Abstract
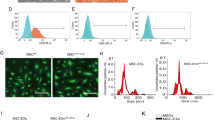
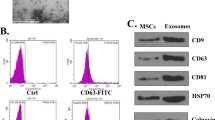
Recent literature has highlighted the therapeutic implication of exosomes (Exos) released by adipose tissue-originated stromal cells (ADSCs) in regenerative medicine. Herein, the current study sought to examine the potential protective effects of ADSC-Exos on neuronal injury following subarachnoid hemorrhage (SAH) by delivering miR-140-5p. Firstly, isolated primary neurons were co-cultured together with well-identified ADSC-Exos. TDP-43-treated neurons were subsequently treated with PKH67-ADSC-Exos and Cy3-miR-140-5p to assess whether ADSC-Exos could transmit miR-140-5p to the recipient neurons to affect their behaviors. Moreover, a luciferase assay was carried out to identify the presumable binding of miR-140-5p to IGFBP5. IGFBP5 rescue experimentation was also performed to testify whether IGFBP5 conferred the impact of miR-140-5p on neuronal damage. The role of PI3K/AKT signaling pathway was further analyzed with the application of its inhibitor miltefosine. Lastly, SAH rat models were developed for in vivo validation. It was found that ADSC-Exos conferred protection against TDP-43-caused neuronal injury by augmenting viability and suppressing cell apoptosis. In addition, miR-140-5p was transmitted from ADSC-Exos to neurons and post-transcriptionally downregulated the expression of IGFBP5. As a result, by means of suppressing IGFBP5 and activating the PI3K/AKT signaling pathway, miR-140-5p from ADSC-Exos induced a neuroprotective effect. Furthermore, in vivo findings substantiated the aforementioned protective role of ADSC-Exos-miR-140-5p, contributing to protection against SAH-caused neurological dysfunction. Collectively, our findings indicated that ADSC-Exos-miR-140-5p could inhibit TDP-43-induced neuronal injury and attenuate neurological dysfunction of SAH rats by inhibiting IGFBP5 and activating the PI3K/Akt signaling pathway.







Similar content being viewed by others
Data Availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- SAH:
-
Subarachnoid hemorrhage
- TDP-43:
-
Transactive response DNA-binding protein 43
- ADSCs:
-
Adipose tissue-derived stromal cells
- IGFBP5:
-
Insulin-like growth factor binding protein 5
- IGF-I:
-
Insulin-like growth factor I
- PI3K:
-
Phosphatidylinositol 3-kinase
- AKT:
-
Protein kinase B
- mTOR:
-
Mammalian target of rapamycin
- HBSS:
-
Hanks’ balanced saline solution
- NC:
-
Negative control
- BSA:
-
Bovine serum albumin
- MAP-2:
-
Microtubule-associated protein 2
- FITC:
-
Fluorescein isothiocyanate
- GAPDH:
-
Glyceraldehyde-3-phosphate dehydrogenase
- ECL:
-
Enhanced chemiluminescence
- FACS:
-
Fluorescence-activated cell sorting
- ANOVA:
-
Analysis of variance
References
Macdonald RL, Schweizer TA (2017) Spontaneous subarachnoid haemorrhage. Lancet 389(10069):655–666. https://doi.org/10.1016/S0140-6736(16)30668-7
Lawton MT, Vates GE (2017) Subarachnoid hemorrhage. N Engl J Med 377(3):257–266. https://doi.org/10.1056/NEJMcp1605827
Macdonald RL (2014) Delayed neurological deterioration after subarachnoid haemorrhage. Nat Rev Neurol 10(1):44–58. https://doi.org/10.1038/nrneurol.2013.246
Sabri M, Lass E, Macdonald RL (2013) Early brain injury: a common mechanism in subarachnoid hemorrhage and global cerebral ischemia. Stroke Res Treat 2013:394036. https://doi.org/10.1155/2013/394036
Schneider UC, Davids AM, Brandenburg S et al (2015) Microglia inflict delayed brain injury after subarachnoid hemorrhage. Acta Neuropathol 130(2):215–231. https://doi.org/10.1007/s00401-015-1440-1
