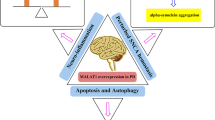
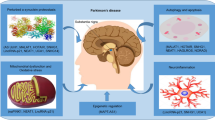
Abstract
Parkinson’s disease (PD) is one of the complex neurodegenerative disorders, primarily characterized by motor deficits, including bradykinesia, tremor, rigidity, and postural instability. The underlying pathophysiology involves the progressive loss of dopaminergic neurons within the substantia nigra pars compacta, leading to dopamine depletion in the basal ganglia circuitry. While motor symptoms are hallmark features of PD, emerging research highlights a wide range of non-motor symptoms, including cognitive impairments, mood disturbances, and autonomic dysfunctions. Inflammasome activation is pivotal in inducing neuroinflammation and promoting disease onset, progression, and severity of PD. Several studies have shown that long noncoding RNAs (lncRNAs) modulate inflammasomes in the pathogenesis of neurodegenerative diseases. Dysregulation of lncRNAs is linked to aberrant gene expression and cellular processes in neurodegeneration, causing the activation of inflammasomes that contribute to neuroinflammation and neurodegeneration. Inflammasomes are cytosolic proteins that form complexes upon activation, inducing inflammation and neuronal cell death. This review explores the significance of lncRNAs in regulating inflammasomes in PD, primarily focusing on specific lncRNAs such as nuclear paraspeckle assembly transcript 1 (NEATNEAT1), X-inactive specific transcript (XIST), growth arrest-specific 5 (GAS5), and HOX transcript antisense RNA (HOTAIR), which have been shown to activate or inhibit the NLRP3 inflammasome and induce the release of proinflammatory cytokines. Moreover, some lncRNAs mediate inflammasome activation through miRNA interactions. Understanding the roles of lncRNAs in inflammasome regulation provides new therapeutic targets for controlling neuroinflammation and reducing the progression of neurodegeneration. Identifying lncRNA-mediated regulatory pathways paves the way for novel therapies in the battle against these devastating neurodegenerative disorders.

Similar content being viewed by others
Data Availability
Not applicable.
Code Availability
Not applicable.
Abbreviations
- AIM2:
-
Absent in melanoma 2
- AKI:
-
Acute kidney injury
- ANRIL:
-
Antisense noncoding RNA in the INK4 locus
- ASC:
-
Apoptosis-associated speck-like protein containing a CARD
- CCI:
-
Chronic constriction injury
- COPD:
-
Chronic obstructive pulmonary disease
- CRNDE:
-
Colorectal neoplasia differentially expressed
- DAMPs:
-
Danger-associated molecular patterns
- DLX6-AS1:
-
Distal-less homeobox 6 antisense 1
- FIRRE:
-
Functional intergenic repeating RNA element
- GAS5:
-
Growth arrest-specific 5
- GBA:
-
Glucocerebrosidase
- GSDMD:
-
Gasdermin D
- HAGLR:
-
HOXD antisense growth-associated long noncoding RNA
- HOTAIR:
-
LncRNA HOX antisense intergenic RNA
- HOTTIP:
-
HOXA transcript at the distal tip
- HOXA11-AS:
-
Homeobox A11 antisense
- HUVEC:
-
Human umbilical vein endothelial cell
- Lfar1:
-
Liver-fibrosis associated lncRNA1
- LRRK2:
-
Leucine-rich repeat kinase 2
- MALAT1:
-
Metastasis-associated lung adenocarcinoma transcript 1
- MDRL:
-
Mitochondrial dynamic-related lncRNA
- Meg3:
-
Maternally expressed gene 3
- MIAT:
-
Myocardial infarction-associated transcript
- MPTP:
-
1-Methyl-4-phenyl-1,2,3,6-tetrahydropyridine
- NACHT:
-
Nucleotide-binding oligomerization domain
- NEAT1:
-
Nuclear paraspeckle assembly transcript 1
- NF-κB:
-
Nuclear factor-κB
- NLRC4:
-
NOD-like receptor family-C
- NLRP1:
-
NLR family pyrin domain containing 1
- NLRs:
-
Nod-like receptors
- NSCLC:
-
Non-small cell lung cancer
- PAMPs:
-
Pattern-associated molecular patterns
- PD:
-
Parkinson’s disease
- Platr4:
-
Pluripotency-associated transcript 4
- PVT1:
-
Plasmacytoma variant translocation 1
- RGMB-AS1:
-
