Abstract
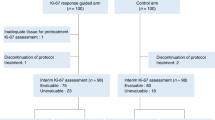
Determine if highly proliferative head and neck squamous cell carcinomas, assessed by pretreatment Ki-67 expression, respond more robustly to induction chemotherapy (IC) that is selectively toxic to cycling cells. Retrospective analysis of 59 patients treated with IC and chemoradiation. IC included either nab-paclitaxel, cisplatin, 5-FU and cetuximab (APF-C, n = 27) or docetaxel, cisplatin, 5-FU +/− cetuximab (TPF+/−C, n = 32). Ki-67 expression was assessed by immunohistochemistry. Tumor response (complete/partial/stable/progressive) at the primary site after two IC cycles was evaluated by visual examination in all patients. In the APF-C sub-group, tumor response (primary site and neck nodes) after two IC cycles was evaluated by computed tomography (CT) and fluorodeoxyglucose-positron emission tomography (FDG-PET)/CT. Ki-67 expression (median 66%, range: 16–97) did not differ across the tumor response categories assessed by visual examination (p = 0.95), CT (p = 0.30), or FDG-PET/CT (p = 0.65). Median decrease in summed SUVmax of measured lesions was 71.6% (range: 8.3–100%). The Pearson correlation coefficient between Ki-67 expression and the percent decrease in summed SUVmax was 0.48 (p = 0.02). Ki-67 expression was not different between those with or without a relapse (median: 60 and 71%, p = 0.10). In multivariate regression analysis (MVA) controlling for p16 positive oropharyngeal SCC status and smoking status, Ki-67 expression was not significantly associated with tumor response by visual examination (coefficient estimate −0.002, standard error 0.010, p = 0.84), CT (coefficient estimate −0.007, standard error 0.011, p = 0.54), FDG-PET/CT (coefficient estimate 0.006, standard error 0.008, p = 0.51), the percent decrease in summed SUVmax (coefficient estimate 0.389, standard error 0.222, p = 0.09), or relapse events (OR = 1.02(95%CI:0.99–1.05), p = 0.28). No significant relationships were found in MVA between pretreatment Ki-67 expression and tumor response to IC or to relapse.

Similar content being viewed by others
References
Parkin DM, Bray F, Ferlay J, Pisani P. Global cancer statistics. 2002. CA Cancer J Clin. 2005;55:74.
Forastiere A, Koch W, Trotti A, Sidransky D. Head and neck cancer. N Engl J Med. 2001;345:1890–900.
Hennessey PT, Westra WH, Califano JA. Human papillomavirus and head and neck squamous cell carcinoma: recent evidence and clinical implications. J Dent Res. 2009;88:300–6.
Adkins D, Ley J, Trinkaus K, Thorstad W, Lewis J Jr, Wildes T, et al. A phase 2 trial of induction nab-paclitaxel and cetuximab given with cisplatin and 5-fluorouracil followed by concurrent cisplatin and radiation for locally advanced squamous cell carcinoma of the head and neck. Cancer. 2013;119:766–73.
Baselga J, Norton L, Albanell J, Kim YM, Mendelsohn J. Recombinant humanized anti-HER2 antibody (Herceptin) enhances the antitumor activity of paclitaxel and doxorubicin against HER2/neu overexpressing human breast cancer xenografts. Cancer Res. 1998;58:2825–31.
Baselga J, Tripathy D, Mendelsohn J, Baughman S, Benz CC, Dantis L, et al. Phase II study of weekly intravenous recombinant humanized anti-p185HER2 monoclonal antibody in patients with HER2/neu-overexpressing metastatic breast cancer. J Clin Oncol. 1996;14:737–44.
Gravalos C, Jimeno A. HER2 in gastric cancer: a new prognostic factor and a novel therapeutic target. Ann Oncol. 2008;19:1523–9.
Tanner M, Hollmen M, Junttila TT, Kapanen AI, Tommola S, Soini Y, et al. Amplification of HER-2 in gastric carcinoma: association with Topoisomerase IIalpha gene amplification, intestinal type, poor prognosis and sensitivity to trastuzumab. Ann Oncol. 2005;16:273–8.
