Abstract
Objective
High burden of rotavirus associated diarrhea has been documented among Indian children. The phased introduction of an indigenous rotavirus vaccine ‘ROTAVAC’ in India's national immunization programme began in 2017. Phase-III trial showed the vaccine to have a low-intussusception-risk profile. However, evaluation of post-licensure trends of intussusception is necessary to assess potential vaccine-associated intussusception risk. This study's objective was to describe the epidemiology of intussusception hospitalizations in children under two years of age in Tamil Nadu and Puducherry following ROTAVAC introduction.
Methods
A cross-sectional surveillance was established in six hospitals in Tamil Nadu and Puducherry. Children under two years of age with intussusception fulfilling Brighton Collaboration’s criteria for level 1 diagnostic certainty were enrolled. Patient and disease characteristics were captured using a standardized questionnaire. Descriptive and inferential statistical analyses were performed using Stata Version 13.
Results
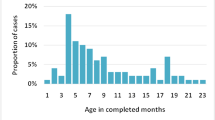
Overall, 287 cases were enrolled and had a median age of seven months. Frequently presenting symptoms were vomiting (78%), abdominal pain (76%), and blood in stool (71%). Abdominal ultrasonography or radiography confirmed diagnosis in 65% of cases and managed by nonoperative measures. Remaining 35% of cases were diagnosed and managed with surgery. Over 98% of the cases had positive treatment outcomes. Age less than five months (OR = 4.36), and hospitalization at a state government health facility (OR = 5.01) were significant predictors for children to receive surgical management.
Conclusions
The present study documents the epidemiology of intussusceptions immediately after the rollout of rotavirus vaccine in Tamil Nadu and Puducherry. No appreciable increase in intussusception hospitalizations was seen in the study hospitals after vaccine introduction.



Similar content being viewed by others
References
World Health Organization. Vaccines and Biologicals: Acute intussusception in infants and children: incidence, clinical representation and management: a global perspective. Available at: https://vaccine-safety-training.org/tl_files/vs/pdf/acute-intussusception-infants-children.pdf. Accessed 20th Jan 2020.
Stringer MD, Pablot SM, Brereton RJ. Paediatric intussusception. Br J Surg. 1992;79:867–76.
Clark A, Tate J, Parashar U, et al. Mortality reduction benefits and intussusception risks of rotavirus vaccination in 135 low-income and middle-income countries: a modelling analysis of current and alternative schedules. Lancet Glob Health. 2019;7:e1541-52.
Bahl R, Saxena M, Bhandari N, et al. Delhi Intussusception Study Hospital Group. Population-based incidence of intussusception and a case-control study to examine the association of intussusception with natural rotavirus infection among Indian children. J Infect Dis. 2009;200:S277–81.
Gupta M, Kanojia R, Singha R, et al. Intussusception rate among under-five-children before introduction of rotavirus vaccine in North India. J Trop Pediatr. 2018;64:326–35.
World Health Organization. Rotavirus. Available at: https://www.who.int/immunization/diseases/rotavirus/en/. Accessed 11th Jan 2020.
World Health Organization. Rotavirus vaccines WHO position paper: January 2013 – recommendations. Vaccine. 2013;31(52):6170–1 Available at: https://www.who.int/wer/2013/wer8805.pdf?ua=1. Accessed 11th Jan 2020.
Kang G, Desai R, Arora R, et al. Diversity of circulating rotavirus strains in children hospitalized with diarrhea in India, 2005–2009. Vaccine. 2013;31:2879–83.
Kumar CP, Venkatasubramanian S, Kang G, et al. Profile and trends of rotavirus gastroenteritis in under 5 children in India, 2012–2014, preliminary report of the Indian national rotavirus surveillance network. Indian Pediatr. 2016;53:619–22.
Shri J P Nadda launches Rotavirus vaccine as part of Universal Immunization Programme; terms it a "historic moment". Available at: http://pib.nic.in/newsite/PrintRelease.aspx?relid=138342. Accessed 11th Jan 2020.
