Abstract
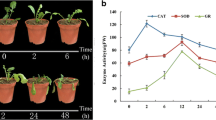
Pineapple (Ananas comosus L. Merr) is a tropical plant with economic importance. We aimed to explore the antioxidase and transcriptome changes of pineapple in response to heat stress using cultivars ‘Xigua’ and ‘MD2′. The antioxidase activity of pineapple at different time points under high temperature (38 °C) was measured. The differentially expressed genes (DEGs) of the two pineapple varieties before and after high temperature treatment were screened and used to construct a protein to protein interaction (PPI) network. Subsequently, the expression of genes with high node degree in the PPI network was verified by qRT-PCR. The antioxidant enzyme activities varied in different cultivars with a time-dependent manner under high temperature. Totally 1214 DEGs was identified in ‘Xigua’ and 261 DEGs were identified in ‘MD2′. The enriched GO terms mainly included ‘response to hydrogen peroxide’ and ‘membrane part’. ‘nitrogen metabolism’ pathway and some secondary substance synthesis pathways were important KEGG pathways. A variety of heat shock proteins (HSPs) was identified to be related to the heat response of pineapple, and most of them showed a significantly high expression level in the high temperature group than that in the normal group. This study laid a certain foundation for the future research on the molecular mechanism of pineapple resistance to high temperature.






Similar content being viewed by others
Availability of data and materials
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Allakhverdiev SI, Kreslavski VD, Klimov VV, Los DA, Carpentier R, Mohanty P (2008) Heat stress: an overview of molecular responses in photosynthesis. Photosynth Res 98(1–3):541–550. https://doi.org/10.1007/s11120-008-9331-0
B L, CN D, (2011) RSEM: accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinformatics 12(18):323–335. https://doi.org/10.1186/1471-2105-12-323
Balfagón D, Zandalinas SI, Baliño P, Muriach M, Gómez-Cadenas A (2018) Involvement of ascorbate peroxidase and heat shock proteins on citrus tolerance to combined conditions of drought and high temperatures. Plant Physiol Bioch 127:194–199. https://doi.org/10.1016/j.plaphy.2018.03.029
Beck EH, Fettig S, Knake C, Hartig K, Bhattarai T (2007) Specific and unspecific responses of plants to cold and drought stress. J Biosci 32(3):501–510. https://doi.org/10.1007/s12038-007-0049-5
Chaudhari RS, Jangale BL, Azeez A, Krishna B, Sane PV, Sane AP (2019) Differential regulation of the banana stress NAC family by individual and combined stresses of drought and heat in susceptible and resistant genotypes. Plant Physiol Bioch 145(37):184–194. https://doi.org/10.1016/j.plaphy.2019.10.040
Chen YE, Zhang CM, Su YQ, Ma J, Zhang ZW, Yuan M, Zhang HY, Yuan S (2017) Responses of photosystem II and antioxidative systems to high light and high temperature co-stress in wheat. Environ Exp Bot 135:45–55. https://doi.org/10.1016/j.envexpbot.2016.12.001
D K, B L, SL S, (2015) HISAT: a fast spliced aligner with low memory requirements. Nat Methods 12(4):357–360. https://doi.org/10.1038/nmeth.3317
Dv K, L Z, BS P, F R, A WV, A N, CJ M, JM Y, O B, M W, et al (2018) GOATOOLS: A Python library for Gene Ontology analyses. Sci Rep 8(1):10872. https://doi.org/10.1038/s41598-018-28948-z
Erkut C, Vasilj A, Boland S, Habermann B, Shevchenko A, Kurzchalia TV (2013) Molecular strategies of the Caenorhabditis elegans dauer larva to survive extreme desiccation. PLoS ONE 8(12):e82473. https://doi.org/10.1371/journal.pone.0082473
Fragkostefanakis S, Röth S, Schleiff E, Scharf KD (2015) Prospects of engineering thermotolerance in crops through modulation of heat stress transcription factor and heat shock protein networks. Plant Cell Environ 38(9):1881–1895. https://doi.org/10.1111/pce.12396
Gong HL, Chen QQ (2021) Exogenous Sucrose Protects Potato Seedlings Against Heat Stress by Enhancing the Antioxidant Defense System. J Soil Sci Plant Nut 6(18):1–9. https://doi.org/10.1007/s42729-021-00457-5
Hayer-Hartl M, Hartl FU, Bracher A (2011) Molecular chaperones in protein folding and proteostasis. Nature 457(7356):324–332. https://doi.org/10.1038/nature10317
He Y, Huang B (2010) Differential responses to heat stress in activities and isozymes of four antioxidant enzymes for two cultivars of Kentucky bluegrass contrasting in heat tolerance. J Am Soc Hortic Sci 135(2):116–124