Fisher B, Costantino J, Redmond C, Poisson R, Bowman D, Couture J, et al. A randomized clinical trial evaluating tamoxifen in the treatment of patients with node-negative breast cancer who have estrogen-receptor-positive tumors. N Engl J Med. 1989;320:479–84.
Paez JG, Janne PA, Lee JC, Tracy S, Greulich H, Gabriel S, et al. EGFR mutations in lung cancer: correlation with clinical response to gefitinib therapy. Science. 2004;304:1497–500.
Amado RG, Wolf M, Peeters M, Van Cutsem E, Siena S, Freeman DJ, et al. Wild-type KRAS is required for panitumumab efficacy in patients with metastatic colorectal cancer. J Clin Oncol. 2008;26:1626–34.
Moroni M, Veronese S, Benvenuti S, Marrapese G, Sartore-Bianchi A, Di Nicolantonio F, et al. Gene copy number for epidermal growth factor receptor (EGFR) and clinical response to antiEGFR treatment in colorectal cancer: a cohort study. Lancet Oncol. 2005;6:279–86.
Bonner JA, Harari PM, Giralt J, Azarnia N, Shin DM, Cohen RB, et al. Radiotherapy plus cetuximab for squamous-cell carcinoma of the head and neck. N Engl J Med. 2006;354:567–78.
Vermorken JB, Mesia R, Rivera F, Remenar E, Kawecki A, Rottey S, et al. Platinum-based chemotherapy plus cetuximab in head and neck cancer. N Engl J Med. 2008;359:1116–27.
Licitra L, Mesia R, Rivera F, Remenar E, Hitt R, Erfan J, et al. Evaluation of EGFR gene copy number as a predictive biomarker for the efficacy of cetuximab in combination with chemotherapy in the first-line treatment of recurrent and/or metastatic squamous cell carcinoma of the head and neck: EXTREME study. Ann Oncol. 2011;22:1078–87.
Scholzen T, Gerdes J. The Ki-67 protein: from the known and the unknown. J Cell Physiol. 2000;182:311–22.
Silva SD, Agostini M, Nishimoto IN, Coletta RD, Alves FA, Lopes MA, et al. Expression of fatty acid synthase, ErbB2 and Ki-67 in head and neck squamous cell carcinoma. A clinicopathological study. Oral Oncol. 2004;40:688–96.
Pich A, Chiusa L, Navone R. Prognostic relevance of cell proliferation in head and neck tumors. Ann Oncol. 2004;15:1319–29.
Lavertu P, Adelstein DJ, Myles J, Secic M. P53 and Ki-67 as outcome predictors for advanced squamous cell cancers of the head and neck treated with chemoradiotherapy. Laryngoscope. 2001;111:1878–92.
Kropveld A, Slootweg PJ, Blankenstein MA, Terhaard CH, Hordijk GJ. Ki-67 and p53 in T2 laryngeal cancer. Laryngoscope. 1998;108:1548–52.
Couture C, Raybaud-Diogene H, Tetu B, Bairati I, Murry D, Allard J, et al. p53 and Ki-67 as markers of radioresistance in head and neck carcinoma. Cancer. 2002;94:713–22.
Homma A, Furuta Y, Oridate N, Nakano Y, Kohashi G, Yagi K, et al. Prognostic significance of clinical parameters and biological markers in patients with squamous cell carcinoma of the head and neck treated with concurrent chemoradiotherapy. Clin Cancer Res. 1999;5:801–6.
Petit T, Wilt M, Velten M, Millon R, Rodier JF, Borel C, et al. Comparative value of tumour grade, hormonal receptors, Ki-67, HER-2 and topoisomerase II alpha status as predictive markers in breast cancer patients treated with neoadjuvant anthracycline-based chemotherapy. Eur J Cancer. 2004;40:205–11.
Ellis MJ, Tao Y, Luo J, A’Hern R, Evans DB, Bhatnagar AS, et al. Outcome prediction for estrogen receptor-positive breast cancer based on postneoadjuvant endocrine therapy tumor characteristics. J Natl Cancer Inst. 2008;100:1380–8.
Oncotypedx. Cited 1/14/2016. http://www.oncotypedx.com/.
