Abstract
Objectives
To estimate the association between serum bisphenol A and premature thelarche in female infants aged 4-mo to 2-y.
Methods
A total of 251 female infants (aged 4 mo to 2 y) with premature thelarche and 33 healthy age-matched control subjects were analyzed. All participants underwent physical examination and serum bisphenol A was measured by ultra-performance liquid chromatography tandem mass spectrometry.
Results
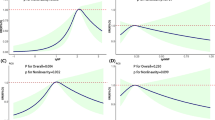
Serum bisphenol A concentration in the premature thelarche group (3.48 ng/ml, 95%CI: 0.09-140.26) was significantly higher than that in the control group (1.70 ng/ml, 95%CI: 0.06–51.78) (p = 0.039). There was no correlation between age and serum bisphenol A (BPA) level. Univariate logistic regression analysis showed that serum BPA concentration positively associated with premature thelarche, and the effect of BPA fell down as the age grew.
Conclusions
This hospital-based study implied that there is an association between serum BPA concentrations and premature thelarche. Additionally, serum BPA levels were markedly higher in infants aged 4-mo to 2-y-old, raising a concern for public health authorities.

Similar content being viewed by others
References
Geens T, Aerts D, Berthot C, Bourguignon JP, Goeyens L, Lecomte P, et al. A review of dietary and non-dietary exposure to bisphenol-A. Food Chem Toxicol. 2012;50:3725–40.
Huang YQ, Wong CK, Zheng JS, Bouwman H, Barra R, Wahlstrom B, et al. Bisphenol A (BPA) in China: a review of sources, environmental levels, and potential human health impacts. Environ Int. 2012;42:91–9.
Liao C, Kannan K. A survey of bisphenol A and other bisphenol analogues in foodstuffs from nine cities in China. Food Addit Contam Part A Chem Anal Control Expo Risk Assess. 2014;31:319–29.
Volkel W, Kiranoglu M, Fromme H. Determination of free and total bisphenol A in urine of infants. Environ Res. 2011;111:143–8.
von Goetz N, Wormuth M, Scheringer M, Hungerbuhler K. Bisphenol a: how the most relevant exposure sources contribute to total consumer exposure. Risk Anal. 2010;30:473–87.
Wang HX, Zhou Y, Tang CX, Wu JG, Chen Y, Jiang QW. Association between bisphenol A exposure and body mass index in Chinese school children: a cross-sectional study. Environ Health. 2012;11:79.
Trasande L, Attina TM, Blustein J. Association between urinary bisphenol A concentration and obesity prevalence in children and adolescents. JAMA. 2012;308:1113–21.
Trasande L, Attina TM, Trachtman H. Bisphenol A exposure is associated with low-grade urinary albumin excretion in children of the United States. Kidney Int. 2013;83:741–8.
Braun JM, Kalkbrenner AE, Calafat AM, Yolton K, Ye X, Dietrich KN, et al. Impact of early-life bisphenol A exposure on behavior and executive function in children. Pediatrics. 2011;128:873–82.
Donohue KM, Miller RL, Perzanowski MS, Just AC, Hoepner LA, Arunajadai S, et al. Prenatal and postnatal bisphenol A exposure and asthma development among inner-city children. J Allergy Clin Immunol. 2013;131:736–42.
Harley KG, Aguilar Schall R, Chevrier J, Tyler K, Aguirre H, Bradman A, et al. Prenatal and postnatal bisphenol A exposure and body mass index in childhood in the CHAMACOS cohort. Environ Health Perspect. 2013;121:514–20.
Mielke H, Gundert-Remy U. Bisphenol A levels in blood depend on age and exposure. Toxicol Lett. 2009;190:32–40.
Edginton AN, Ritter L. Predicting plasma concentrations of bisphenol A in children younger than 2 years of age after typical feeding schedules, using a physiologically based toxicokinetic model. Environ Health Perspect. 2009;117:645–52.
Diamantopoulos S, Bao Y. Gynecomastia and premature thelarche: a guide for practitioners. Pediatr Rev. 2007;28:e57–68.
