Abstract
Purpose
Microencapsulation is a technique employed in the development of controlled drug delivery systems. It is beneficial in reducing dosing frequency, ensuring targeted drug delivery, and improving drug bioavailability. The objective of this study was to evaluate the effectiveness of Cyperus esculentus starch and its derivative in the development of microbeads for sustained delivery of ibuprofen.
Methods
Ibuprofen-loaded microbeads were prepared by ionotropic gelation using Cyperus esculentus starch and its derivative in combination with sodium alginate at concentrations of 1:1, 1:2, and 2:1 and calcium chloride solution as cross-linker. Morphology of the microbeads by scanning electron microscopy (SEM), Fourier transform infrared (FT-IR), and differential scanning calorimetry (DSC) were investigated. Entrapment efficiency, swelling capacity, mucoadhesion, and in vitro drug release were also evaluated.
Results
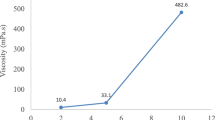
SEM showed that the microbeads were spherical to irregular in shape, FT-IR returned prominent peaks specific for ibuprofen and absence of new peaks, DSC revealed evidence of entrapment of ibuprofen in the microbeads, and entrapment efficiency ranged between 46.05 and 89.86%. Microbeads prepared with native and cross-linked starches showed better mucoadhesion, and those prepared with cross-linked starch blend exhibited highest swelling capacity. In vitro release was pH dependent, and increasing the concentration of cross-linked starch in the blend caused greater retardation of drug release (45.19%) than the formulations containing native starch-alginate blend or sodium alginate alone.
Conclusion
The blend of cross-linked Cyperus esculentus starch and sodium alginate shows propensity to sustain ibuprofen release and could be exploited for targeted delivery especially to the lower GIT.








Similar content being viewed by others
References
Kumar VV, Sivakumar T, Tamizh TM. (2011). Colon targeting drug delivery system: a review on recent approaches. Int J Pharm Bio Sci. 2011;2(1):11–9.
Berkland C, Kipper MJ, Narasimhan B, Kim KK, Pack DW. Microsphere size, precipitation kinetics and drug distribution control drug release from biodegradable polyanhydride microspheres. J Ctrl Rel. 2004;8:129–41.
Patil P, Chavanke D, Wagh M. A review on ionotropic gelation method: novel approach for controlled gastroretentive gelispheres. Int J Pharm Pharm Sci. 2012;4:27–32.
Belyaeva E, Valle DD, Neufeld RJ, Ponceleta D. New approach to the formulation of hydrogel beads by emulsification/thermal gelation using a static mixer. Chem Eng Sci. 2004;59(2):2913–20.
Aswathy KS, Abraham AM, Jomy L, Mehaladevi R, John RK. Formulation and evaluation of Etodolac alginate beads prepared by ionotropic gelation for sustained release. J Sci Innov Res. 2014;3:527–53.
Fontes GC, Calado VMA, Rossi AM, Miguez da Rocha-Leão HM. Characterization of antibiotic-loaded alginate-OSA starch microbeads produced by ionotropic pregelation. Biomed Res Int. 2013:1–12.
Hari BNV, Praneeth T, Prathyusha T, Mounika K, Devi DR. Development of starch-gelatin complex microspheres as sustained release delivery system. J Adv Pharm Tech Res. 2012;3:182–7.
Okunlola A, Adewusi SA. Development of theophylline microbeads using pregelatinized breadfruit starch (Artocarpus altilis) as a novel co-polymer for controlled release. Adv Pharm Bull. 2019;9:93–101.
Blemur L, Canh T, Marococci L, Pietrangeli P, Mateescu MA. Carboxymethyl starch/alginate microspheres containing diamine oxidase for intestinal targeting. Biotechnol Appl Biochem. 2016;63:344–53.
Jha A, Bhattachraya A. Preparation and evaluation of sweet potato starch-blended sodium alginaye microbeads. Asian J Pharm. 2009;3:229–303.
Biswas N, Sahoo RK. Tapioca starch blended alginate mucoadhesive-floating beads for intragastric delivery of metoprolol tartrate. Int J Biol Macromol. 2016;83:61–70.
Kim YJ, Kim J, Park GH, Yang YL, Yoon Y, Kim S, et al. Multifunctional drug delivery system using starch-alginate beads for controlled release. Biol Pharm Bull. 2005;28:394–7.
Leo E, Forni F, Bernabe MT. Surface drug removal from ibuprofen-loaded PLA microspheres. Int J Pharm. 2000;196:1–9.