Prosigna. Cited 1/14/2016. http://prosigna.com.
Perez EA. Microtubule inhibitors: differentiating tubulin-inhibiting agents based on mechanisms of action, clinical activity, and resistance. Mol Cancer Ther. 2009;8:2086–95.
Yewale C, Baradia D, Vhora I, Patil S, Misra A. Epidermal growth factor receptor targeting in cancer: a review of trends and strategies. Biomaterials. 2013;34:8690–707.
Schell A, Ley J, Wu N, Trinkaus K, Wildes TM, Michel L, et al. Nab-paclitaxel-based compared to docetaxel-based induction chemotherapy regimens for locally advanced squamous cell carcinoma of the head and neck. Cancer Med. 2015;4:481–9.
Ensley JF, Jacobs JR, Weaver A, Kinzie J, Crissman J, Kish JA, et al. Correlation between response to cisplatinum-combination chemotherapy and subsequent radiotherapy in previously untreated patients with advanced squamous cell cancers of the head and neck. Cancer. 1984;54:811–4.
Forastiere AA, Goepfert H, Maor M, Pajak TF, Weber R, Morrison W, et al. Concurrent chemotherapy and radiotherapy for organ preservation in advanced laryngeal cancer. N Engl J Med. 2003;349:2091–8.
Therasse P, Arbuck SG, Eisenhauer EA, Wanders J, Kaplan RS, Rubinstein L, et al. New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst. 2000;92:205–16.
Young H, Baum R, Cremerius U, Herholz K, Hoekstra O, Lammertsma A, et al. Measurement of clinical and subclinical tumour response using [18 F]-fluorodeoxyglucose and positron emission tomography: review and 1999 EORTC recommendations. Eur J cancer. 1999;35:1773–82.
Scantlebury JB, Luo J, Thorstad WL, El-Mofty SK, Lewis JS Jr. Cyclin D1-a prognostic marker in oropharyngeal squamous cell carcinoma that is tightly associated with high-risk human papillomavirus status. Hum Pathol. 2013;44:1672–80.
Dowsett M, Smith IE, Ebbs SR, Dixon JM, Skene A, A’Hern R, et al. Prognostic value of Ki67 expression after short-term presurgical endocrine therapy for primary breast cancer. J Natl Cancer Inst. 2007;99:167–70.
Goetz MP, Suman VJ. Window-of-opportunity trials in the preoperative setting: insights into drug development for estrogen receptor–positive breast cancer. J Clin Oncol. 2016;34:1970–2.
Cohen EE, Karrison TG, Kocherginsky M, Mueller J, Egan R, Huang CH, et al. Phase III randomized trial of induction chemotherapy in patients with N2 or N3 locally advanced head and neck cancer. J Clin Oncol. 2014;32(25):2735–43.
Haddad R, O’Neill A, Rabinowits G, Tishler R, Khuri F, Adkins D, et al. Induction chemotherapy followed by concurrent chemoradiotherapy (sequential chemoradiotherapy) versus concurrent chemoradiotherapy alone in locally advanced head and neck cancer (PARADIGM): a randomised phase 3 trial. Lancet Oncol. 2013;14(3):257–64.
Funding
We thank the Alvin J. Siteman Cancer Center at Washington University School of Medicine and Barnes-Jewish Hospital in St. Louis, Missouri for the use of the Clinical Trials Core, which provided protocol development and clinical trial support and for use of the Imaging and Response Assessment Core. The Siteman Cancer Center is supported in part by NCI Cancer Center Support Grant #P30 CA91842, (Eberlein, PI).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. Adkins has received Research Funding from Celgene. All other authors have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study, as part of a registry protocol.
Additional information
Presented in abstract form at the 2015 Annual Meeting of the American Society of Clinical Oncology.
Rights and permissions
About this article
Cite this article
Chatzkel, J., Lewis, J.S., Ley, J.C. et al. Correlation of Ki-67 Proliferative Antigen Expression and Tumor Response to Induction Chemotherapy Containing Cell Cycle-Specific Agents in Head and Neck Squamous Cell Carcinoma. Head and Neck Pathol 11, 338–345 (2017). https://doi.org/10.1007/s12105-016-0775-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12105-016-0775-9