Xia DY, Zhang HS, Wu LY, Zhang XS, Zhou ML, Hang CH (2017) Pentoxifylline alleviates early brain injury after experimental subarachnoid hemorrhage in rats: possibly via inhibiting TLR 4/NF-kappaB signaling pathway. Neurochem Res 42(4):963–974. https://doi.org/10.1007/s11064-016-2129-0
Xie Y, Liu W, Zhang X et al (2015) Human albumin improves long-term behavioral sequelae after subarachnoid hemorrhage through neurovascular remodeling. Crit Care Med 43(10):e440-449. https://doi.org/10.1097/CCM.0000000000001193
Nagata E, Nonaka T, Moriya Y et al (2016) Inositol hexakisphosphate kinase 2 promotes cell death in cells with cytoplasmic TDP-43 aggregation. Mol Neurobiol 53(8):5377–5383. https://doi.org/10.1007/s12035-015-9470-1
Barmada SJ, Skibinski G, Korb E, Rao EJ, Wu JY, Finkbeiner S (2010) Cytoplasmic mislocalization of TDP-43 is toxic to neurons and enhanced by a mutation associated with familial amyotrophic lateral sclerosis. J Neurosci 30(2):639–649. https://doi.org/10.1523/JNEUROSCI.4988-09.2010
He T, Zuo Y, Ai-Zakwani K et al (2018) Subarachnoid hemorrhage enhances the expression of TDP-43 in the brain of experimental rats and human subjects. Exp Ther Med 16(4):3363–3368. https://doi.org/10.3892/etm.2018.6636
Huang T, He D, Kleiner G, Kuluz J (2007) Neuron-like differentiation of adipose-derived stem cells from infant piglets in vitro. J Spinal Cord Med 30(Suppl 1):S35-40. https://doi.org/10.1080/10790268.2007.11753967
Pegtel DM, Gould SJ (2019) Exosomes. Annu Rev Biochem 88:487–514. https://doi.org/10.1146/annurev-biochem-013118-111902
Ma X, Huang M, Zheng M et al (2020) ADSCs-derived extracellular vesicles alleviate neuronal damage, promote neurogenesis and rescue memory loss in mice with Alzheimer’s disease. J Control Release 327:688–702. https://doi.org/10.1016/j.jconrel.2020.09.019
Hwang S, Park SK, Lee HY et al (2014) miR-140-5p suppresses BMP2-mediated osteogenesis in undifferentiated human mesenchymal stem cells. FEBS Lett 588(17):2957–2963. https://doi.org/10.1016/j.febslet.2014.05.048
Wei Z, Qiao S, Zhao J et al (2019) miRNA-181a over-expression in mesenchymal stem cell-derived exosomes influenced inflammatory response after myocardial ischemia-reperfusion injury. Life Sci 232:116632. https://doi.org/10.1016/j.lfs.2019.116632
Wang S, Cui Y, Xu J, Gao H (2019) miR-140-5p attenuates neuroinflammation and brain injury in rats following intracerebral hemorrhage by targeting TLR4. Inflammation 42(5):1869–1877. https://doi.org/10.1007/s10753-019-01049-3
Qiao D, Xu J, Le C et al (2014) Insulin-like growth factor binding protein 5 (IGFBP5) mediates methamphetamine-induced dopaminergic neuron apoptosis. Toxicol Lett 230(3):444–453. https://doi.org/10.1016/j.toxlet.2014.08.010
Yu L, Lu Y, Han X et al (2016) microRNA-140-5p inhibits colorectal cancer invasion and metastasis by targeting ADAMTS5 and IGFBP5. Stem Cell Res Ther 7(1):180. https://doi.org/10.1186/s13287-016-0438-5
Tardif G, Hum D, Pelletier JP, Duval N, Martel-Pelletier J (2009) Regulation of the IGFBP-5 and MMP-13 genes by the microRNAs miR-140 and miR-27a in human osteoarthritic chondrocytes. BMC Musculoskelet Disord 10:148. https://doi.org/10.1186/1471-2474-10-148
Chen K, Hou J, Song Y et al (2018) Chi-miR-3031 regulates beta-casein via the PI3K/AKT-mTOR signaling pathway in goat mammary epithelial cells (GMECs). BMC Vet Res 14(1):369. https://doi.org/10.1186/s12917-018-1695-6
Qin X, Yu S, Zhou L et al (2017) Cisplatin-resistant lung cancer cell-derived exosomes increase cisplatin resistance of recipient cells in exosomal miR-100-5p-dependent manner. Int J Nanomedicine 12:3721–3733. https://doi.org/10.2147/IJN.S131516
van Balkom BW, de Jong OG, Smits M et al (2013) Endothelial cells require miR-214 to secrete exosomes that suppress senescence and induce angiogenesis in human and mouse endothelial cells. Blood 121(19):3997-4006-S39915-3915. https://doi.org/10.1182/blood-2013-02-478925