Repulsive guidance molecule bone morphogenetic protein coreceptor b antisense RNA 1
- RMRP:
-
RNA component of mitochondrial RNAase P
- SNHG:
-
Small nucleolar RNA host gene
- TLR:
-
Toll-like receptors
- UC:
-
Ulcerative colitis
- VA-HF:
-
Ventricular arrhythmia associated with heart failure
- XIST:
-
X-inactive specific transcript
- ZFAS1:
-
Zinc finger antisense 1
References
Tang Y, Le W (2016) Differential roles of M1 and M2 microglia in neurodegenerative diseases. Mol Neurobiol 53(2):1181–1194. https://doi.org/10.1007/s12035-014-9070-5
Simon DK, Tanner CM, Brundin P (2020) Parkinson disease epidemiology, pathology, genetics, and pathophysiology. Clin Geriatr Med 36(1):1–12. https://doi.org/10.1016/j.cger.2019.08.002
Ou Z, Pan J, Tang S, Duan D, Yu D, Nong H, Wang Z (2021) Global trends in the incidence, prevalence, and years lived with disability of Parkinson’s disease in 204 countries/territories from 1990 to 2019. Front Public Health 9:776847. https://doi.org/10.3389/fpubh.2021.776847
Stefanis L (2012) alpha-Synuclein in Parkinson’s disease. Cold Spring Harb Perspect Med 2(2):a009399. https://doi.org/10.1101/cshperspect.a009399
Xiong Y, Dawson TM, Dawson VL (2017) Models of LRRK2-associated Parkinson’s Disease. Adv Neurobiol 14:163–191. https://doi.org/10.1007/978-3-319-49969-7_9
Wang Q, Liu Y, Zhou J (2015) Neuroinflammation in Parkinson’s disease and its potential as therapeutic target. Transl Neurodegener 4:19. https://doi.org/10.1186/s40035-015-0042-0
Yang I, Han SJ, Kaur G, Crane C, Parsa AT (2010) The role of microglia in central nervous system immunity and glioma immunology. J Clin Neurosci 17(1):6–10. https://doi.org/10.1016/j.jocn.2009.05.006
Chen WW, Zhang X, Huang WJ (2016) Role of neuroinflammation in neurodegenerative diseases (Review). Mol Med Rep 13(4):3391–3396. https://doi.org/10.3892/mmr.2016.4948
Ramesh G, MacLean AG, Philipp MT (2013) Cytokines and chemokines at the crossroads of neuroinflammation, neurodegeneration, and neuropathic pain. Mediators Inflamm 2013:480739. https://doi.org/10.1155/2013/480739
Xu L, He D, Bai Y (2016) Microglia-mediated inflammation and neurodegenerative disease. Mol Neurobiol 53(10):6709–6715. https://doi.org/10.1007/s12035-015-9593-4
Azam S, Jakaria M, Kim IS, Kim J, Haque ME, Choi DK (2019) Regulation of toll-like receptor (TLR) signaling pathway by polyphenols in the treatment of age-linked neurodegenerative diseases: focus on TLR4 signaling. Front Immunol 10:1000. https://doi.org/10.3389/fimmu.2019.01000
Panicker N, Kam TI, Wang H, Neifert S, Chou SC, Kumar M, Brahmachari S, Jhaldiyal A, Hinkle JT, Akkentli F, Mao X, Xu E, Karuppagounder SS, Hsu ET, Kang SU, Pletnikova O, Troncoso J, Dawson VL, Dawson TM (2022) Neuronal NLRP3 is a parkin substrate that drives neurodegeneration in Parkinson’s disease. Neuron 110(15):2422-2437 e2429. https://doi.org/10.1016/j.neuron.2022.05.009
Jarroux J, Morillon A, Pinskaya M (2017) History, discovery, and classification of lncRNAs. Adv Exp Med Biol 1008:1–46. https://doi.org/10.1007/978-981-10-5203-3_1
St Laurent G, Wahlestedt C, Kapranov P (2015) The Landscape of long noncoding RNA classification. Trends Genet 31(5):239–251. https://doi.org/10.1016/j.tig.2015.03.007
Hombach S, Kretz M (2016) Noncoding RNAs: classification, biology and functioning. Adv Exp Med Biol 937:3–17. https://doi.org/10.1007/978-3-319-42059-2_1
Ma L, Bajic VB, Zhang Z (2013) On the classification of long noncoding RNAs. RNA Biol 10(6):925–933. https://doi.org/10.4161/rna.24604
Quinn JJ, Chang HY (2016) Unique features of long noncoding RNA biogenesis and function. Nat Rev Genet 17(1):47–62. https://doi.org/10.1038/nrg.2015.10
Ali T, Grote P (2020) Beyond the RNA-dependent function of lncRNA genes. eLife 9: https://doi.org/10.7554/eLife.60583
Bridges MC, Daulagala AC, Kourtidis A (2021) LNCcation: lncRNA localization and function. J Cell Biol 220:2. https://doi.org/10.1083/jcb.202009045