Hildebrandt TM (2018) Synthesis versus degradation: directions of amino acid metabolism during Arabidopsis abiotic stress response. Plant Mol Biol 98(1–2):121–135. https://doi.org/10.1007/s11103-018-0767-0
Ivanova NN, Khomich LM, Perova IB, Eller KI (2019) Pineapple juice nutritional profile. Voprosy pitaniia 88(2):73–82. https://doi.org/10.24411/0042-8833-2019-10020
Lavania D, Dhingra A, Siddiqui MH, Al-Whaibi MH, Grover A (2015) Current status of the production of high temperature tolerant transgenic crops for cultivation in warmer climates. Plant Physiol Bioch 86:100–108. https://doi.org/10.1016/j.plaphy.2014.11.019
Lavhale SG, Kalunke RM, Giri AP (2018) Structural, functional and evolutionary diversity of 4-coumarate-CoA ligase in plants. Planta 248(5):1063–1078. https://doi.org/10.1007/s00425-018-2965-z
M P, GM P, CM A, TC C, JT M, SL S, (2015) StringTie enables improved reconstruction of a transcriptome from RNA-seq reads. Nat Biotechnol 33(3):290–295. https://doi.org/10.1038/nbt.3122
Mi L, W H, S A, (2014) Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol 15(12):550. https://doi.org/10.1186/s13059-014-0550-8
Mittler R (2006) Abiotic stress, the field environment and stress combination. Trends Plant Sci 11(1):15–19
Pyatrikas DV, Rikhvanov EG, Fedoseeva IV, Varakina NN, Voinikov VK (2014) Mitochondrial retrograde regulation of HSP101 expression in Arabidopsis thaliana under heat stress and amiodarone action. Russ J Plant Physl 61(1):80–89
Rathnavelu V, Alitheen NB, Sohila S, Kanagesan S, Ramesh R (2016) Potential role of bromelain in clinical and therapeutic applications. Biomed Rep 5(3):283–288. https://doi.org/10.3892/br.2016.720
Sanewski GM, Bartholomew DP, Paull RE (2018) The pineapple: botany, production and uses. CABI
Schieber M, Chandel NS (2014) ROS function in redox signaling and oxidative stress. Curr Biol 24(10):R453–R462
Shafqat W, Jaskani MJ, Maqbool R, Chattha WS, Ali Z, Naqvi SA, Haider MS, Khan IA, Vincent CI (2021) Heat shock protein and aquaporin expression enhance water conserving behavior of citrus under water deficits and high temperature conditions. Environ Exp Bot 181. https://doi.org/10.1016/j.envexpbot.2020.104270
Shao L, Shu Z, Sun SL, Peng CL, Wang XJ, Lin ZF (2007) Antioxidlation of Anthocyanins in photosynthesis under high temperature stress. J Integr Plant Biol 49(9):1341–1351. https://doi.org/10.1111/j.1744-7909.2007.00527.x
Shekhar S, Mishra D, Gayali S, Buragohain AK, Chakraborty S, Chakraborty N (2016) Comparison of proteomic and metabolomic profiles of two contrasting ecotypes of sweetpotato (Ipomoea batata L.). J Proteomics 143:306–317. https://doi.org/10.1016/j.jprot.2016.03.028
Sun H, Li Y, Feng S, Zou W, Guo K, Fan C, Si S, Peng L (2013) Analysis of five rice 4-coumarate:coenzyme A ligase enzyme activity and stress response for potential roles in lignin and flavonoid biosynthesis in rice. Biochem Bioph Res Co 430(3):1151–1156. https://doi.org/10.1016/j.bbrc.2012.12.019
Szabados L, Savouré A (2010) Proline: a multifunctional amino acid. Trends Plant Sci 15(2):89–97. https://doi.org/10.1016/j.tplants.2009.11.009
Tang Y, Horikoshi M, Li W (2016) ggfortify: unified interface to visualize statistical results of popular R packages. The R Journal 8(2):478–489
Torun H (2019) Time-course analysis of salicylic acid effects on ROS regulation and antioxidant defense in roots of hulled and hulless barley under combined stress of drought, heat and salinity. Physiol Plant 165(2):169–182. https://doi.org/10.1111/ppl.12798
Tripathy BC, Oelmüller R (2012) Reactive oxygen species generation and signaling in plants. Plant Signal Behav 7(12):1621–1633. https://doi.org/10.4161/psb.22455
Ul Haq S, Khan A, Ali M, Khattak AM, Gai WX, Zhang HX, Wei AM, Gong ZH (2019) Heat Shock Proteins: Dynamic Biomolecules to Counter Plant Biotic and Abiotic Stresses. Int J Mol Sci 20(21):5321–5345. https://doi.org/10.3390/ijms20215321
Vives-Peris V, Gómez-Cadenas A, Pérez-Clemente RM (2017) Citrus plants exude proline and phytohormones under abiotic stress conditions. Plant Cell Rep 36(12):1971–1984. https://doi.org/10.1007/s00299-017-2214-0
Volkov RA, Panchuk II, Mullineaux PM, Schöffl F (2006) Heat stress-induced H2O2 is required for effective expression of heat shock genes in Arabidopsis. Plant Mol Biol 61(4–5):733–746. https://doi.org/10.1007/s11103-006-0045-4
