Abstract
Background
Immune checkpoint inhibitors (ICIs) plus chemotherapy (CT) have strikingly expanded the therapeutic landscape for advanced non-small cell lung cancer (NSCLC), but little is known about which is superior. We performed a meta-analysis that compared the efficacy and safety of PD-1 inhibitor + CT with PD-L1 inhibitor + CT.
Methods
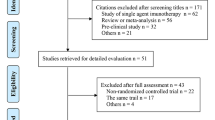
PubMed, Embase, Web of Science, Cochrane Library, and major international scientific meetings were searched for relevant randomized controlled trials (RCTs), and the indirect analysis was performed for PD-1 + CT vs PD-L1 + CT. The outcomes included progression-free survival (PFS), overall survival (OS), objective response rate (ORR) and treatment-related adverse events (TRAEs).
Results
8 phase III RCTs with 4253 patients comparing PD-1/PD-L1 + CT in NSCLC were included. The PD-1 + CT led to notably longer OS most in low/negative expression of PD-L1 for NSCLC patients compared with PD-L1 + CT. In terms of Grade 3–5 TRAEs, the results showed that PD-1 + CT and PD-L1 + CT exclusively increased the risk of adverse incidence than CT alone, especially for PD-L1 + CT (p < 0.00001). For subgroups including female, young patients, patients with nonsmoker, and EGFR/ALK wild-type, PD-1 + CT was associated with prolonged OS (p < 0.05). Meanwhile, for no liver metastasis of NSCLC patients, we found obviously OS advantage for patients treated with PD-1 + CT compared to PD-L1 + CT.
Conclusions
ICIs + CT seemed to be more effective first-line regimen and PD-1 + CT could be recommended as the first-rank therapy for advanced NSCLC patients with low/negative expression of PD-L1. However, we should be particularly vigilant about the occurrence of the Grade 3–5 TRAEs.




Similar content being viewed by others
References
Siegel RL, Miller KD. Jemal A (2018) cancer statistics. Ca Cancer J Clin. 2018;68:7–30. https://doi.org/10.3322/caac.21442.
Fitzmaurice C, Akinyemiju TF, Al Lami FH, et al. Global, regional, and national cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life-years for 29 cancer groups, 1990 to 2016: a systematic analysis for the global burden of disease study. JAMA Oncol. 2018;4:1553–688. https://doi.org/10.1001/jamaoncol.2018.2706.
Chen Y, Peng X, Zhou Y, Xia K, Zhuang W. Comparing the benefits of chemoradiotherapy and chemotherapy for resectable stage III A/N2 non-small cell lung cancer: a meta-analysis. World J Surg Oncol. 2018;16:8. https://doi.org/10.1186/s12957-018-1313-x.
Herbst RS, Morgensztern D, Boshoff C. The biology and management of non-small cell lung cancer. Nature. 2018;553:446–54. https://doi.org/10.1038/nature25183.
Reck M, Rabe KF. Precision diagnosis and treatment for advanced non–small-cell lung cancer. N Engl J Med. 2017;377:849–61. https://doi.org/10.1056/NEJMra1703413.
Laurie S, Solomon B, Seymour L, et al. Randomised, double-blind trial of carboplatin and paclitaxel with daily oral cediranib or placebo in patients with advanced non-small cell lung cancer: NCIC Clinical Trials Group study BR29. Eur J Cancer. 2014;50:706–12. https://doi.org/10.1016/j.ejca.2013.11.032.
Paz-Ares LG, Biesma B, Heigener D, et al. Phase III, randomized, double-blind, placebo-controlled trial of gemcitabine/cisplatin alone or with sorafenib for the first-line treatment of advanced, nonsquamous non–small-cell lung cancer. J Clin Oncol. 2012;30:3084–92. https://doi.org/10.1200/JCO.2011.39.7646.
Hall RD, Le TM, Haggstrom DE, Gentzler RD. Angiogenesis inhibition as a therapeutic strategy in non-small cell lung cancer (NSCLC). Transl Lung Cancer Res. 2015;4:515. https://doi.org/10.3978/j.issn.2218-6751.2015.06.09.
