Abstract
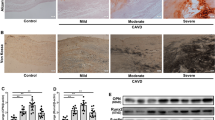
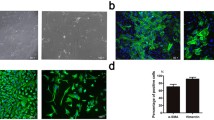
This study aims to elucidate the role of miR-129/miR-342 loaded in exosomes derived from vascular smooth muscle cells (VSMCs) stimulated by intermittent hypoxia in calcified aortic valvular disease (CAVD). Bioinformatics analysis was conducted to identify differentially expressed miRs in VSMCs-derived exosomes and CAVD samples, and their potential target genes were predicted. VSMCs were exposed to intermittent hypoxia to induce stimulation, followed by isolation of exosomes. Valvular interstitial cells (VICs) were cultured in vitro to investigate the impact of miR-129/miR-342 on VICs’ osteogenic differentiation and aortic valve calcification with eIF2α. A CAVD mouse model was established using ApoE knockout mice for in vivo validation. In CAVD samples, miR-129 and miR-342 were downregulated, while eIF2α and ATF4 were upregulated. miR-129 and miR-342 exhibited inhibitory effects on eIF2α through targeted regulation. Exosomes released from intermittently hypoxia-stimulated VSMCs contained miR-129 and miR-342. Overexpression of miR-129 and miR-342, or silencing ATF4, suppressed VICs’ osteogenic differentiation and aortic valve calcification, which could be rescued by overexpressed eIF2α. Collectively, intermittent hypoxia stimulation of VSMCs leads to the secretion of exosomes that activate the miR-129/miR-342 dual pathway, thereby inhibiting the eIF2α/ATF4 axis and attenuating VICs’ osteogenic differentiation and CAVD progression.
Graphical abstract










Similar content being viewed by others
Data availability
The data supporting this study’s findings are available on request from the corresponding author.
References
Alano CC, Garnier P, Ying W, Higashi Y, Kauppinen TM, Swanson RA (2010) NAD+ depletion is necessary and sufficient for poly(ADP-ribose) polymerase-1-mediated neuronal death. J Neurosci 30(8):2967–2978. https://doi.org/10.1523/JNEUROSCI.5552-09.2010
Asano K (2021) Origin of translational control by eIF2alpha phosphorylation: insights from genome-wide translational profiling studies in fission yeast. Curr Genet 67(3):359–368. https://doi.org/10.1007/s00294-020-01149-w
Balogh E, Toth A, Mehes G, Trencsenyi G, Paragh G, Jeney V (2019) Hypoxia triggers osteochondrogenic differentiation of vascular smooth muscle cells in an hif-1 (hypoxia-inducible factor 1)-dependent and reactive oxygen species-dependent manner. Arterioscler Thromb Vasc Biol 39(6):1088–1099. https://doi.org/10.1161/ATVBAHA.119.312509
Barile L, Vassalli G (2017) Exosomes: therapy delivery tools and biomarkers of diseases. Pharmacol Ther 174:63–78. https://doi.org/10.1016/j.pharmthera.2017.02.020
Barile L, Moccetti T, Marban E, Vassalli G (2017) Roles of exosomes in cardioprotection. Eur Heart J 38(18):1372–1379. https://doi.org/10.1093/eurheartj/ehw304
Beckmann E, Grau JB, Sainger R, Poggio P, Ferrari G (2010) Insights into the use of biomarkers in calcific aortic valve disease. J Heart Valve Dis 19(4):441–452
Blaser MC, Aikawa E (2018) Roles and regulation of extracellular vesicles in cardiovascular mineral metabolism. Front Cardiovasc Med 5:187. https://doi.org/10.3389/fcvm.2018.00187
Cho KI, Sakuma I, Sohn IS, Jo SH, Koh KK (2018) Inflammatory and metabolic mechanisms underlying the calcific aortic valve disease. Atherosclerosis 277:60–65. https://doi.org/10.1016/j.atherosclerosis.2018.08.029
Dickhout JG, Carlisle RE, Jerome DE et al (2012) Integrated stress response modulates cellular redox state via induction of cystathionine gamma-lyase: cross-talk between integrated stress response and thiol metabolism. J Biol Chem 287(10):7603–7614. https://doi.org/10.1074/jbc.M111.304576
Fu Z, Li F, Jia L et al (2019) Histone deacetylase 6 reduction promotes aortic valve calcification via an endoplasmic reticulum stress-mediated osteogenic pathway. J Thorac Cardiovasc Surg 158(2):408–417. https://doi.org/10.1016/j.jtcvs.2018.10.136
Geng J, Xu H, Fu W et al (2020) Rosuvastatin protects against endothelial cell apoptosis in vitro and alleviates atherosclerosis in ApoE(-/-) mice by suppressing endoplasmic reticulum stress. Exp Ther Med 20(1):550–560. https://doi.org/10.3892/etm.2020.8733