Sivagurunathan N, Ambatt ATS, Calivarathan L (2022) Role of long noncoding RNAs in the pathogenesis of Alzheimer’s and Parkinson’s diseases. Curr Aging Sci 15(2):84–96. https://doi.org/10.2174/1874609815666220126095847
Cortini F, Roma F, Villa C (2019) Emerging roles of long noncoding RNAs in the pathogenesis of Alzheimer’s disease. Ageing Res Rev 50:19–26. https://doi.org/10.1016/j.arr.2019.01.001
Haque ME, Akther M, Jakaria M, Kim IS, Azam S, Choi DK (2020) Targeting the microglial NLRP3 inflammasome and its role in Parkinson’s disease. Mov Disord 35(1):20–33. https://doi.org/10.1002/mds.27874
Yang S, Yang H, Luo Y, Deng X, Zhou Y, Hu B (2021) Long noncoding RNAs in neurodegenerative diseases. Neurochem Int 148:105096. https://doi.org/10.1016/j.neuint.2021.105096
Brahadeeswaran S, Sivagurunathan N, Calivarathan L (2022) Inflammasome signaling in the aging brain and age-related neurodegenerative diseases. Mol Neurobiol 59(4):2288–2304. https://doi.org/10.1007/s12035-021-02683-5
Guo H, Callaway JB, Ting JP (2015) Inflammasomes: mechanism of action, role in disease, and therapeutics. Nat Med 21(7):677–687. https://doi.org/10.1038/nm.3893
Strowig T, Henao-Mejia J, Elinav E, Flavell R (2012) Inflammasomes in health and disease. Nature 481(7381):278–286. https://doi.org/10.1038/nature10759
Broz P, Dixit VM (2016) Inflammasomes: mechanism of assembly, regulation and signalling. Nat Rev Immunol 16(7):407–420. https://doi.org/10.1038/nri.2016.58
He Y, Hara H, Nunez G (2016) Mechanism and regulation of NLRP3 inflammasome activation. Trends Biochem Sci 41(12):1012–1021. https://doi.org/10.1016/j.tibs.2016.09.002
Chen X, He WT, Hu L, Li J, Fang Y, Wang X, Xu X, Wang Z, Huang K, Han J (2016) Pyroptosis is driven by non-selective gasdermin-D pore and its morphology is different from MLKL channel-mediated necroptosis. Cell Res 26(9):1007–1020. https://doi.org/10.1038/cr.2016.100
Man SM, Karki R, Kanneganti TD (2017) Molecular mechanisms and functions of pyroptosis, inflammatory caspases and inflammasomes in infectious diseases. Immunol Rev 277(1):61–75. https://doi.org/10.1111/imr.12534
Yu P, Zhang X, Liu N, Tang L, Peng C, Chen X (2021) Pyroptosis: mechanisms and diseases. Signal Transduct Target Ther 6(1):128. https://doi.org/10.1038/s41392-021-00507-5
Munoz-Planillo R, Kuffa P, Martinez-Colon G, Smith BL, Rajendiran TM, Nunez G (2013) K(+) efflux is the common trigger of NLRP3 inflammasome activation by bacterial toxins and particulate matter. Immunity 38(6):1142–1153. https://doi.org/10.1016/j.immuni.2013.05.016
Abais JM, Xia M, Zhang Y, Boini KM, Li PL (2015) Redox regulation of NLRP3 inflammasomes: ROS as trigger or effector? Antioxid Redox Signal 22(13):1111–1129. https://doi.org/10.1089/ars.2014.5994
Chevriaux A, Pilot T, Derangere V, Simonin H, Martine P, Chalmin F, Ghiringhelli F, Rebe C (2020) Cathepsin B is required for NLRP3 inflammasome activation in macrophages, through NLRP3 interaction. Front Cell Dev Biol 8:167. https://doi.org/10.3389/fcell.2020.00167
Murakami T, Ockinger J, Yu J, Byles V, McColl A, Hofer AM, Horng T (2012) Critical role for calcium mobilization in activation of the NLRP3 inflammasome. Proc Natl Acad Sci U S A 109(28):11282–11287. https://doi.org/10.1073/pnas.1117765109
Shimada K, Crother TR, Karlin J, Dagvadorj J, Chiba N, Chen S, Ramanujan VK, Wolf AJ, Vergnes L, Ojcius DM, Rentsendorj A, Vargas M, Guerrero C, Wang Y, Fitzgerald KA, Underhill DM, Town T, Arditi M (2012) Oxidized mitochondrial DNA activates the NLRP3 inflammasome during apoptosis. Immunity 36(3):401–414. https://doi.org/10.1016/j.immuni.2012.01.009
Davis BK, Wen H, Ting JP (2011) The inflammasome NLRs in immunity, inflammation, and associated diseases. Annu Rev Immunol 29:707–735. https://doi.org/10.1146/annurev-immunol-031210-101405