Garon EB, Ciuleanu T-E, Arrieta O, et al. Ramucirumab plus docetaxel versus placebo plus docetaxel for second-line treatment of stage IV non-small-cell lung cancer after disease progression on platinum-based therapy (REVEL): a multicentre, double-blind, randomised phase 3 trial. Lancet. 2014;384:665–73. https://doi.org/10.1016/S0140-6736(14)60845-X.
Reck M, Kaiser R, Mellemgaard A, et al. Docetaxel plus nintedanib versus docetaxel plus placebo in patients with previously treated non-small-cell lung cancer (LUME-Lung 1): a phase 3, double-blind, randomised controlled trial. Lancet Oncol. 2014;15:143–55. https://doi.org/10.1016/S1470-2045(13)70586-2.
Reck M, Popat S, Reinmuth N, De Ruysscher D, Kerr K, Peters S. Metastatic non-small-cell lung cancer (NSCLC): ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2014;25:iii27-iii39.
Borghaei H, Paz-Ares L, Horn L, et al. Nivolumab versus docetaxel in advanced nonsquamous non–small-cell lung cancer. N Engl J Med. 2015;373:1627–39. https://doi.org/10.1056/NEJMoa1507643.
Brahmer J, Reckamp KL, Baas P, et al. Nivolumab versus docetaxel in advanced squamous-cell non–small-cell lung cancer. N Engl J Med. 2015;373:123–35. https://doi.org/10.1056/NEJMoa1504627.
Gandhi L, Rodríguez-Abreu D, Gadgeel S, et al. Pembrolizumab plus chemotherapy in metastatic non–small-cell lung cancer. N Engl J Med. 2018;378:2078–92. https://doi.org/10.1056/NEJMoa1801005.
Antonia SJ, Villegas A, Daniel D, et al. Durvalumab after chemoradiotherapy in stage III non–small-cell lung cancer. N Engl J Med. 2017;377:1919–29. https://doi.org/10.1056/NEJMoa1709937.
Wei SC, Levine JH, Cogdill AP, et al. Distinct cellular mechanisms underlie anti-CTLA-4 and anti-PD-1 checkpoint blockade. Cell. 2017;170(1120–1133):e1117. https://doi.org/10.1016/j.cell.2017.07.024.
Wang C, Qiao W, Jiang Y, et al. The landscape of immune checkpoint inhibitor plus chemotherapy versus immunotherapy for advanced non-small-cell lung cancer: a systematic review and meta-analysis. J Cell Physiol. 2019. https://doi.org/10.1002/jcp.29371.
Zhou Y, Lin Z, Zhang X, et al. First-line treatment for patients with advanced non-small cell lung carcinoma and high PD-L1 expression: pembrolizumab or pembrolizumab plus chemotherapy. J Immunother Cancer. 2019;7:120. https://doi.org/10.1186/s40425-019-0600-6.
Socinski MA, Jotte RM, Cappuzzo F, et al. Atezolizumab for first-line treatment of metastatic nonsquamousNSCLC. N Engl J Med. 2018;378:2288–301. https://doi.org/10.1056/NEJMoa1716948.
Paz-Ares L, Luft A, Vicente D, et al. Pembrolizumab plus chemotherapy for squamous non–small-cell lung cancer. N Engl J Med. 2018;379:2040–51. https://doi.org/10.1056/NEJMoa1810865.
Zhang Y, Zhou H, Zhang L. Which is the optimal immunotherapy for advanced squamous non-small-cell lung cancer in combination with chemotherapy: anti-PD-1 or anti-PD-L1? J Immunother Cancer. 2018;6:135. https://doi.org/10.1186/s40425-018-0427-6.
Paz-Ares LG, Luft A, Tafreshi A, et al. Phase 3 study of carboplatin-paclitaxel/nab-paclitaxel (Chemo) with or without pembrolizumab (Pembro) for patients (Pts) with metastatic squamous (Sq) non-small cell lung cancer (NSCLC): American Society of Clinical Oncology, 2018.
