng W, Zhang H, Yu J, Xu X (2019) Self-microemulsifying drug delivery system for improved oral delivery of limonene: preparation, characterization, in vitro and in vivo evaluation. AAPS PharmSciTech. 2019;20(4):153. https://doi.org/10.1208/s12249-019-1361-8
Grassiri B, Zambito Y, Bernkop-Schnürch A. Strategies to prolong the residence time of drug delivery systems on ocular surface. Advances in Colloid and Interface Science. 2021;288. https://doi.org/10.1016/j.cis.2020.102342.
Peter B Taylor, Eileen M Burd, And Khalid F Tabbara. Corneal and intraocular penetration of topical and subconjunctival fusidic acid. British Journal of Ophthalmology. 1987; 71 :598–601. https://doi.org/10.1136/bjo.71.8.598.
Cé R, Pacheco BZ, Ciocheta TM, Barbosa FS, de CS Alves A, Dallemole DR, Lavayen V, Guterres SS, Steppe M, Pohlmann AR. Antibacterial activity against Gram-positive bacteria using fusidic acid-loaded lipid-core nanocapsules. React Funct Polym. 2021;162. https://doi.org/10.1016/j.reactfunctpolym.2021.104876.
Jyoti K, Malik G, Chaudhary M, Sharma M, Goswami M, Katare OP, Singh SB, Madan J (2020) Chitosan and phospholipid assisted topical fusidic acid drug delivery in burn wound: strategies to conquer pharmaceutical and clinical challenges, opportunities and future panorama. Int J BiolMacromol. 2020;161:325–335. https://doi.org/10.1016/j.ijbiomac.2020.05.230.
Okur ME, Ayla S, Yozgatl Vı, Aksu NB, Yoltase A, Orak D, Sipahi H, Okur NÜ. Evaluation of burn wound healing activity of novel fusidic acid loaded microemulsion based gel in male Wistar albino rats. Saudi Pharmaceut J. 2020;28: 338–348. https://doi.org/10.1016/j.jsps.2020.01.015.
Aksu NB, Yozgatl Vı, Okur ME, Ayla S, Yoltaş A, Okur NÜ. Preparation and evaluation of QbD based fusidic acid loaded in situ gel formulations for burn wound treatment. J Drug Deliv Sci Technol. 2019;52: 110–121. https://doi.org/10.1016/j.jddst.2019.04.015.
Chhibber T, Wadhwa S, Chadha P, Sharma G, Katare OP. Phospholipid structured microemulsion as effective carrier system with potential in methicillin sensitive Staphylococcus aureus (MSSA) involved burn wound infection. J Drug Target. 2015;23 (10): 943–52. https://doi.org/10.3109/1061186X.2015.1048518
Wadhwa S, Singh B, Sharma G, Raza K, Katare OP. Liposomal fusidic acid as a potential delivery system: a new paradigm in the treatment of chronic plaque psoriasis. Drug Deliv. 2016;23(4): 1204–13. https://doi.org/10.3109/10717544.2015.1110845
Rashid MA, Naz T, Abbas M, Nazir S, Younas N, Majeed S, Qureshi N, Akhtar MN. Chloramphenicol loaded microemulsions: development, characterization and stability. Coll Interf Sci Commun. 2019;28: 41–48. https://doi.org/10.1016/j.colcom.2018.11.006.
Kumar R, Sinha VR. Preparation and optimization of voriconazole microemulsion forocular delivery. Coll Surf B: Biointer 2014;117:82–88. https://doi.org/10.1016/j.colsurfb.2014.02.007.
Nazar MF, Khan AM, Shah SS. Microemulsion system with improved loading of piroxicam: a study of microstructure. AAPS PharmSciTech. 2009;10(4):1286–94. https://doi.org/10.1208/s12249-009-9328-9.
Lopez-Alemany A, Montes-Mico R, Gracia-Valldecabres M. Ocular physiology and artificial tears. J Am Optom Assoc. 1999;70:455–60.
Radomska-Soukharev A, Wojciechowska J. Microemulsions as potential ocular drug delivery systems: phase diagrams and physical properties depending on ingredients. Acta Pol Pharm. 2005;62(6):465–71.
Ammar HO, Salama HA, Ghorab M, Mahmoud AA. Nanoemulsion as a potential ophthalmic delivery system for dorzolamide hydrochloride. AAPS PharmSciTech. 2009;10:808–19. https://doi.org/10.1208/s12249-009-9268-4.