Kelley N, Jeltema D, Duan Y, He Y (2019) The NLRP3 inflammasome: an overview of mechanisms of activation and regulation. Int J Mol Sci 20:13. https://doi.org/10.3390/ijms20133328
Wan P, Su W, Zhang Y, Li Z, Deng C, Li J, Jiang N, Huang S, Long E, Zhuo Y (2020) LncRNA H19 initiates microglial pyroptosis and neuronal death in retinal ischemia/reperfusion injury. Cell Death Differ 27(1):176–191. https://doi.org/10.1038/s41418-019-0351-4
Liu Y, Luo Y, Zhang A, Wang Z, Wang X, Yu Q, Zhang Z, Zhu Z, Wang K, Chen L, Nie X, Zhang JH, Zhang J, Fang Y, Su Z, Chen S (2022) Long noncoding RNA H19 promotes NLRP3-mediated pyroptosis after subarachnoid hemorrhage in rats. Transl Stroke Res. https://doi.org/10.1007/s12975-022-01104-6
Zhang P, Cao L, Zhou R, Yang X, Wu M (2019) The lncRNA NEAT1 promotes activation of inflammasomes in macrophages. Nat Commun 10(1):1495. https://doi.org/10.1038/s41467-019-09482-6
Zhang M, Zheng Y, Sun Y, Li S, Chen L, Jin X, Hou X, Liu X, Chen Q, Li J, Liu M, Zheng X, Zhang Y, Wu J, Yu B (2019) Knockdown of NEAT1 induces tolerogenic phenotype in dendritic cells by inhibiting activation of NLRP3 inflammasome. Theranostics 9(12):3425–3442. https://doi.org/10.7150/thno.33178
An Q, Zhou Z, Xie Y, Sun Y, Zhang H, Cao Y (2021) Knockdown of long noncoding RNA NEAT1 relieves the inflammatory response of spinal cord injury through targeting miR-211-5p/MAPK1 axis. Bioengineered 12(1):2702–2712. https://doi.org/10.1080/21655979.2021.1930925
Yao T, Song Y, Li S, Gu J, Yan X (2022) Inhibition of lncRNA NEAT1 protects endothelial cells against hypoxia/reoxygenation-induced NLRP3 inflammasome activation by targeting the miR-204/BRCC3 axis. Mol Med Rep 25:1. https://doi.org/10.3892/mmr.2021.12548
Dai W, Wang M, Wang P, Wen J, Wang J, Cha S, Xiao X, He Y, Shu R, Bai D (2021) lncRNA NEAT1 ameliorates LPS-induced inflammation in MG63 cells by activating autophagy and suppressing the NLRP3 inflammasome. Int J Mol Med 47(2):607–620. https://doi.org/10.3892/ijmm.2020.4827
Ren N, Jiang T, Wang C, Xie S, Xing Y, Piao D, Zhang T, Zhu Y (2020) LncRNA ADAMTS9-AS2 inhibits gastric cancer (GC) development and sensitizes chemoresistant GC cells to cisplatin by regulating miR-223-3p/NLRP3 axis. Aging (Albany NY) 12(11):11025–11041. https://doi.org/10.18632/aging.103314
Zhang K, Shi Z, Zhang M, Dong X, Zheng L, Li G, Han X, Yao Z, Han T, Hong W (2020) Silencing lncRNA Lfar1 alleviates the classical activation and pyoptosis of macrophage in hepatic fibrosis. Cell Death Dis 11(2):132. https://doi.org/10.1038/s41419-020-2323-5
Liang J, Wang Q, Li JQ, Guo T, Yu D (2020) Long noncoding RNA MEG3 promotes cerebral ischemia-reperfusion injury through increasing pyroptosis by targeting miR-485/AIM2 axis. Exp Neurol 325:113139. https://doi.org/10.1016/j.expneurol.2019.113139
Meng J, Ding T, Chen Y, Long T, Xu Q, Lian W, Liu W (2021) LncRNA-Meg3 promotes Nlrp3-mediated microglial inflammation by targeting miR-7a-5p. Int Immunopharmacol 90:107141. https://doi.org/10.1016/j.intimp.2020.107141
Liao H, Zhang S, Qiao J (2020) Silencing of long noncoding RNA MEG3 alleviates lipopolysaccharide-induced acute lung injury by acting as a molecular sponge of microRNA-7b to modulate NLRP3. Aging (Albany NY) 12(20):20198–20211. https://doi.org/10.18632/aging.103752
Zou DM, Zhou SM, Li LH, Zhou JL, Tang ZM, Wang SH (2020) Knockdown of long noncoding RNAs of maternally expressed 3 alleviates hyperoxia-induced lung injury via inhibiting thioredoxin-interacting protein-mediated pyroptosis by binding to miR-18a. Am J Pathol 190(5):994–1005. https://doi.org/10.1016/j.ajpath.2019.12.013
Xue Z, Zhang Z, Liu H, Li W, Guo X, Zhang Z, Liu Y, Jia L, Li Y, Ren Y, Yang H, Zhang L, Zhang Q, Da Y, Hao J, Yao Z, Zhang R (2019) lincRNA-Cox2 regulates NLRP3 inflammasome and autophagy mediated neuroinflammation. Cell Death Differ 26(1):130–145. https://doi.org/10.1038/s41418-018-0105-8