Shri JP Nadda launches expansion of rotavirus vaccine under universal immunization Programme. http://pib.nic.in/newsite/PrintRelease.aspx?relid=158549. Accessed 11th Jan 2020.
Reddy S, Nair NP, Giri S, et al. Indian Intussusception Surveillance Network. Safety monitoring of ROTAVAC vaccine and etiological investigation of intussusception in India: study protocol. BMC Public Health. 2018;18:898.
Bines JE, Kohl KS, Forster J, et al. Brighton Collaboration Intussusception Working Group. Acute intussusception in infants and children as an adverse event following immunization: case definition and guidelines of data collection, analysis, and presentation. Vaccine. 2004;22:569–74.
Singh JV, Kamath V, Shetty R, et al. Retrospective surveillance for intussusception in children aged less than five years at two tertiary care centers in India. Vaccine. 2014;32:A95-8.
Bhowmick K, Kang G, Bose A, et al. Retrospective surveillance for intussusception in children aged less than five years in a South Indian tertiary-care hospital. J Health PopulNutr. 2009;27:660–5.
Mehendale S, Kumar CPG, Venkatasubramanian S, Prasanna T. Intussusception in children aged less than five years. Indian J Pediatr. 2016;83:1087–92.
Srinivasan R, Girish Kumar CP, Naaraayan SA, et al. Intussusception hospitalizations before rotavirus vaccine introduction: retrospective data from two referral hospitals in Tamil Nadu, India. Vaccine. 2018;36:7820–5.
Simon NM, Joseph J, Philip RR, Sukumaran TU, Philip R. Intussusception: single center experience of 10 years. Indian Pediatr. 2019;56:29–32.
Jiang J, Jiang B, Parashar U, Nguyen T, Bines J, Patel MM. Childhood intussusception: a literature review. PLoSOne. 2013;8:e68482.
National Immunization Schedule (NIS) for Infants, Children and Pregnant Women. Ministry of Health and Family Welfare, Government of India. Available at: https://mohfw.gov.in/sites/default/files/245453521061489663873.pdf. Accessed 14th Jan 2020.
Acknowledgements
The authors are thankful to the participants, parents/guardians, and surveillance staff at each of the study sites for their contributions to this study
Funding
This study was supported by a grant from Bill and Melinda Gates Foundation to the Translational Health Science and Technology Institute (OPP1165083) and the CDC Foundation, Atlanta with, the Christian Medical College (CMC), Vellore as sub-awardee.
Author information
Authors and Affiliations
Contributions
Analysis of data – NSR, CPGK, SK, SS, NS, VT; Interpretation of data – NSR, SK, CPGK, BS, NS, BHK, MK, PR, RM; Drafting of the article – CPGK, AS, PD, JMK; Critically revising drafts of the article – SK, CPGK, BS, NS, RG; Acquisition of data and final approval of submitted version – All authors. Dr. Gagandeep Kang, The Wellcome Trust Research Laboratory, Division of Gastrointestinal Sciences, Christian Medical College, Vellore, Tamil Nadu will act as guarantor for this paper.
Corresponding author
Ethics declarations
Ethics Approval
Ethical clearance has been obtained by each of the participating institutes from their respective ethical clearance committees/institute review boards.
Conflict of Interest
None.
Consent to participate and Consent for publication
Consent for participation and for publication and sharing of data have been obtained from the guardians of each of the participants (cases and controls) prior to enrolment in the study by each of the participating institutes
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kumar, C.P.G., N, S.R., Subramanian, S. et al. Epidemiology of Intussusception Hospitalizations in Children Under 2 Years of Age Post Rotavirus Vaccine Introduction in Tamil Nadu and Puducherry, India. Indian J Pediatr 88 (Suppl 1), 124–130 (2021). https://doi.org/10.1007/s12098-020-03597-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12098-020-03597-1