Abstract
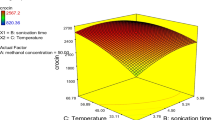
In this study, response surface methodology (RSM) was applied to optimize the extraction of picrocrocin, safranal, and crocin, three major ingredients of saffron. The process parameters included ethanol concentration (0–100 %), extraction time (2–7 h), and temperature (5–85 °C). The extracted compounds were measured spectrophotometrically at 257, 330, and 440 nm at which picrocrocin, safranal, and crocin had the maximum absorbance values, respectively. Four models including linear, linear squares, linear interactions, and full quadratic were fitted to the final data. As expected, the best model was the full quadratic with R 2 values of 83.91, 86.60, and 92.42 % for the picrocrocin, safranal, and crocin contents, respectively. Our results indicated that high temperatures, short times, and moderate concentrations of ethanol had the superlative impact on the extraction efficiencies of the compounds. According to the response surface analysis, ethanol concentration of 33.33 %, extraction time of 2.0 h, and temperature of 85.0 °C were found to be as the optimum conditions of the process under which the empirical amounts of E % 11cm λ max were 1,190.47 ± 154.45, 474.02 ± 95.00, and 2,311.68 ± 57.37 for the picrocrocin, safranal, and crocin contents, respectively. While the theoretical values for the same responses were 1,237.27, 652.08, and 2,821.23.










Similar content being viewed by others
References
Baş D, Boyacı İH (2007a) Modeling and optimization I: usability of response surface methodology. J Food Eng 78(3):836–845
Baş D, Boyacı İH (2007b) Modeling and optimization II: comparison of estimation capabilities of response surface methodology with artificial neural networks in a biochemical reaction. J Food Eng 78(3):846–854
Bergoin-Lefort M, Raynaud C, Vilarem G, Talou T (2006) Influence of dehydration on key odour compounds of saffron. Dev Food Sci 43:513–516
Bezerra MA, Santelli RE, Oliveira EP, Villar LS, Escaleira LA (2008) Response surface methodology (RSM) as a tool for optimization in analytical chemistry. Talanta 76(5):965–977
Bolandi M, Ghoddusi HB (2006) Flavour and colour changes during processing and storage of saffron (Crocus sativus L.). Dev Food Sci 43:323–326
Chen Y, Zhang H, Tian X, Zhao C, Cai L, Liu Y, Chen C (2008) Antioxidant potential of crocins and ethanol extracts of gardenia jasminoides ELLIS and crocus sativus L.: a relationship investigation between antioxidant activity and crocin contents. Food Chem 109(3):484–492
Granato, Daniel, Branco, Gabriel Favalli, & de Araْjo Calado, Verônica Maria. (2010). Experimental design and application of response surface methodology for proccess modelling and optimization: a review. Food Research International, In Press, Corrected Proof
ISO. (2002). ISO 3632/TS Saffron (Crocus sativus L.)-Part 2 Test methods. France
Kadkhodaee, R, & Hemmati-Kakhki, A. (2006). Ultrasonic extraction of active compounds from saffron. Paper presented at the II International Symposium on Saffron Biology and Technology 739
Kanakis CD, Daferera DJ, Tarantilis PA, Polissiou MG (2004) Qualitative determination of volatile compounds and quantitative evaluation of safranal and 4-hydroxy-2,6,6-trimethyl-1-cyclohexene-1-carboxaldehyde (HTCC) in greek saffron. J Agric Food Chem 52(14):4515–4521
Lage M, Cantrell CL (2009) Quantification of saffron (Crocus sativus L.) metabolites crocins, picrocrocin and safranal for quality determination of the spice grown under different environmental Moroccan conditions. Sci Hortic 121(3):366–373
Licón CC, Carmona M, Rubio R, Molina A, Berruga MI (2012) Preliminary study of saffron (Crocus sativus L. stigmas) color extraction in a dairy matrix. Dyes Pigm 92(3):1355–1360
Loskutov AV, Beninger CW, Hosfield GL, Sink KC (2000) Development of an improved procedure for extraction and quantitation of safranal in stigmas of Crocus sativus L. using high performance liquid chromatography. Food Chem 69(1):87–95
Lozano P, Castellar MR, Simancas MJ, Iborra JL (1999) A quantitative high-performance liquid chromatographic method to analyse commercial saffron (Crocus sativus L.) products. J Chromatogr A 830(2):477–483
Lozano P, Delgado D, Gómez D, Rubio M, Iborra JL (2000) A non-destructive method to determine the safranal content of saffron (Crocus sativus L.) by supercritical carbon dioxide extraction combined with high-performance liquid chromatography and gas chromatography. J Biochem Biophys Methods 43(1–3):367–378