Liu J, Yao L, Zhang M, Jiang J, Yang M, Wang Y (2019) Downregulation of LncRNA-XIST inhibited development of non-small cell lung cancer by activating miR-335/SOD2/ROS signal pathway mediated pyroptotic cell death. Aging (Albany NY) 11(18):7830–7846. https://doi.org/10.18632/aging.102291
Ma M, Pei Y, Wang X, Feng J, Zhang Y, Gao MQ (2019) LncRNA XIST mediates bovine mammary epithelial cell inflammatory response via NF-kappaB/NLRP3 inflammasome pathway. Cell Prolif 52(1):e12525. https://doi.org/10.1111/cpr.12525
Yu SY, Dong B, Tang L, Zhou SH (2018) LncRNA MALAT1 sponges miR-133 to promote NLRP3 inflammasome expression in ischemia-reperfusion injured heart. Int J Cardiol 254:50. https://doi.org/10.1016/j.ijcard.2017.10.071
Wang S, Lin Y, Li F, Qin Z, Zhou Z, Gao L, Yang Z, Wang Z, Wu B (2020) An NF-kappaB-driven lncRNA orchestrates colitis and circadian clock. Sci Adv 6:42. https://doi.org/10.1126/sciadv.abb5202
Yi H, Peng R, Zhang LY, Sun Y, Peng HM, Liu HD, Yu LJ, Li AL, Zhang YJ, Jiang WH, Zhang Z (2017) LincRNA-Gm4419 knockdown ameliorates NF-kappaB/NLRP3 inflammasome-mediated inflammation in diabetic nephropathy. Cell Death Dis 8(2):e2583. https://doi.org/10.1038/cddis.2016.451
Brocker CN, Kim D, Melia T, Karri K, Velenosi TJ, Takahashi S, Aibara D, Bonzo JA, Levi M, Waxman DJ, Gonzalez FJ (2020) Long noncoding RNA Gm15441 attenuates hepatic inflammasome activation in response to PPARA agonism and fasting. Nat Commun 11(1):5847. https://doi.org/10.1038/s41467-020-19554-7
Deng X, Guo J, Sun Z, Liu L, Zhao T, Li J, Tang G, Zhang H, Wang W, Cao S, Zhu D, Tao T, Cao G, Baryshnikov PI, Chen C, Zhao Z, Chen L, Zhang H (2020) Brucella-induced downregulation of lncRNA Gm28309 triggers macrophages inflammatory response through the miR-3068-5p/NF-kappaB pathway. Front Immunol 11:581517. https://doi.org/10.3389/fimmu.2020.581517
Luo D, Dai W, Feng X, Ding C, Shao Q, Xiao R, Zhao N, Peng W, Yang Y, Cui Y, Liu F, Qian K (2021) Suppression of lncRNA NLRP3 inhibits NLRP3-triggered inflammatory responses in early acute lung injury. Cell Death Dis 12(10):898. https://doi.org/10.1038/s41419-021-04180-y
Chi K, Geng X, Liu C, Zhang Y, Cui J, Cai G, Chen X, Wang F, Hong Q (2021) LncRNA-HOTAIR promotes endothelial cell pyroptosis by regulating the miR-22/NLRP3 axis in hyperuricaemia. J Cell Mol Med 25(17):8504–8521. https://doi.org/10.1111/jcmm.16812
Zhang C, Gong Y, Li N, Liu X, Zhang Y, Ye F, Guo Q, Zheng J (2021) Long noncoding RNA Kcnq1ot1 promotes sC5b-9-induced podocyte pyroptosis by inhibiting miR-486a-3p and upregulating NLRP3. Am J Physiol Cell Physiol 320(3):C355–C364. https://doi.org/10.1152/ajpcell.00403.2020
Wang J, Jiao P, Wei X, Zhou Y (2021) Silencing long noncoding RNA Kcnq1ot1 limits acute kidney injury by promoting miR-204-5p and blocking the activation of NLRP3 inflammasome. Front Physiol 12:721524. https://doi.org/10.3389/fphys.2021.721524
Han Y, Huang Y, Yang Q, Jia L, Zheng Y, Li W (2022) Long noncoding RNA SNHG5 mediates periodontal inflammation through the NF-kappaB signalling pathway. J Clin Periodontol 49(10):1038–1051. https://doi.org/10.1111/jcpe.13684
Cao B, Wang T, Qu Q, Kang T, Yang Q (2018) Long noncoding RNA SNHG1 promotes neuroinflammation in Parkinson’s disease via regulating miR-7/NLRP3 pathway. Neuroscience 388:118–127. https://doi.org/10.1016/j.neuroscience.2018.07.019
Lin Y, Wang S, Gao L, Zhou Z, Yang Z, Lin J, Ren S, Xing H, Wu B (2021) Oscillating lncRNA Platr4 regulates NLRP3 inflammasome to ameliorate nonalcoholic steatohepatitis in mice. Theranostics 11(1):426–444. https://doi.org/10.7150/thno.50281
Liu XH, Wu LM, Wang JL, Dong XH, Zhang SC, Li XH, Xu H, Liu DB, Li ZH, Liu ZM, Wu SG, Hu YW (2021) Long noncoding RNA RP11–490M8.1 inhibits lipopolysaccharide-induced pyroptosis of human umbilical vein endothelial cells via the TLR4/NF-kappaB pathway. Immunobiology 226(5):152133. https://doi.org/10.1016/j.imbio.2021.152133