Malakar J, Nayak AK, Das A. Modified starch (cationized)-alginate beads containing aceclofenac: formulation optimization, using central composite design. Starch-Starke. 2013;65:603–12.
Rapee KR, Wirinta K, Pongjanyakul TT. Modification of alginate beads using gelatinized and ungelatinized arrowroot (Tacca leontopetaloides) starch for drug delivery. Int J Biol Macromol. 2018;118:683–92.
Fontes GC, Finotelli PV, Rossi AM, Rocha-Leão MHM. Optimization of penicillin G microencapsulation with OSA starch by factorial design. Chem Eng Trans. 2012;27:85–90.
Martins M, Barros AA, Quraishi S, Gurikov P, Raman SP, Smirnova I, et al. Preparation of macroporous alginate-based aerogels for biomedical applications. J Supercrit Fluids. 2015;106:152–7.
Feng J, Dou J, Wu Z, Yin D, Wu W. Controlled release of biological control agents for preventing aflatoxin contamination from starch-alginate beads. Molecules. 2019;24:1858.
Onyido I, Sha’Ato R, Nnamonu LA. Environmentally friendly formulations of Trifluralin based on alginate modified starch. J Environ Prot. 2012;3:1085–93.
Singh B, Sharma DK, Gupta A. The controlled and sustained release of a fungicide from starch and alginate beads. J Environ Sci Health. 2009;44:113–22.
Hoover R. Composition, molecular structure, and physicochemical properties of tuber and root starches: a review. Carbohydr Polym. 2001;45:253–67.
Morthy SN. Starch and starch derivatives in food. Trends Carbohyd Chem. 1996;425:133–9.
Abano EE, Amoah KK. Effect of moisture content on the physical properties of tigernut (Cyperus esculentus). Asian J Agric Res. 2001;5:56–66.
Rubert J, Sebastià N, Soriano JM, Soler C, Mañes J. One-year monitoring of aflatoxins and ochratoxin a in tiger-nuts and their beverages. Food Chem. 2011;127:822–6.
Azubuike CP, Fabiyi RO, Oseni BA, Igwilo CI. Evaluation of disintegrant potential of carboxymethyl starch derived from Cyperus esculentus (Cyperaceae) tubers. Trop J Nat Prdt Res. 2019;3:246–51.
Builders PF, Anwunobi PA, Mbah CC, Adikwu MA. New direct compression excipient from tigernut starch: physicochemical and functional properties. AAPS PharmSciTech. 2013;14:818–27.
Okorie O, Azaka JE, Ibeshi E. Evaluation of the suspending properties of cyperus esculentus (Tiger nut) starch in sulphadimidine suspension. Am J Biomed Sci Eng. 2016;1:1–7.
Onyinye DO, Azubuike CP, Aloko S, Ologunagba MO, Igwilo CI. Characterization and disintegrant potential of phosphorylated tiger nut (Cyperus esculentus) starch in immediate release ibuprofen tablet formulation. Dhaka Univ J Pharm Sci. 2019;18:21–9.
Olayemi B, Isimi CY, Ekere K, Ajeh JI, Okoh JE, Emeje M. Green preparation of citric acid cross-linked starch for improvement of physico-chemical properties of Cyperus starch. In press; Turkish J Pharm. 2020. https://doi.org/10.4274/tjps.65624.
Wolfe MM, Lichtenstein DR, Singh G. Gastrointestinal toxicity of non-steroidal anti-inflammatory drugs. N Engl J Med. 1999;340:1888–99.
Hemalatha CH, Vasavi G, Kumar A, Sriram N. Formulation and development of gliclazide microspheres for pharmaceutical evaluations. Int J Adv Pharm. 2014;4:83–92.
Sharma VK, Bhattacharya A. Release of metformin hydrochloride from Ispaghula-sodium alginate beads adhered on cock intestinal mucosa. Indian J Pharm Educ Res. 2008;42:363–70.
Siepmann J, Streubel A, Peppas NA. Understanding and predicting drug delivery from hydrophilic matrix tablets using the sequential layer model. Pharm Res. 2002;19:306–14.
Artifin DY, Lee LY, Wang CH. Mathematical modeling and simulation of drug release from microspheres: implication to drug delivery systems. Adv Drug Deliv Rev. 2006;58:1274–325.
Al-Musa S, Fara DA, Badwan AA. Evaluation of parameters involved in preparation and release of drug loaded in crosslinked matrices of alginate. J Ctrl Rel. 1999;57:223–32.
Odeku OA, Okunlola A, Lamprecht A. Microbead design for sustained drug release using four natural gums. Int J Biol Macromol. 2013;58:113–20.
