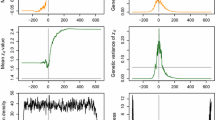
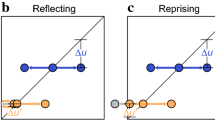
Abstract
Considerable research effort has been spent to understand why most organisms disperse despite the clear costs of doing so. One aspect of dispersal evolution that has received recent attention is a process known as spatial sorting, which has been referred to as the “shy younger sibling” of natural selection. Spatial sorting is the process, whereby variation in dispersal ability is sorted along density clines and will, in nature, often be a transient phenomenon. Despite this transience, spatial sorting is likely a general mechanism behind the evolution of nonzero dispersal rates in spatiotemporally varying environments. While most often transient, spatial sorting is persistent on invasion fronts, where its effect cannot be ignored, causing rapid evolution of traits related to dispersal. Spatial sorting is captured in several elegant models, yet these models require a high level of mathematical sophistication and are not accessible to most evolutionary biologists or their students. Here, we frame spatial sorting in terms of the classic haploid and diploid models of natural selection. We show that, on an invasion front, spatial sorting can be conceptualised precisely as selection operating through space rather than (as with natural selection) time, and that genotypes can be viewed as having both spatial and temporal aspects of fitness. Viewing fitness in this way shows that, on invasion fronts, organisms maximise spatiotemporal fitness, rather than traditional (temporal) fitness. The resultant model is strikingly similar to classic models of natural selection under gene flow. This similarity renders the model easy to understand (and to teach), but also suggests that many established theoretical results around natural selection could apply equally to spatial sorting.



Similar content being viewed by others
References
Balkau B J, Feldman M W (1973) Selection for migration modification. Genetics 74:171–174
Benichou O, Calvez V, Meunier N, Voituriez R (2012) Front acceleration by dynamic selection in Fisher population waves. Phys Rev E 86:041908
Bouin E, Calvez V (2014) Travelling waves for the cane toads equation with bounded traits. Nonlinearity 27:2233
Bouin E, Calvez V, Meunier N et al (2012) Invasion fronts with variable motility: phenotype selection, spatial sorting and wave acceleration. C R Math 350:761–766
Burgess S C, Baskett M L, Grosberg R K et al (2016) When is dispersal for dispersal? Unifying marine and terrestrial perspectives: when is dispersal for dispersal? Biol Rev 91:867–882. https://doi.org/10.1111/brv.12198
Burton O J, Travis J M J, Phillips B L (2010) Trade-offs and the evolution of life-histories during range expansion. Ecol Lett 13:1210–1220. https://doi.org/10.1111/j.1461-0248.2010.01505.x
Case T J, Taper M L (2000) Interspecific competition, environmental gradients, gene flow, and the coevolution of species’ borders. Am Nat 155:583–605
Crow J F, Kimura M (1970) An introduction to population genetics theory. Burgess Publishing Company, Minneapolis
Cwynar L C, MacDonald G M (1987) Geographical variation of lodgepole pine in relation to population history. Am Nat 129:463. https://doi.org/10.1086/284651
Deforet M, Carmona-Fontaine C, Korolev K S, Xavier J B (2017) A simple rule for the evolution of fast dispersal at the edge of expanding populations. arXiv:171107955 [q-bio]
Ellner S P, Schreiber S J (2012) Temporally variable dispersal and demography can accelerate the spread of invading species. Theor Popul Biol 82:283–298
Fisher R A (1937) The wave advance of advantageous genes. Ann Eugen 7:355–369
Fronhofer E A, Altermatt F (2015) Eco-evolutionary feedbacks during experimental range expansions. Nat Commun 6:6844. https://doi.org/10.1038/ncomms7844
Gandon S (1999) Kin competition, the cost of inbreeding and the evolution of dispersal. J Theor Biol 200:345–364. https://doi.org/10.1006/jtbi.1999.0994
Haldane J B S (1924) A mathematical theory of natural and artificial selection—I. Trans Camb Philos Soc 23:19–41
Hamilton W D, May R M (1977) Dispersal in stable habitats. Nature 269:578–581
Hartl D L, Clark AG, Clark AG (1997) Principles of population genetics. Sinauer Associates, Sunderland
Hastings A (1983) Can spatial variation alone lead to selection for dispersal? Theor Popul Biol 24:244–251
Holt R (1985) Population dynamics in two-patch environments: some anomalous consequences of an optimal habitat distribution. Theor Popul Biol 28:181–208. https://doi.org/10.1016/0040-5809(85)90027-9
Hughes C L, Dytham C, Hill J K (2003) Evolutionary trade-offs between reproduction and dispersal in populations at expanding range boundaries. Proc R Soc Biol Sci Ser B 270:S147–S150
Johnson M L, Gaines M S (1990) Evolution of dispersal: theoretical models and empirical tests using birds and mammals. Annu Rev Ecol Syst 21:449–480