Mo R, Li J, Chen Y, Ding Y (2022) lncRNA GAS5 promotes pyroptosis in COPD by functioning as a ceRNA to regulate the miR-223-3p/NLRP3 axis. Mol Med Rep 26:1. https://doi.org/10.3892/mmr.2022.12735
You L, Zheng Y, Yang J, Hou Q, Wang L, Zhang Y, Zhao C, Xie R (2022) LncRNA MDRL mitigates atherosclerosis through miR-361/SQSTM1/NLRP3 signaling. Mediators Inflamm 2022:5463505. https://doi.org/10.1155/2022/5463505
Liang Y, Wang B, Huang H, Wang M, Wu Q, Zhao Y, He Y (2021) Silenced SOX2-OT alleviates ventricular arrhythmia associated with heart failure by inhibiting NLRP3 expression via regulating miR-2355-3p. Immun Inflamm Dis 9(1):255–264. https://doi.org/10.1002/iid3.388
Zhang X, Huang Z, Wang Y, Wang T, Li J, Xi P (2021) Long noncoding RNA RMRP contributes to sepsis-induced acute kidney injury. Yonsei Med J 62(3):262–273. https://doi.org/10.3349/ymj.2021.62.3.262
Wang Z, Kun Y, Lei Z, Dawei W, Lin P, Jibo W (2021) LncRNA MIAT downregulates IL-1beta, TNF-a to suppress macrophage inflammation but is suppressed by ATP-induced NLRP3 inflammasome activation. Cell Cycle 20(2):194–203. https://doi.org/10.1080/15384101.2020.1867788
Shen M, Pan X, Gao Y, Ye H, Zhang J, Chen Y, Pan M, Huang W, Xu X, Zhao Y, Jin L (2022) LncRNA CRNDE exacerbates IgA nephropathy progression by promoting NLRP3 inflammasome activation in macrophages. Immunol Invest 51(5):1515–1527. https://doi.org/10.1080/08820139.2021.1989461
Cao H, Han X, Jia Y, Zhang B (2021) Inhibition of long noncoding RNA HOXA11-AS against neuroinflammation in Parkinson’s disease model via targeting miR-124-3p mediated FSTL1/NF-kappaB axis. Aging (Albany NY) 13(8):11455–11469. https://doi.org/10.18632/aging.202837
Zhang Q, Zhou L, Xie H, Zhang H, Gao X (2021) HAGLR aggravates neuropathic pain and promotes inflammatory response and apoptosis of lipopolysaccharide-treated SH-SY5Y cells by sequestering miR-182-5p from ATAT1 and activating NLRP3 inflammasome. Neurochem Int 145:105001. https://doi.org/10.1016/j.neuint.2021.105001
Hu J, Wu H, Wang D, Yang Z, Dong J (2019) LncRNA ANRIL promotes NLRP3 inflammasome activation in uric acid nephropathy through miR-122-5p/BRCC3 axis. Biochimie 157:102–110. https://doi.org/10.1016/j.biochi.2018.10.011
Zang Y, Zhou X, Wang Q, Li X, Huang H (2018) LncRNA FIRRE/NF-kB feedback loop contributes to OGD/R injury of cerebral microglial cells. Biochem Biophys Res Commun 501(1):131–138. https://doi.org/10.1016/j.bbrc.2018.04.194
Janni A, Gattuccio F, Porcelli P, Morici V, Gabrielli M, Romano V (1977) Correlation between pituitary gonadotropin response to GnRH and testicular histology in subjects suffering from dyspermia. Acta Eur Fertil 8(2):143–154
Xu Z, Xi K (2019) LncRNA RGMB-AS1 promotes laryngeal squamous cell carcinoma cells progression via sponging miR-22/NLRP3 axis. Biomed Pharmacother 118:109222. https://doi.org/10.1016/j.biopha.2019.109222
Mi L, Min X, Chai Y, Zhang J, Chen X (2022) NLRP1 inflammasomes: a potential target for the treatment of several types of brain injury. Front Immunol 13:863774. https://doi.org/10.3389/fimmu.2022.863774
Vontell RT, de Rivero Vaccari JP, Sun X, Gultekin SH, Bramlett HM, Dietrich WD, Keane RW (2023) Identification of inflammasome signaling proteins in neurons and microglia in early and intermediate stages of Alzheimer’s disease. Brain Pathol 33(4):e13142. https://doi.org/10.1111/bpa.13142
Voet S, Srinivasan S, Lamkanfi M, van Loo G (2019) Inflammasomes in neuroinflammatory and neurodegenerative diseases. Embo Mol Med 11:6. https://doi.org/10.15252/emmm.201810248
Feng X, Zhan F, Luo D, Hu J, Wei G, Hua F, Xu G (2021) LncRNA 4344 promotes NLRP3-related neuroinflammation and cognitive impairment by targeting miR-138-5p. Brain Behav Immun 98:283–298. https://doi.org/10.1016/j.bbi.2021.08.230