Akin-Ajani OD, Ajala TO, Ikehin M. Date mucilage as co-polymer in metformin-loaded microbeads for controlled release. J Excip Food Chem. 2019;10:3–12.
Martic M, Tatic I, Markovic S, Kujundzic N, Kostrun S. Synthesis, biological activity and molecular modelling studies of novel COX-1 inhibitors. Eur J Med Chem. 2004;39:141–51.
Mahaveer DK, Anandrao RK, Mahadevappa YK, Tejraj MA. In vitro release study of verapamil hydrochloride through sodium alginate interpenetrating monolithic membranes. Drug Dev Ind Pharm. 2001;27:1107–14.
Pahwa R, Kumar V, Kohli K. Alginate beads prepared by ionotropic gelation technique: formulation design. Res J Chem Sci. 2015;5:45–7.
Singh PK, Shukla TS, Easwari TS, Kumar S, Chudhary R. Formulation development and evaluation of mucoadhesive oral dosage form containing clarithromycin using different mucoadhesive polymers. Int J Pharm Sci Health Care. 2012;2:159–71.
Shivhare UD, Marthar VB, Shrivastava CG, Ramteke VI. Preparation of microbeads by different techniques and study of their influence. J Adv Pharm Educ Res. 2013;3:279–89.
Al-Kassas RS, Al-Gohary OMN, Al-Faadhel MM. Controlling of systemic absorption of gliclazide through incorporation into alginate beads. Int J Pharm. 2007;341:230–7.
Deasy PB, Collins AEM, Maccarthy DJ, Russell RJ. Use of strips containing tetracycline hydrochloride or metronidazole for the treatment of advanced periodontal disease. J Pharm Pharmacol. 1989;41:694–9.
Kowalski G, Kijowska K, Witczak K, Kuterasiński K, Łukasiewicz M. Synthesis and effect of structure on swelling properties of hydrogels based on high methylated pectin and acrylic polymers. Polymers. 2019;11:1–16.
Alexander A, Ajazuddin S, Tripathi DK, Verma T, Maurya J, Patel S. Mechanism responsible for mucoadhesion of mucoadhesive drug delivery system: a review. Int J Appl Biol Pharm Technol. 2011;2:434–45.
Jelvehgari M, Vajihe M, Seyed HM. Preparation and evaluation of mucoadhesive beads/discs of alginate and algino-pectinate of piroxicam for colon-specific drug delivery via oral route. Jundishapur J Nat Pharm Prdt. 2014;9:1–10.
Cui S, Yao B, Gao M, Sun X, Gou D, Hu J, et al. Effects of pectin structure and crosslinking method on the properties of cross-linked pectin nanofibers. Carbohydr Polym. 2017;157:766–74.
Khan AB, Mahamana R, Pal E. Review on mucoadhesive drug delivery system: novel approaches in modern era. RGUHS J Pharm Sci. 2014;4:128–41.
Raida S. Controlling of systemic absorption of gliclazide through incorporation into alginate beads. J Microencapsul. 2007;341:230–7.
El Maghraby GM, El Zayat EM, Al Anazi FK. Development of modified in situ gelling oral liquid sustained release formulation of dextromethorphan. Drug Dev Ind Pharm. 2012;38:971–8.
Narra K, Dhanalekshmi U, Rangaraj G, Raja D, Kumar CS, Reddy PN, et al. Effect of formulation variables on rifampicin loaded alginate beads. Iranian J Pharm Res. 2012;11:715–21.
Shah VP, Tsong Y, Sathe P, Liu JP. In-vitro dissolution profile comparison-statistics and analysis of the similarity factor, f2. Pharm Res. 1998;5:889–96.
Turner S, Federici C, Hite M, Fassihi R. Formulation development and human in-vitro/in-vivo correlation fora novel, monolithic controlled-release matrix system of high load and highly water-soluble drug niacin. Drug Dev Ind Pharm. 2004;30(8):797–807.
Korsmeyer RW, Gurny R, Doelker E, Buri P, Peppas NA. Mechanisms of solute release from porous hydrophilic polymers. Int J Pharm. 1983;15:25–35.
Acknowledgments
The authors appreciate the staff of the Department of Pharmaceutical Technology and Raw Materials Development for the support received during the course of this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Olayemi, O.J., Apeji, Y.E. & Isimi, C.Y. Formulation and Evaluation of Cyperus esculentus (Tiger Nut) Starch-Alginate Microbeads in the Oral Delivery of Ibuprofen. J Pharm Innov 17, 366–375 (2022). https://doi.org/10.1007/s12247-020-09509-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12247-020-09509-2