Geng Z, Xu F, Zhang Y (2016) MiR-129-5p-mediated Beclin-1 suppression inhibits endothelial cell autophagy in atherosclerosis. Am J Transl Res 8(4):1886–1894
Goody PR, Hosen MR, Christmann D et al (2020) Aortic valve stenosis: from basic mechanisms to novel therapeutic targets. Arterioscler Thromb Vasc Biol 40(4):885–900. https://doi.org/10.1161/ATVBAHA.119.313067
Heo J, Yang HC, Rhee WJ, Kang H (2020) Vascular smooth muscle cell-derived exosomal micrornas regulate endothelial cell migration under pdgf stimulation. Cells 9(3):33. https://doi.org/10.3390/cells9030639
Huang P, Peslak SA, Lan X et al (2020) The HRI-regulated transcription factor ATF4 activates BCL11A transcription to silence fetal hemoglobin expression. Blood 135(24):2121–2132. https://doi.org/10.1182/blood.2020005301
Hutcheson JD, Aikawa E, Merryman WD (2014) Potential drug targets for calcific aortic valve disease. Nat Rev Cardiol 11(4):218–231. https://doi.org/10.1038/nrcardio.2014.1
Jung YY, Kim KC, Park MH et al (2018) Atherosclerosis is exacerbated by chitinase-3-like-1 in amyloid precursor protein transgenic mice. Theranostics 8(3):749–766. https://doi.org/10.7150/thno.20183
Kapustin AN, Shanahan CM (2016) Emerging roles for vascular smooth muscle cell exosomes in calcification and coagulation. J Physiol 594(11):2905–2914. https://doi.org/10.1113/JP271340
Kyotani Y, Takasawa S, Yoshizumi M (2019) Proliferative pathways of vascular smooth muscle cells in response to intermittent hypoxia. Int J Mol Sci 20(11):2706. https://doi.org/10.3390/ijms20112706
Latif N, Sarathchandra P, Chester AH, Yacoub MH (2015) Expression of smooth muscle cell markers and co-activators in calcified aortic valves. Eur Heart J 36(21):1335–1345. https://doi.org/10.1093/eurheartj/eht547
Li J, Xue H, Li T et al (2019) Exosomes derived from mesenchymal stem cells attenuate the progression of atherosclerosis in ApoE(-/-) mice via miR-let7 mediated infiltration and polarization of M2 macrophage. Biochem Biophys Res Commun 510(4):565–572. https://doi.org/10.1016/j.bbrc.2019.02.005
Li N, Bai Y, Zhou G et al (2020) miR-214 attenuates aortic valve calcification by regulating osteogenic differentiation of valvular interstitial cells. Mol Ther Nucleic Acids 22:971–980. https://doi.org/10.1016/j.omtn.2020.10.016
Masuda M, Miyazaki-Anzai S, Levi M, Ting TC, Miyazaki M (2013) PERK-eIF2alpha-ATF4-CHOP signaling contributes to TNFalpha-induced vascular calcification. J Am Heart Assoc 2(5):e000238. https://doi.org/10.1161/JAHA.113.000238
Majumdar U, Manivannan S, Basu M et al (2021) Nitric oxide prevents aortic valve calcification by S-nitrosylation of USP9X to activate NOTCH signaling. Sci Adv 7(6):304. https://doi.org/10.1126/sciadv.abe3706
Masuda M, Ting TC, Levi M, Saunders SJ, Miyazaki-Anzai S, Miyazaki M (2012) Activating transcription factor 4 regulates stearate-induced vascular calcification. J Lipid Res 53(8):1543–1552. https://doi.org/10.1194/jlr.M025981
Navarrete-Opazo A, Mitchell GS (2014) Therapeutic potential of intermittent hypoxia: a matter of dose. Am J Physiol Regul Integr Comp Physiol 307(10):R1181-1197. https://doi.org/10.1152/ajpregu.00208.2014
Onat UI, Yildirim AD, Tufanli O et al (2019) Intercepting the lipid-induced integrated stress response reduces atherosclerosis. J Am Coll Cardiol 73(10):1149–1169. https://doi.org/10.1016/j.jacc.2018.12.055
Pan W, Liang J, Tang H et al (2020) Differentially expressed microRNA profiles in exosomes from vascular smooth muscle cells associated with coronary artery calcification. Int J Biochem Cell Biol 118:105645. https://doi.org/10.1016/j.biocel.2019.105645
Peeters F, Meex SJR, Dweck MR et al (2018) Calcific aortic valve stenosis: hard disease in the heart: a biomolecular approach towards diagnosis and treatment. Eur Heart J 39(28):2618–2624. https://doi.org/10.1093/eurheartj/ehx653
Peltonen T, Ohukainen P, Ruskoaho H, Rysa J (2017) Targeting vasoactive peptides for managing calcific aortic valve disease. Ann Med 49(1):63–74. https://doi.org/10.1080/07853890.2016.1231933
Qiu H, Shi S, Wang S, Peng H, Ding SJ, Wang L (2018) Proteomic profiling exosomes from vascular smooth muscle cell. Proteomics Clin Appl 12(5):e1700097. https://doi.org/10.1002/prca.201700097
Raddatz MA, Madhur MS, Merryman WD (2019) Adaptive immune cells in calcific aortic valve disease. Am J Physiol Heart Circ Physiol 317(1):H141–H155. https://doi.org/10.1152/ajpheart.00100.2019