Song A, Yang Y, He H, Sun J, Chang Q, Xue Q (2021) Inhibition of long noncoding RNA KCNQ1OT1 attenuates neuroinflammation and neuronal apoptosis through regulating NLRP3 expression via sponging miR-30e-3p. J Inflamm Res 14:1731–1742. https://doi.org/10.2147/JIR.S291274
Gong H, Wan X, Zhang Y, Liang S (2021) Downregulation of HOTAIR reduces neuronal pyroptosis by targeting miR-455-3p/NLRP1 axis in propofol-treated neurons in vitro. Neurochem Res 46(5):1141–1150. https://doi.org/10.1007/s11064-021-03249-6
Guo C, Sun L, Chen X, Zhang D (2013) Oxidative stress, mitochondrial damage and neurodegenerative diseases. Neural Regen Res 8(21):2003–2014. https://doi.org/10.3969/j.issn.1673-5374.2013.21.009
Wang J, Zhao J, Hu P, Gao L, Tian S, He Z (2022) Long noncoding RNA HOTAIR in central nervous system disorders: new insights in pathogenesis, diagnosis, and therapeutic potential. Front Mol Neurosci 15:949095. https://doi.org/10.3389/fnmol.2022.949095
Xiao X, Tan Z, Jia M, Zhou X, Wu K, Ding Y, Li W (2021) Long noncoding RNA SNHG1 knockdown ameliorates apoptosis, oxidative stress and inflammation in models of Parkinson’s disease by inhibiting the miR-125b-5p/MAPK1 axis. Neuropsychiatr Dis Treat 17:1153–1163. https://doi.org/10.2147/NDT.S286778
Lang Y, Zhang H, Yu H, Li Y, Liu X, Li M (2022) Long noncoding RNA myocardial infarction-associated transcript promotes 1-Methyl-4-phenylpyridinium ion-induced neuronal inflammation and oxidative stress in Parkinson’s disease through regulating microRNA-221-3p/ transforming growth factor /nuclear factor E2-related factor 2 axis. Bioengineered 13(1):930–940. https://doi.org/10.1080/21655979.2021.2015527
Zhou S, Zhang D, Guo J, Chen Z, Chen Y, Zhang J (2020) Long noncoding RNA NORAD functions as a microRNA-204-5p sponge to repress the progression of Parkinson’s disease in vitro by increasing the solute carrier family 5 member 3 expression. IUBMB Life 72(9):2045–2055. https://doi.org/10.1002/iub.2344
Kopp F, Elguindy MM, Yalvac ME, Zhang H, Chen B, Gillett FA, Lee S, Sivakumar S, Yu H, Xie Y, Mishra P, Sahenk Z, Mendell JT (2019) PUMILIO hyperactivity drives premature aging of Norad-deficient mice. Elife 8. https://doi.org/10.7554/eLife.42650
Lyu Y, Bai L, Qin C (2019) Long noncoding RNAs in neurodevelopment and Parkinson’s disease. Animal Model Exp Med 2(4):239–251. https://doi.org/10.1002/ame2.12093
Ma X, Wang Y, Yin H, Hua L, Zhang X, Xiao J, Yuan Q, Wang S, Liu Y, Zhang S, Wang Y (2021) Down-regulated long noncoding RNA RMST ameliorates dopaminergic neuron damage in Parkinson’s disease rats via regulation of TLR/NF-kappaB signaling pathway. Brain Res Bull 174:22–30. https://doi.org/10.1016/j.brainresbull.2021.04.026
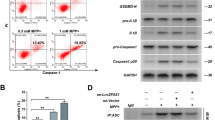
Cai LJ, Tu L, Huang XM, Huang J, Qiu N, Xie GH, Liao JX, Du W, Zhang YY, Tian JY (2020) LncRNA MALAT1 facilitates inflammasome activation via epigenetic suppression of Nrf2 in Parkinson’s disease. Mol Brain 13(1):130. https://doi.org/10.1186/s13041-020-00656-8
Xu X, Zhang Y (2022) Regulation of oxidative stress by long noncoding RNAs in central nervous system disorders. Front Mol Neurosci 15:931704. https://doi.org/10.3389/fnmol.2022.931704
Elkouris M, Kouroupi G, Vourvoukelis A, Papagiannakis N, Kaltezioti V, Matsas R, Stefanis L, Xilouri M, Politis PK (2019) Long noncoding RNAs associated with neurodegeneration-linked genes are reduced in Parkinson’s disease patients. Front Cell Neurosci 13:58. https://doi.org/10.3389/fncel.2019.00058
Ni Y, Huang H, Chen Y, Cao M, Zhou H, Zhang Y (2017) Investigation of long noncoding RNA expression profiles in the substantia nigra of Parkinson’s disease. Cell Mol Neurobiol 37(2):329–338. https://doi.org/10.1007/s10571-016-0373-0