Xiaomei W, Jing Z, Zhi L, Jian X, Yeqiang L, Hui R (2019) Introduction performance and main cultivation techniques of “Xigua.” Fruit trees in South China 10(4):127–130
Xie T, Chen C, Li C, Liu J, Liu C, He Y (2018) Genome-wide investigation of WRKY gene family in pineapple: evolution and expression profiles during development and stress. BMC Genomics 19(1):472–490. https://doi.org/10.1186/s12864-018-4880-x
Yang X, Cushman JC, Borland AM, Edwards EJ, Wullschleger SD, Tuskan GA, Owen NA, Griffiths H, Smith JA, De Paoli HC et al (2015) A roadmap for research on crassulacean acid metabolism (CAM) to enhance sustainable food and bioenergy production in a hotter, drier world. New Phytol 207(3):491–504. https://doi.org/10.1111/nph.13393
Yang X, Pang X, Xu L, Fang R, Huang X, Guan P, Lu W, Zhang Z (2009) Accumulation of soluble sugars in peel at high temperature leads to stay-green ripe banana fruit. J Exp Bot 60(14):4051–4062. https://doi.org/10.1093/jxb/erp238
Yongya M, Tujian X, Weisheng S, Yuhua M (2020) Introduction performance and cultivation techniques of MD2 in Guizhou hot area. Agr Tech Serv 37(11):19–20
Zandalinas SI, Balfagón D, Arbona V, Gómez-Cadenas A (2017) Modulation of Antioxidant Defense System Is Associated with Combined Drought and Heat Stress Tolerance in Citrus. Front Plant Sci 8(7):953. https://doi.org/10.3389/fpls.2017.00953
Zhang CH, Ma T, Luo WC, Xu JM, Liu JQ, Wan DS (2015) Identification of 4CL Genes in Desert Poplars and Their Changes in Expression in Response to Salt Stress. Genes 6(3):901–917. https://doi.org/10.3390/genes6030901
Zhang K, Cui H, Cao S, Yan L, Li M, Sun Y (2019) Overexpression of CrCOMT from Carex rigescens increases salt stress and modulates melatonin synthesis in Arabidopsis thaliana. Plant Cell Rep 38(12):1501–1514. https://doi.org/10.1007/s00299-019-02461-7
Zhu H, Zhang Y, Tang R, Qu H, Duan X, Jiang Y (2019) Banana sRNAome and degradome identify microRNAs functioning in differential responses to temperature stress. BMC Genomics 20(1):20–33. https://doi.org/10.1186/s12864-018-5395-1
Funding
This study is supported by the Special Research Project of Tropical Crops College of Yunnan Agricultural University (No. 2019RYZX001); Central Public-Interest Scientific Institution Basal Research Fund for Chinese Academy of Tropical Agricultural Sciences (No. 1630062020030); Key Research & Development Project of Shanxi Province (No. 201803D221011-5); and Scientific Research Fund of Yunnan Provincial Department of Education (No. 2020j0250).
Author information
Authors and Affiliations
Contributions
WZ devise the research and drafted the manuscript. ZM and SL acquired and analyzed the data. WY did a statistical analysis of the data. JM revised the manuscript for important intellectual content. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Communicated by Yuan Qin.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
12042_2021_9287_MOESM1_ESM.tif
Supplementary file1 Figure S1. qRT-PCR verification the expression level between XGBL_T (38 °C) and XGBL_C (25 °C). * and ** represents p < 0.05 and p < 0.01, respectively. (TIF 863 KB)
12042_2021_9287_MOESM2_ESM.tif
Supplementary file2 Figure S2. qRT-PCR verification the expression level between MD2_T (38 °C) and MD2_C (25 °C). * and ** represents p < 0.05 and p < 0.01, respectively. (TIF 878 KB)
12042_2021_9287_MOESM6_ESM.xlsx
Supplementary file6 Table S4. The topological properties of protein nodes in the PPI network of 'MD2′ cultivar. (XLSX 59 KB)
12042_2021_9287_MOESM7_ESM.xlsx
Supplementary file7 Table S5. The topological properties of protein nodes in the PPI network of 'Xigua' cultivar. (XLSX 227 KB)
12042_2021_9287_MOESM8_ESM.xls
Supplementary file8 Table S6. The topological properties of protein nodes in the PPI network of 'Xigua' cultivar. (XLS 18 KB)
12042_2021_9287_MOESM9_ESM.xls
Supplementary file9 Table S7. The topological properties of protein nodes in the PPI network of 'MD2′ cultivar. (XLS 16 KB)
Rights and permissions
About this article
Cite this article
Zhao, W., Ma, Z., Liu, S. et al. Transcriptome Profiling Reveals Potential Genes and Pathways Supporting Ananas comosus L. Merr’s High Temperature Stress Tolerance. Tropical Plant Biol. 14, 132–142 (2021). https://doi.org/10.1007/s12042-021-09287-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12042-021-09287-2