Jotte RM, Cappuzzo F, Vynnychenko I, et al. IMpower131: Primary PFS and safety analysis of a randomized phase III study of atezolizumab+ carboplatin+ paclitaxel or nab-paclitaxel vs carboplatin+ nab-paclitaxel as 1L therapy in advanced squamous NSCLC. J Clin Oncol. 2018;36:LBA9000.
Barlesi F, Vansteenkiste J, Spigel D, et al. Avelumab versus docetaxel in patients with platinum-treated advanced non-small-cell lung cancer (JAVELIN Lung 200): an open-label, randomised, phase 3 study. Lancet Oncol. 2018;19:1468–79. https://doi.org/10.1016/S1470-2045(18)30673-9.
Langer CJ, Gadgeel SM, Borghaei H, et al. Carboplatin and pemetrexed with or without pembrolizumab for advanced, non-squamous non-small-cell lung cancer: a randomised, phase 2 cohort of the open-label KEYNOTE-021 study. Lancet Oncol. 2016;17:1497–508. https://doi.org/10.1016/S1470-2045(16)30498-3.
West H, McCleod M, Hussein M, et al. Atezolizumab in combination with carboplatin plus nab-paclitaxel chemotherapy compared with chemotherapy alone as first-line treatment for metastatic non-squamous non-small-cell lung cancer (IMpower130): a multicentre, randomised, open-label, phase 3 trial. Lancet Oncol. 2019;20:924–37. https://doi.org/10.1016/S1470-2045(19)30167-6.
Papadimitrakopoulou V, Cobo M, Bordoni R, Dubray-Longeras P, Szalai Z, Ursol G. IMpower132: PFS and safety results with 1L atezolizumab+ carboplatin/cisplatin+ pemetrexed in stage IV non-squamous NSCLC. J Thorac Oncol. 2018;13:S332–333.
Higgins JP, Altman DG, Gøtzsche PC, et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ. 2011;343:d5928. https://doi.org/10.1136/bmj.d5928.
Chen Y, Zhou Y, Tang L, et al. Immune-checkpoint inhibitors as the first line treatment of advanced non-small cell lung cancer: a meta-analysis of randomized controlled trials. J Cancer. 2019;10:6261. https://doi.org/10.7150/jca.34677.
Zhou Y, Chen C, Zhang X, et al. Immune-checkpoint inhibitor plus chemotherapy versus conventional chemotherapy for first-line treatment in advanced non-small cell lung carcinoma: a systematic review and meta-analysis. J Immunother Cancer. 2018;6:155. https://doi.org/10.1186/s40425-018-0477-9.
Ribas A, Wolchok JD. Cancer immunotherapy using checkpoint blockade. Science. 2018;359:1350–5. https://doi.org/10.1126/science.aar4060.
Zou W, Wolchok JD, Chen L. PD-L1 (B7–H1) and PD-1 pathway blockade for cancer therapy: mechanisms, response biomarkers, and combinations. Sci Transl Med. 2016;8:328. https://doi.org/10.1126/scitranslmed.aad7118.
Herbst RS, Baas P, Kim D-W, et al. Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): a randomised controlled trial. Lancet. 2016;387:1540–50. https://doi.org/10.1016/S0140-6736(15)01281-7.
Rittmeyer A, Barlesi F, Waterkamp D, et al. Atezolizumab versus docetaxel in patients with previously treated non-small-cell lung cancer (OAK): a phase 3, open-label, multicentre randomised controlled trial. Lancet. 2017;389:255–65. https://doi.org/10.1016/S0140-6736(16)32517-X.
Luo W, Wang Z, Tian P, Li W. Safety and tolerability of PD-1/PD-L1 inhibitors in the treatment of non-small cell lung cancer: a meta-analysis of randomized controlled trials. J Cancer Res Clin Oncol. 2018;144:1851–9. https://doi.org/10.1007/s00432-018-2707-4.