Lun P, Ji T, Wan DH, Liu X, Chen XD, Yu S, Sun P (2022) HOTTIP downregulation reduces neuronal damage and microglial activation in Parkinson’s disease cell and mouse models. Neural Regen Res 17(4):887–897. https://doi.org/10.4103/1673-5374.322475
Zhang Q, Huang XM, Liao JX, Dong YK, Zhu JL, He CC, Huang J, Tang YW, Wu D, Tian JY (2021) LncRNA HOTAIR promotes neuronal damage through facilitating NLRP3 mediated-pyroptosis activation in Parkinson’s disease via regulation of miR-326/ELAVL1 axis. Cell Mol Neurobiol 41(8):1773–1786. https://doi.org/10.1007/s10571-020-00946-8
Sun Q, Zhang Y, Wang S, Yang F, Cai H, Xing Y, Chen Z, Chen J (2021) NEAT1 decreasing suppresses Parkinson’s disease progression via acting as miR-1301-3p sponge. J Mol Neurosci 71(2):369–378. https://doi.org/10.1007/s12031-020-01660-2
He J, Xuan X, Jiang M, Li J, Li N, Nie T (2021) Long noncoding RNA SNHG1 relieves microglia activation by downregulating miR-329-3p expression in an in vitro model of cerebral infarction. Exp Ther Med 22(4):1148. https://doi.org/10.3892/etm.2021.10581
Xu W, Zhang L, Geng Y, Liu Y, Zhang N (2020) Long noncoding RNA GAS5 promotes microglial inflammatory response in Parkinson’s disease by regulating NLRP3 pathway through sponging miR-223-3p. Int Immunopharmacol 85:106614. https://doi.org/10.1016/j.intimp.2020.106614
Zhu Z, Huang P, Sun R, Li X, Li W, Gong W (2022) A novel long-noncoding RNA LncZFAS1 prevents MPP(+)-induced neuroinflammation through MIB1 activation. Mol Neurobiol 59(2):778–799. https://doi.org/10.1007/s12035-021-02619-z
Liu L, Zhou T, Li T, Liang Z, Luo X (2022) LncRNA DLX6-AS1 promotes microglial inflammatory response in Parkinson’s disease by regulating the miR-223-3p/NRP1 axis. Behav Brain Res 431:113923. https://doi.org/10.1016/j.bbr.2022.113923
Lv Q, Wang Z, Zhong Z, Huang W (2020) Role of long noncoding RNAs in Parkinson’s disease: putative biomarkers and therapeutic targets. Parkinsons Dis 2020:5374307. https://doi.org/10.1155/2020/5374307
Wang W, Yang N, Yang YH, Wen R, Liu CF, Zhang TN (2021) Noncoding RNAs: master Regulators of inflammasomes in inflammatory diseases. J Inflamm Res 14:5023–5050. https://doi.org/10.2147/JIR.S332840
Funding
The study was financially supported by the Science & Engineering Research Board and the Council of Scientific and Industrial Research, Govt. of India, and the Council of Scientific and Industrial Research, Govt. of India, for a Junior Research Fellowship. CL acknowledges the Science & Engineering Research Board, Govt. of India, for Extra Mural Research Grant [EMR/2017/002793], the Indian Council of Medical Research, Govt. of India, for Extra Mural Research Grant [File Number 36/14/2020/TOX/BMS], and UGC for Startup Research Grant. This study is supported via funding from Prince Sattam bin Abdulaziz University project number (PSAU/2023/R/1444).
Author information
Authors and Affiliations
Contributions
NS, MPR, HA, and LC developed the concept, designed, and supported in collecting references and writing the manuscript.
Corresponding author
Ethics declarations
Ethics Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
All contributing authors agree to the publication of this article.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• Unraveling the role of lncRNAs in the pathogenesis of Parkinson’s disease
• Significance of inflammasome activation by lncRNAs in Parkinson’s disease.
• Understanding the mechanism of lncRNAs in regulating inflammasome-mediated neuroinflammation in Parkinson’s disease.
• A novel insight on lncRNAs for targeted therapies for Parkinson’s disease.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sivagurunathan, N., Rahamathulla, M.P., Al-Dossary, H. et al. Emerging Role of Long Noncoding RNAs in Regulating Inflammasome-Mediated Neurodegeneration in Parkinson’s Disease. Mol Neurobiol (2023). https://doi.org/10.1007/s12035-023-03809-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12035-023-03809-7