Santos CX, Hafstad AD, Beretta M et al (2016) Targeted redox inhibition of protein phosphatase 1 by Nox4 regulates eIF2alpha-mediated stress signaling. EMBO J 35(3):319–334. https://doi.org/10.15252/embj.201592394
Sathyamurthy I, Alex S (2015) Calcific aortic valve disease: is it another face of atherosclerosis? Indian Heart J 67(5):503–506. https://doi.org/10.1016/j.ihj.2015.07.033
Schlotter F, Halu A, Goto S et al (2018) Spatiotemporal multi-omics mapping generates a molecular atlas of the aortic valve and reveals networks driving disease. Circulation 138(4):377–393. https://doi.org/10.1161/CIRCULATIONAHA.117.032291
Simplicio CL, Purita J, Murrell W, Santos GS, Dos Santos RG, Lana J (2020) Extracorporeal shock wave therapy mechanisms in musculoskeletal regenerative medicine. J Clin Orthop Trauma 11(Suppl 3):S309–S318. https://doi.org/10.1016/j.jcot.2020.02.004
Wang L, Xia JW, Ke ZP, Zhang BH (2019) Blockade of NEAT1 represses inflammation response and lipid uptake via modulating miR-342-3p in human macrophages THP-1 cells. J Cell Physiol 234(4):5319–5326. https://doi.org/10.1002/jcp.27340
Wang Q, Lin P, Feng L, Ren Q, Xie X, Zhang B (2022) Ameliorative effect of allicin on vascular calcification via inhibiting endoplasmic reticulum stress. Vascular 30(5):999–1007. https://doi.org/10.1177/17085381211035291
Xiao N, Zhang J, Chen C, Wan Y, Wang N, Yang J (2019) miR-129-5p improves cardiac function in rats with chronic heart failure through targeting HMGB1. Mamm Genome 30(9–10):276–288. https://doi.org/10.1007/s00335-019-09817-0
Xu F, Zhong JY, Lin X et al (2020) Melatonin alleviates vascular calcification and ageing through exosomal miR-204/miR-211 cluster in a paracrine manner. J Pineal Res 68(3):e12631. https://doi.org/10.1111/jpi.12631
Ye Q, Tian GP, Cheng HP et al (2018) MicroRNA-134 promotes the development of atherosclerosis via the ANGPTL4/LPL pathway in apolipoprotein e knockout mice. J Atheroscler Thromb 25(3):244–253. https://doi.org/10.5551/jat.40212
Yi GW, Zeng QT, Mao XB et al (2011) Overexpression of CXCL16 promotes a vulnerable plaque phenotype in apolipoprotein e-knockout mice. Cytokine 53(3):320–326. https://doi.org/10.1016/j.cyto.2010.11.016
Yu C, Li L, Xie F et al (2018) LncRNA TUG1 sponges miR-204-5p to promote osteoblast differentiation through upregulating Runx2 in aortic valve calcification. Cardiovasc Res 114(1):168–179. https://doi.org/10.1093/cvr/cvx180
Zhang H, Zhang N, Jiang W, Lun X (2021) Clinical significance of the long non-coding RNA NEAT1/miR-129-5p axis in the diagnosis and prognosis for patients with chronic heart failure. Exp Ther Med 21(5):512. https://doi.org/10.3892/etm.2021.9943
Zhang Y, He L, Tu M et al (2021) The ameliorative effect of terpinen-4-ol on ER stress-induced vascular calcification depends on SIRT1-mediated regulation of PERK acetylation. Pharmacol Res 170:105629. https://doi.org/10.1016/j.phrs.2021.105629
Acknowledgment
Not applicable.
Funding
This study was supported by Henan Province Medical Science and Technology Tackling Key Problems Joint Construction Project (LHGJ20220281).
Author information
Authors and Affiliations
Contributions
CH, LY and XH wrote the paper and conceived and designed the experiments; WS, HZ and XZ analyzed the data; GH and JX collected and provided the sample for this study. All authors have read and approved the final submitted manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethics statement
The study was conducted under the approval of the Ethics Committee of The First Affiliated Hospital of Zhengzhou University. All animal experiments were performed following the guidelines for laboratory animal care and use issued by the National Institutes of Health.
Consent for publication
Not applicable.
Consent to participate
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Huang, C., Han, X., Yang, L. et al. Exosomal miR-129 and miR-342 derived from intermittent hypoxia-stimulated vascular smooth muscle cells inhibit the eIF2α/ATF4 axis from preventing calcified aortic valvular disease. J. Cell Commun. Signal. 17, 1449–1467 (2023). https://doi.org/10.1007/s12079-023-00785-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12079-023-00785-4