Abstract
Purpose
To identify the relevant factors affecting the prognosis and survival time of colon cancer and construct a survival prediction model.
Methods
Data on postoperative stage I–III colon cancer patients were obtained from the Surveillance, Epidemiology, and End Results database. We used R project to analyze the data. Univariate and multivariate Cox regression analyses were performed for independent factors correlated with overall survival from colon cancer. The C-index was used to screen the factors that had the greatest influence in overall survival after surgery in colon cancer patients. Receiver operating characteristic (ROC) curve was made according to the Risk score and calculated to validate the predictive accuracy of the model. In addition, we used decision curve analysis (DCA) to evaluate the clinical benefits and utility of the nomogram. We created a model survival curve to determine the difference in prognosis between patients in the low-risk group and those in the high-risk group.
Results
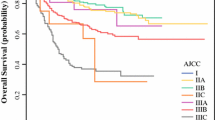
Univariate and multifactor COX analyses showed that the race, Grade, tumor size, N-stage and T-stage were independent risk factors affecting survival time of patients. The analysis of ROC and DCA showed the nomogram prediction model constructed based on the above indicators has good predictive effects.
Conclusion
Overall, the nomogram constructed in this study has good predictive effects. It can provide a reference for future clinicians to evaluate the prognosis of colon cancer patients.





Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424.
Stoffel EM, Murphy CC. Epidemiology and mechanisms of the increasing incidence of colon and rectal cancers in young adults. Gastroenterology. 2020;158(2):341–53.
Siegel RL, Miller KD, Goding Sauer A, Fedewa SA, Butterly LF, Anderson JC, et al. Colorectal cancer statistics, 2020. CA Cancer J Clin. 2020;70(3):145–64.
Collins GS, Reitsma JB, Altman DG, Moons KG. Transparent Reporting of a multivariable prediction model for Individual Prognosis Or Diagnosis (TRIPOD): the TRIPOD Statement. Br J Surg. 2015;102(3):148–58.
Wang Z, Wang Y, Yang Y, Luo Y, Liu J, Xu Y, et al. A competing-risk nomogram to predict cause-specific death in elderly patients with colorectal cancer after surgery (especially for colon cancer). World J Surg Oncol. 2020;18(1):30.
Wu Q, Wang WJ, Huang YQ, Fang SY, Guan YJ. Nomograms for estimating survival in patients with liver-only colorectal metastases: a retrospective study. Int J Surg. 2018;60:1–8.
Guan X, Ma CX, Quan JC, Zhao ZX, Chen HP, Sun P, et al. A prognostic index model to individually predict clinical outcomes for colorectal cancer with synchronous bone metastasis. J Cancer. 2020;11(15):4366–72.
Wang S, Yang L, Ci B, Maclean M, Gerber DE, Xiao G, et al. Development and validation of a nomogram prognostic model for SCLC patients. J Thorac Oncol. 2018;13(9):1338–48.
Chen S, Lai Y, He Z, Li J, He X, Shen R, et al. Establishment and validation of a predictive nomogram model for non-small cell lung cancer patients with chronic hepatitis B viral infection. J Transl Med. 2018;16(1):116.
Tang LQ, Li CF, Li J, Chen WH, Chen QY, Yuan LX, et al. Establishment and validation of prognostic nomograms for endemic nasopharyngeal carcinoma. J Natl Cancer Inst. 2015;108(1):djv291.
Rex DK, Boland CR, Dominitz JA, Giardiello FM, Johnson DA, Kaltenbach T, et al. Colorectal cancer screening: recommendations for physicians and patients from the U.S. Multi-Society Task Force on Colorectal Cancer. Am J Gastroenterol. 2017;112(7):1016–30.
Brenner H, Stock C, Hoffmeister M. Effect of screening sigmoidoscopy and screening colonoscopy on colorectal cancer incidence and mortality: systematic review and meta-analysis of randomised controlled trials and observational studies. BMJ. 2014;348: g2467.
Dai W, Mo S, Xiang W, Han L, Li Q, Wang R, et al. The critical role of tumor size in predicting prognosis for T1 Colon cancer. Oncologist. 2020;25(3):244–51.
Saha S, Shaik M, Johnston G, Saha SK, Berbiglia L, Hicks M, et al. Tumor size predicts long-term survival in colon cancer: an analysis of the National Cancer Data Base. Am J Surg. 2015;209(3):570–4.
Dai W, Li Y, Meng X, Cai S, Li Q, Cai G. Does tumor size have its prognostic role in colorectal cancer? Re-evaluating its value in colorectal adenocarcinoma with different macroscopic growth pattern. Int J Surg. 2017;45:105–12.
Yan Q, Zhang K, Guo K, Liu S, Wasan HS, Jin H, et al. Value of tumor size as a prognostic factor in metastatic colorectal cancer patients after chemotherapy: a population-based study. Future Oncol. 2019;15(15):1745–58.
Santullo F, Biondi A, Cananzi FCM, Fico V, Tirelli F, Ricci R, et al. Tumor size as a prognostic factor in patients with stage IIa colon cancer. Am J Surg. 2018;215(1):71–7.
Delattre JF, Selce Oguz Erdogan A, Cohen R, Shi Q, Emile JF, Taieb J, et al. A comprehensive overview of tumour deposits in colorectal cancer: Towards a next TNM classification. Cancer Treat Rev. 2022;103:102325.
Lea D, Håland S, Hagland HR, Søreide K. Accuracy of TNM staging in colorectal cancer: a review of current culprits, the modern role of morphology and stepping-stones for improvements in the molecular era. Scand J Gastroenterol. 2014;49(10):1153–63.
Chen YG, Liu YL, Liu Z, Ge X, Wang XS. Comparative study of oncologic outcomes after multivisceral resection for stage IIC colorectal cancer between inflammatory and malignant adhesion. Zhonghua Wei Chang Wai Ke Za Zhi. 2011;14(4):267–70.
Funding
This study was funded by Basic research funding for higher education institutions in Heilongjiang Province in 2020 (2020-KYYWF-0032).
Author information
Authors and Affiliations
Contributions
Contributions: (I) FZ and YS contributed to Conception and design; (II) FZ was involved in administrative support; (III) FZ, YS, KN contributed to provision of study materials or patients; (IV) FZ, JZ, and CZ were involved in collection and assembly of data; (V) FZ, JG contributed to data analysis and interpretation; (VI) All authors were involved in manuscript writing and final approval of manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The study was approved by Ethical Committee of The Second Affiliated Hospital of Qiqihaer Medical University and conducted in accordance with the ethical standards.
Informed consent
Subjects signed the informed consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhao, F., Sun, Y., Zhao, J. et al. Clinical characteristics and prognosis analysis of postoperative patients with stage I–III colon cancer based on SEER database. Clin Transl Oncol 26, 225–230 (2024). https://doi.org/10.1007/s12094-023-03239-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-023-03239-w