Abstract
Objective
To delineate the association of postoperative complications with clinicopathologic factors and to identify risk factors for tumor recurrence and mortality after tumor resection in patients with colorectal cancer (CRC).
Methods
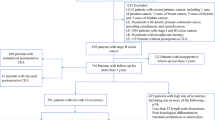
The clinical data of 1144 patients with CRC who underwent surgical intervention between 2003 and 2013 were retrieved. Correlations of postoperative complications with clinicopathologic factors were examined using univariate analysis. Risk factors for tumor recurrence and mortality of the patients after tumor resection were identified using multivariate Cox proportional hazards models. Time to relapse and overall survival were analyzed using log-rank test of Kaplan–Meier analysis.
Results
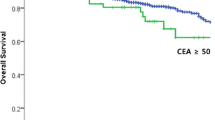
Blood carcinoembryonic antigen (CEA) significantly correlated with early symptoms, preoperative manifestations, and tumor pathology. Low differentiation grade of tumor increased the risk of recurrence after surgery in all patients with CRC. In the same cohort of patients, elevated blood CEA, low differentiation grade of tumor, laparotomy, smoking history, and TNM stage IV and III increased the mortality risk after tumor resection. In patients with advanced colon cancer, risk for postoperative mortality was increased by blood CEA, advanced tumor stage, and low tumor differentiation grade; while in those with advanced rectal cancer, blood CEA, pathologic type other than mucinous/adenocarcinoma, and laparotomy were identified as significant risk factors. In both groups of patients, postoperative chemotherapy significantly reduced the risk of mortality.
Conclusions
The present work has identified clinical factors increasing the risk of recurrence as well as mortality after surgery in more than 1,000 patients with CRC. Postoperative chemotherapy is associated with a significant reduction in the risk of mortality. All of these findings should provide insights into the better management of critically ill patients with CRC.




Similar content being viewed by others
References
Bonnington SN, Rutter MD. Surveillance of colonic polyps: are we getting it right? World J Gastroenterol. 2016;22(6):1925–34.
Vogelstein B, Fearon ER, Hamilton SR, Kern SE, Preisinger AC, Leppert M, et al. Genetic alterations during colorectal-tumor development. N Engl J Med. 1988;319(9):525–32.
Sameer AS, Parray FQ, Dar MA, Nissar S, Banday MZ, Rasool S, et al. Cyclin D1 G870A polymorphism and risk of colorectal cancer: a case control study. Mol Med Rep. 2013;7(3):811–5.
Sameer AS. Colorectal cancer: molecular mutations and polymorphisms. Front Oncol. 2013;3:114.
Lasry A, Zinger A, Ben-Neriah Y. Inflammatory networks underlying colorectal cancer. Nat Immunol. 2016;17(3):230–40.
Housseau F, Wu S, Wick EC, Fan H, Wu X, Llosa NJ, et al. Redundant innate and adaptive sources of IL-17 production drive colon tumorigenesis. Cancer Res. doi:10.1158/0008-5472.CAN-15-0749 (2016e-pub ahead of print Feb 15).
DeSantis CE, Lin CC, Mariotto AB, Siegel RL, Stein KD, Kramer JL, et al. Cancer treatment and survivorship statistics, 2014. CA Cancer J Clin. 2014;64(4):252–71.
Siegel R, Desantis C, Jemal A. Colorectal cancer statistics, 2014. CA Cancer J Clin. 2014;64(2):104–17.
Sweetser S, Jones A, Smyrk TC, Sinicrope FA. Sessile serrated polyps are precursors of colon carcinomas predominantly with deficient DNA mismatch repair. Clin Gastroenterol Hepatol. doi:10.1016/j.cgh.2016.01.021 (2016e-pub ahead of print Feb 16).
Sinicrope FA, Okamoto K, Kasi PM, Kawakami H. Molecular biomarkers in the personalized treatment of colorectal cancer. Clin Gastroenterol Hepatol. doi:10.1016/j.cgh.2016.02.008 (2016e-pub ahead of print Feb 9).
Ruibal Morell A. CEA serum levels in non-neoplastic disease. Int J Biol Mark. 1992;7(3):160–6.
Sorensen CG, Karlsson WK, Pommergaard HC, Burcharth J, Rosenberg J. The diagnostic accuracy of carcinoembryonic antigen to detect colorectal cancer recurrence—a systematic review. Int J Surg. 2016;25:134–44.
Basu A, Seth S, Chauhan AK, Bansal N, Arora K, Mahaur A. Comparative study of tumor markers in patients with colorectal carcinoma before and after chemotherapy. Ann Transl Med. 2016;4(4):71.
Ikeguchi M, Ashida K, Saito H. New prognostic indicator is useful for predicting the survival of patients with unresectable advanced colorectal cancer. Hepatogastroenterology. 2015;62(140):859–62.
Eren T, Burcu B, Tombalak E, Ozdemir T, Leblebici M, Ozemir IA, et al. Clinical significance of the glasgow prognostic score for survival after colorectal cancer surgery. J Gastrointest Surg. doi:10.1007/s11605-016-3114-2 (2016e-pub ahead of print Feb 29).
Kirchhoff P, Clavien PA, Hahnloser D. Complications in colorectal surgery: risk factors and preventive strategies. Patient Saf Surg. 2010;4(1):5.
Pecorelli N, Amodeo S, Frasson M, Vignali A, Zuliani W, Braga M. Ten-year outcomes following laparoscopic colorectal resection: results of a randomized controlled trial. Int J Colorectal Dis. doi:10.1007/s00384-016-2587-5 (2016e-pub ahead of print Apr 18).
Patankar SK, Larach SW, Ferrara A, Williamson PR, Gallagher JT, DeJesus S, et al. Prospective comparison of laparoscopic vs. open resections for colorectal adenocarcinoma over a ten-year period. Dis Colon Rectum. 2003;46(5):601–11.
Braga M, Pecorelli N, Frasson M, Vignali A, Zuliani W, Carlo VD. Long-term outcomes after laparoscopic colectomy. World J Gastrointest Oncol. 2011;3(3):43–8.
O’Connell MJ, Campbell ME, Goldberg RM, Grothey A, Seitz JF, Benedetti JK, et al. Survival following recurrence in stage II and III colon cancer: findings from the ACCENT data set. J Clin Oncol. 2008;26(14):2336–41.
Kobayashi H, Mochizuki H, Sugihara K, Morita T, Kotake K, Teramoto T, et al. Characteristics of recurrence and surveillance tools after curative resection for colorectal cancer: a multicenter study. Surgery. 2007;141(1):67–75.
Acknowledgements
This work was supported by the Beijing Municipal Administration of Hospitals Clinical Medicine Development of Special Funding (ZYLX201504) and Support Project of High-level Teachers in Beijing Municipal Universities in the Period of 13th Five–year Plan (IDHT20170516).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors declare that they have no conflict of interest.
Ethics approval and informed consent
The study protocol was approved by the Ethics Committee of the Second Hospital of Hebei Medical University and conducted in accordance with Helsinki’s Declaration. All the patients gave their written informed consent.
Rights and permissions
About this article
Cite this article
Bai, Z., Wang, J., Wang, T. et al. Clinicopathologic parameters associated with postoperative complications and risk factors for tumor recurrence and mortality after tumor resection of patients with colorectal cancer. Clin Transl Oncol 20, 176–192 (2018). https://doi.org/10.1007/s12094-017-1708-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-017-1708-0