O'Kane GM, Labbé C, Doherty MK, Young K, Albaba H, Leighl NB. Monitoring and management of immune-related adverse events associated with programmed cell death protein-1 axis inhibitors in lung cancer. Oncologist. 2018;22:70. https://doi.org/10.1634/theoncologist.2016-0164.
Ma K, Lu Y, Jiang S, Tang J, Li X, Zhang Y. The relative risk and incidence of immune checkpoint inhibitors related pneumonitis in patients with advanced cancer: a meta-analysis. Front Pharmacol. 2018;9:1430. https://doi.org/10.3389/fphar.2018.01430.
Su Q, Zhang X, Shen X, Hou Y, Sun Z, Gao ZH. Risk of immune-related colitis with PD-1/PD-L1 inhibitors vs chemotherapy in solid tumors: systems assessment. J Cancer. 2018;9:1614. https://doi.org/10.7150/jca.24200.
El-Osta H, Jafri S. Predictors for clinical benefit of immune checkpoint inhibitors in advanced non-small-cell lung cancer: a meta-analysis. Immunotherapy. 2019;11:189–99. https://doi.org/10.2217/imt-2018-0086.
Conforti F, Pala L, Bagnardi V, et al. Cancer immunotherapy efficacy and patients' sex: a systematic review and meta-analysis. Lancet Oncol. 2018;19:737–46. https://doi.org/10.1016/S1470-2045(18)30261-4.
El-Osta HE, Mott FE, Burt BM, Wang DY, Sabichi AL. Predictors of benefits from frontline chemoimmunotherapy in stage IV non-small-cell lung cancer: a meta-analysis. OncoImmunology. 2019;8:e1665974. https://doi.org/10.1080/2162402X.2019.1665974.
Ninomiya K, Oze I, Kato Y, et al. Influence of age on the efficacy of immune checkpoint inhibitors in advanced cancers: a systematic review and meta-analysis. Acta Oncol. 2019;1:1–8. https://doi.org/10.1080/0284186X.2019.1695062.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (81372436), and Science and Technology Open Cooperation Project of Henan Province (182106000062).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared that no conflict interest exists.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
12094_2020_2502_MOESM1_ESM.tif
Figure S1 Forest plots of hazard ratios comparing OS between patients treated with PD-1 + CT or PD-L1 + CT according to PD-L1 expression. PD-1: anti-PD-1 inhibitors combined with chemotherapy; PD-L1: anti-PD-L1 inhibitors combined with chemotherapy; *PD-L1 high (TC3 or IC3). Low PD-L1 expression: PDL1 1-49% or TC1/2 (TC ≥1% and <50%) or IC1/2 (IC ≥1% and <10%). High PD-L1 expression: PD-L1 ≥ 50% or TC3 (TC≥50%) or IC3 (IC≥10%). TC: tumor cells. IC: tumor-infiltrating immune cells. (TIF 23054 kb)
12094_2020_2502_MOESM2_ESM.tif
Figure S2 Forest plots of hazard ratios comparing PFS between patients treated with PD-1 + CT or PD-L1 + CT according to PD-L1 expression. PD-1: anti-PD-1 inhibitors combined with chemotherapy; PD-L1: anti-PD-L1 inhibitors combined with chemotherapy; *PD-L1 high (TC3 or IC3). Low PD-L1 expression: PDL1 1-49% or TC1/2 (TC ≥1% and <50%) or IC1/2 (IC ≥1% and <10%). High PD-L1 expression: PD-L1 ≥ 50% or TC3 (TC≥50%) or IC3 (IC≥10%). TC: tumor cells. IC: tumor-infiltrating immune cells (TIF 23852 kb)
Rights and permissions
About this article
Cite this article
Yang, Y., Luo, H., Zheng, X.L. et al. The optimal immune checkpoint inhibitors combined with chemotherapy for advanced non-small-cell lung cancer: a systematic review and meta-analysis. Clin Transl Oncol 23, 1117–1127 (2021). https://doi.org/10.1007/s12094-020-02502-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-020-02502-8