Vieira LB, Casimiro MW, Santos RG. Surface tension of aqueous amoxicillin+ peg systems. Colloid Interf Sci Commun. 2018;24:93–7. https://doi.org/10.1016/j.colcom.2018.04.006.
Kahlweit M, Busse G, Faulhaber B, Eibl H. Preparing nontoxic microemulsions. Langmuir. 1995;11(11):4185–7. https://doi.org/10.1021/la970583z.
Lu GW, Gao P. Handbook of non-invasive drug delivery systems, William Andrew Publishing; 2010;59–94. https://doi.org/10.1016/B978-0-8155-2025-2.10003-4.
Silva-Cunha A, da Silva GR, de Castro WV, Fialho SL. Evaluation of the pharmacokinetics and ocular tolerance of a microemulsion containing tacrolimus. J Ocul Pharmacol Ther. 2014;30(1):59–65. https://doi.org/10.1089/jop.2013.0128.
Raid GA, Wen J. Microemulsions as drug delivery systems. Pharmaceutical manufacturing handbook: production and processes, edited by Shayne Cox Gad. 2007;769–792. https://doi.org/10.1002/9780470259818.ch20.
Ustundag ON, Caglar ES, Siafaka PI. Novel ocular drug delivery systems: an update on microemulsions. J Ocul Pharmacol Ther. 2020;36:342–54. https://doi.org/10.1089/jop.2019.0135.
Nidhin M, Indumathy R, Sreeram KJ, Nair BU. Synthesis of iron oxide nanoparticles of narrow size distribution on polysaccharide templates. Bull Mater Sci. 2008;31:93–6. https://doi.org/10.1007/s12034-008-0016-2.
Jadhav CM, Shinde SM, Kate VK, Payghan SA. Investigating application of nonaqueous microemulsion for drug delivery. Asian J Biomed Pharm Sci. 2014;4(29):1–9. https://doi.org/10.1007/s40097-014-0141-y.
Araujo J, Vega E, Lopes C, Egea MA, Garcia ML, Souto EB. Effect of polymer viscosity on physicochemical properties and ocular tolerance of FB-loaded PLGA nanospheres. Colloids Surf B Biointerfaces. 2009;2009(72):48–56. https://doi.org/10.1016/j.colsurfb.2009.03.028.
Kalam MA, Alshamsan A, Aljuffali IA, Mishra AK, Sultana Y. Delivery of gatifloxacin using microemulsion as vehicle: formulation, evaluation, transcorneal permeation and aqueous humor drug determination. Drug Deliv. 2016;23 (3): 896–907. https://doi.org/10.3109/10717544.2014.920432.
Morsi N, Ibrahim M, Refai H, El Sorogy H. Nanoemulsion-based electrolyte triggered in situ gel for ocular delivery of acetazolamide. Eur J Pharm Sci. 2017;104:302–14. https://doi.org/10.1016/j.ejps.2017.04.013.
Lv F-F, Zheng L-Q, Tung C-H. Phase behavior of the microemulsions and the stability of the chloramphenicol in the microemulsion-based ocular drug delivery system. Int J Pharm. 2005;301:237–46. https://doi.org/10.1016/j.ijpharm.2005.06.006.
Lipinski CA, Lombardo F, Dominy BW, Feeney PJ. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv Drug Deliv Rev. 2001;46:3–26. https://doi.org/10.1016/s0169-409x(00)00129-0.
Wong HL, Bendayan R, Rauth AM, Xue HY, Babakhanian K, Wu XY. A mechanistic study of enhanced doxorubicin uptake and retention in multidrug resistant breast cancer cells using a polymer-lipid hybrid nanoparticle system. J Pharmacol Exp Ther. 2006;317:1372–81. https://doi.org/10.1124/jpet.106.101154.
Nachari Y, Jabbari M. A case study on the partitioning of pharmaceutical compound naproxen in edible oil-water system in the presence of ionic and nonionic surfactants. J Taiwan Inst Chem Eng. 2021;000:1–5. https://doi.org/10.1016/j.jtice.2021.02.012.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lamoudi, L., Akretche, S., Hadjsadok, A. et al. Fusidic Acid Microemulsion Based on a Pseudoternary Phase Diagram: Development, Characterization, and Evaluation. J Pharm Innov 18, 621–628 (2023). https://doi.org/10.1007/s12247-022-09668-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12247-022-09668-4