Larriuz-Serrano MC, Perez-Cardona CM, Ramos-Valencia G, Bourdony CJ. Natural history and incidence of premature thelarche in Puerto Rican girls aged 6 months to 8 years diagnosed between 1990 and 1995. P R Health Sci J. 2001;20:13–8.
Marshall WA, Tanner JM. Variations in pattern of pubertal changes in girls. Arch Dis Child. 1969;44:291–303.
Hornung RW, Reed LD. Estimation of average concentration in the presence of nondetectable values. Appl Occup Environ Hyg. 1990;5:46–51.
Ye X, Zhou X, Wong LY, Calafat AM. Concentrations of bisphenol A and seven other phenols in pooled sera from 3–11 year old children: 2001–2002. National health and nutrition examination survey. Environ Sci Technol. 2012;46:12664–71.
Taylor JA, Vom Saal FS, Welshons WV, Drury B, Rottinghaus G, Hunt PA, et al. Similarity of bisphenol A pharmacokinetics in rhesus monkeys and mice: relevance for human exposure. Environ Health Perspect. 2011;119:422–30.
WANG L, HE J-b, LI F-h, Liu C. A serological survey of Bisphenol A in Shenzhen City. Pract Prev Med. 2005;12:241–2.
Zhou J, Zhang Q, Han K, Zhang L, Zhao F. Determination of bisphenol A (BPA) and diethylstilbestrol (DES) in milk powder for the middle and senior age by RP-HPLC. Sci Technol Food Ind. 2007;28:233–4.
Xuan D, Chen J. Determination of BPA in nursing bottle by solid phase extraction—high performance liquid chromatography. Mod Prev Med. 2008;35:4663–5.
Menon PS, Vijayakumar M. Precocious puberty–perspectives on diagnosis and management. Indian J Pediatr. 2014;81:76–83.
Chou YY, Huang PC, Lee CC, Wu MH, Lin SJ. Phthalate exposure in girls during early puberty. J Pediatr Endocrinol Metab. 2009;22:69–77.
Chapin RE, Adams J, Boekelheide K, Gray Jr LE, Hayward SW, Lees PS, et al. NTP-CERHR expert panel report on the reproductive and developmental toxicity of bisphenol A. Birth Defects Res B Dev Reprod Toxicol. 2008;83:157–395.
Wolff MS, Teitelbaum SL, Pinney SM, Windham G, Liao L, Biro F, et al. Investigation of relationships between urinary biomarkers of phytoestrogens, phthalates, and phenols and pubertal stages in girls. Environ Health Perspect. 2010;118:1039–46.
Wolff MS, Britton JA, Boguski L, Hochman S, Maloney N, Serra N, et al. Environmental exposures and puberty in inner-city girls. Environ Res. 2008;107:393–400.
Savchuk I, Soder O, Svechnikov K. Mouse leydig cells with different androgen production potential are resistant to estrogenic stimuli but responsive to bisphenol a which attenuates testosterone metabolism. PLoS ONE. 2013;8:e71722.
Losa-Ward SM, Todd KL, McCaffrey KA, Tsutsui K, Patisaul HB. Disrupted organization of RFamide pathways in the hypothalamus is associated with advanced puberty in female rats neonatally exposed to bisphenol A. Biol Reprod. 2012;87:28.
Acknowledgments
The authors would like to thank the children and their parents who participated in the study.
Contributions
LHC, YLF, LL: Conceived and designed the research; JRS, WQC, XZC: Collected the data; LHC, YLF: Analyzed the data; YLF: Act as guarantor for this paper; LHC, JRS, YLF: Wrote the paper.
Conflict of Interest
None.
Source of Funding
The Medical Science and Technology Program of Zhejiang province (2012RCA039) funded the study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, Lh., Shi, Jr., Fang, Yl. et al. Serum Bisphenol A Concentration and Premature Thelarche in Female Infants Aged 4-month to 2-year. Indian J Pediatr 82, 221–224 (2015). https://doi.org/10.1007/s12098-014-1548-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12098-014-1548-7