Krug P J, Zimmer R K (2004) Developmental dimorphism: consequences for larval behavior and dispersal potential in a marine gastropod. Biol Bull 207:233–246. https://doi.org/10.2307/1543212
Lenormand T (2002) Gene flow and the limits to natural selection. Trends Ecol Evol 17:183–189
Levin S A, Cohen D, Hastings A (1984) Dispersal strategies in patchy environments. Theor Popul Biol 26:165–191. https://doi.org/10.1016/0040-5809(84)90028-5
Lombaert E, Estoup A, Facon B et al (2014) Rapid increase in dispersal during range expansion in the invasive ladybird Harmonia axyridis. J Evol Biol 27:508–517
McPeek MA, Holt RD (1992) The evolution of dispersal in spatially and temporally varying environments. Am Nat 140:1010–1027
Moody M E (1988) The evolution of migration in subdivided populations. I. Haploids. J Theor Biol 131:1–14. https://doi.org/10.1016/S0022-5193(88)80116-4
Nagylaki T (1992) Introduction to theoretical population genetics. Springer, Berlin
Ochocki B M, Miller T E X (2017) Rapid evolution of dispersal ability makes biological invasions faster and more variable. Nat Commun 8:ncomms14315. https://doi.org/10.1038/ncomms14315
Olivieri I, Michalakis Y, Gouyon P-H (1995) Metapopulation genetics and the evolution of dispersal. Am Nat 146:202–228
Otto S P, Day T (2007) A biologist’s guide to mathematical modeling in ecology and evolution. Princeton University Press, Princeton
Peischl S, Gilbert KJ (2018) Evolution of dispersal can rescue populations from expansion load. bioRxiv. https://doi.org/10.1101/483883
Peischl S, Dupanloup I, Kirkpatrick M, Excoffier L (2013) On the accumulation of deleterious mutations during range expansions. Mol Ecol 22:5972–5982. https://doi.org/10.1111/mec.12524
Peischl S, Kirkpatrick M, Excoffier L (2015) Expansion load and the evolutionary dynamics of a species range. American Naturalist, in press
Perkins T A, Phillips B L, Baskett M L, Hastings A (2013) Evolution of dispersal and life-history interact to drive accelerating spread of an invasive species. Ecol Lett 16:1079–1087. https://doi.org/10.1111/ele.12136
Perkins S E, Boettiger C, Phillips B L (2016) After the games are over: life-history trade-offs drive dispersal attenuation following range expansion. Ecol Evol 6:6425–6434. https://doi.org/10.1002/ece3.2314
Phillips B L (2015) Evolutionary processes make invasion speed difficult to predict. Biol Invasions 17:1949–1960. https://doi.org/10.1007/s10530-015-0849-8
Phillips B L, Brown G P, Travis J M J, Shine R (2008) Reid’s paradox revisited: the evolution of dispersal in range-shifting populations. Am Nat 172:S34–S48
Phillips B L, Brown G P, Shine R (2010) Evolutionarily accelerated invasions: the rate of dispersal evolves upwards during range advance of cane toads. J Evol Biol 23:2595–2601. https://doi.org/10.1111/j.1420-9101.2010.02118.x
Ronce O (2007) How does it feel to be like a rolling stone? Ten questions about dispersal evolution. Annu Rev Ecol Evol Syst 38:231–253. https://doi.org/10.1146/annurev.ecolsys.38.091206.095611
Shine R, Brown G P, Phillips B L (2011) An evolutionary process that assembles phenotypes through space rather than through time. Proc Natl Acad Sci 108:5708–5711. https://doi.org/10.1073/pnas.1018989108
Simmons A D, Thomas C D (2004) Changes in dispersal during species’ range expansions. Am Nat 164:378–395
Skellam J G (1951) Random dispersal in theoretical populations. Biometrika 38:196–218
Slatkin M, Excoffier L (2012) Serial founder effects during range expansion: a spatial analog of genetic drift. Genetics 191:171–181. https://doi.org/10.1534/genetics.112.139022
Travis J M J, Dytham C (2002) Dispersal evolution during invasions. Evol Ecol Res 4:1119–1129
van Ditmarsch D, Boyle K E, Sakhtah H et al (2013) Convergent evolution of hyperswarming leads to impaired biofilm formation in pathogenic bacteria. Cell Rep 4:697–708
Van Valen L (1971) Group selection and the evolution of dispersal. Evolution 25:591–598
Weiss-Lehman C, Hufbauer R A, Melbourne B A (2017) Rapid trait evolution drives increased speed and variance in experimental range expansions. Nat Commun 8:ncomms14303. https://doi.org/10.1038/ncomms14303
Wright S (1931) Evolution in Mendelian populations. Genetics 16:97–159
Wright S (1940) Breeding structure of populations in relation to speciation. Am Nat 74:232–248. https://doi.org/10.1086/280891
Acknowledgements
The manuscript was improved by thoughtful comments from Stephan Peischl, Susan Frances Bailey, and Frédéric Guillaume.
Funding
BLP was supported by the Australian Research Council (DP160101730, FT160100198).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Phillips, B.L., Perkins, T.A. Spatial sorting as the spatial analogue of natural selection. Theor Ecol 12, 155–163 (2019). https://doi.org/10.1007/s12080-019-0412-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12080-019-0412-9