Maggi L, Carmona M, Zalacain A, Kanakis CD, Anastasaki E, Tarantilis PA, Alonso GL (2010) Changes in saffron volatile profile according to its storage time. Food Res Int 43(5):1329–1334
Maggi L, Sanchez AM, Carmona M, Kanakis CD, Anastasaki E, Tarantilis PA, Alonso GL (2011) Rapid determination of safranal in the quality control of saffron spice (Crocus sativus L.). Food Chem 127(1):369–373
Melnyk JP, Wang S, Marcone MF (2010) Chemical and biological properties of the world's most expensive spice: saffron. Food Res Int 43(8):1981–1989
Minitab (2010). Minitab® Inc (Version 16.1.1)
Mohajeri SA, Hosseinzadeh H, Keyhanfar F, Aghamohammadian J (2010) Extraction of crocin from saffron (Crocus sativus) using molecularly imprinted polymer solid-phase extraction. J Sep Sci 33(15):2302–2309
Moraga AR, Nohales PF, Perez JA, Gomez-Gomez L (2004) Glucosylation of the saffron apocarotenoid crocetin by a glucosyltransferase isolated from Crocus sativus stigmas. Planta 219(6):955–966
Mousavi S, Tayarani N, Parsaee H (2010) Protective effect of saffron extract and crocin on reactive oxygen species-mediated high glucose-induced toxicity in PC12 cells. Cell Mol Neurobiol 30(2):185–191
Orfanou O, Tsimidou M (1996) Evaluation of the colouring strength of saffron spice by UV-Vis spectrometry. Food Chem 57(3):463–469
Orfanou, Olga, Tsimidou, Maria, & George, Charalambous (1995) Influence of selected additives on the stability of saffron pigments in aqueous extracts Developments in Food Science (Vol. Volume 37, pp. 881–894): Elsevier
Raina BL, Agarwal SG, Bhatia AK, Gaur GS (1996) Changes in pigments and volatiles of saffron (Crocus sativus L.) during processing and storage. J Sci Food Agric 71(1):27–32
Sánchez AM, Carmona M, del Campo CP, Alonso GL (2009) Solid-phase extraction for picrocrocin determination in the quality control of saffron spice (Crocus sativus L.). Food Chem 116(3):792–798
Selim K, Tsimidou M, Biliaderis CG (2000) Kinetic studies of degradation of saffron carotenoids encapsulated in amorphous polymer matrices. Food Chem 71(2):199–206
Sereshti H, Heidari R, Samadi S (2014) Determination of volatile components of saffron by optimised ultrasound-assisted extraction in tandem with dispersive liquid–liquid microextraction followed by gas chromatography–mass spectrometry. Food Chem 143:499–505
Serrano-Díaz J, Sánchez AM, Maggi L, Carmona M, Alonso GL (2011) Synergic effect of water-soluble components on the coloring strength of saffron spice. J Food Compos Anal 24(6):873–879
Verma R, Middha D (2010) Analysis of saffron (Crocus sativus L.) stigma components by LC–MS–MS. Chromatographia 71(1):117–123
Yang B, Liu X, Gao Y (2009) Extraction optimization of bioactive compounds (crocin, geniposide and total phenolic compounds) from Gardenia (Gardenia jasminoides Ellis) fruits with response surface methodology. Innov Food Sci Emerg Technol 10(4):610–615
Yilmaz A, Nyberg N, Mølgaard P, Asili J, Jaroszewski J (2010) H NMR metabolic fingerprinting of saffron extracts. Metabolomics 6(4):511–517
Zeng Y, Yan F, Tang L, Chen F (2003) Increased crocin production and induction frequency of stigma-like-structure from floral organs of Crocus sativus L. by precursor feeding. Plant Cell, Tissue Organ Cult 72(2):185–191
Zougagh M, Ríos A, Valcárcel M (2006) Determination of total safranal by in situ acid hydrolysis in supercritical fluid media: Application to the quality control of commercial saffron. Anal Chim Acta 578(2):117–121
Acknowledgement
Iran National Science Foundation and the Research Institute of Food Science and Technology should be acknowledged for their financial support. Great thanks is also dedicated to Mr. Ali Ghayur Kazemi and Mrs. Zahra Khodabakhshi for their technical assistance in performing the experiments.
Compliance with Ethics Requirements
ᅟ
Conflict of Interest
Messiah Sarfarazi declares that he has no conflict of interest.
Seid Mahdi Jafari declares that he has no conflict of interest.
Qadir Rajabzadeh declares that he has no conflict of interest.
We must include the following sentence to make sure that readers are aware that there are no ethical issues with human or animal subjects:
This article does not contain any studies with human or animal subjects.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sarfarazi, M., Jafari, S.M. & Rajabzadeh, G. Extraction Optimization of Saffron Nutraceuticals Through Response Surface Methodology. Food Anal. Methods 8, 2273–2285 (2015). https://doi.org/10.1007/s12161-014-9995-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-014-9995-3