Chen X, Jiang M, Li H et al (2020) CX3CL1/CX3CR1 axis attenuates early brain injury via promoting the delivery of exosomal microRNA-124 from neuron to microglia after subarachnoid hemorrhage. J Neuroinflammation 17(1):209. https://doi.org/10.1186/s12974-020-01882-6
Chen D, Wang X, Huang J, Cui S, Zhang L (2020) CDKN1B mediates apoptosis of neuronal cells and inflammation induced by oxyhemoglobin via miR-502-5p after subarachnoid hemorrhage. J Mol Neurosci 70(7):1073–1080. https://doi.org/10.1007/s12031-020-01512-z
Li Y, Wang J, Li Z et al (2019) Propoxyphene mediates oxyhemoglobin-induced injury in rat cortical neurons through up-regulation of active-beta-catenin. Front Pharmacol 10:1616. https://doi.org/10.3389/fphar.2019.01616
Tsai AC, Pan SL, Liao CH et al (2010) Moscatilin, a bibenzyl derivative from the India orchid Dendrobium loddigesii, suppresses tumor angiogenesis and growth in vitro and in vivo. Cancer Lett 292(2):163–170. https://doi.org/10.1016/j.canlet.2009.11.020
Sugawara T, Ayer R, Jadhav V, Zhang JH (2008) A new grading system evaluating bleeding scale in filament perforation subarachnoid hemorrhage rat model. J Neurosci Methods 167(2):327–334. https://doi.org/10.1016/j.jneumeth.2007.08.004
Liu F, Chen Y, Hu Q et al (2015) MFGE8/integrin beta3 pathway alleviates apoptosis and inflammation in early brain injury after subarachnoid hemorrhage in rats. Exp Neurol 272:120–127. https://doi.org/10.1016/j.expneurol.2015.04.016
Trabolsi C, Takash Chamoun W, Hijazi A, Nicoletti C, Maresca M, Nasser M (2021) Study of neuroprotection by a combination of the biological antioxidant (eucalyptus extract) and the antihypertensive drug candesartan against chronic cerebral ischemia in rats. Molecules 26(4):839. https://doi.org/10.3390/molecules26040839
Zhu Q, Enkhjargal B, Huang L et al (2018) Aggf1 attenuates neuroinflammation and BBB disruption via PI3K/Akt/NF-kappaB pathway after subarachnoid hemorrhage in rats. J Neuroinflammation 15(1):178. https://doi.org/10.1186/s12974-018-1211-8
Drommelschmidt K, Serdar M, Bendix I et al (2017) Mesenchymal stem cell-derived extracellular vesicles ameliorate inflammation-induced preterm brain injury. Brain Behav Immun 60:220–232. https://doi.org/10.1016/j.bbi.2016.11.011
Yan BC, Wang J, Rui Y et al (2019) Neuroprotective effects of gabapentin against cerebral ischemia reperfusion-induced neuronal autophagic injury via regulation of the PI3K/Akt/mTOR signaling pathways. J Neuropathol Exp Neurol 78(2):157–171. https://doi.org/10.1093/jnen/nly119
Peroni D, Scambi I, Pasini A et al (2008) Stem molecular signature of adipose-derived stromal cells. Exp Cell Res 314(3):603–615. https://doi.org/10.1016/j.yexcr.2007.10.007
Xin H, Li Y, Cui Y, Yang JJ, Zhang ZG, Chopp M (2013) Systemic administration of exosomes released from mesenchymal stromal cells promote functional recovery and neurovascular plasticity after stroke in rats. J Cereb Blood Flow Metab 33(11):1711–1715. https://doi.org/10.1038/jcbfm.2013.152
Bonafede R, Scambi I, Peroni D et al (2016) Exosome derived from murine adipose-derived stromal cells: neuroprotective effect on in vitro model of amyotrophic lateral sclerosis. Exp Cell Res 340(1):150–158. https://doi.org/10.1016/j.yexcr.2015.12.009
Lee M, Liu T, Im W, Kim M (2016) Exosomes from adipose-derived stem cells ameliorate phenotype of Huntington’s disease in vitro model. Eur J Neurosci 44(4):2114–2119. https://doi.org/10.1111/ejn.13275
Tao SC, Yuan T, Zhang YL, Yin WJ, Guo SC, Zhang CQ (2017) Exosomes derived from miR-140-5p-overexpressing human synovial mesenchymal stem cells enhance cartilage tissue regeneration and prevent osteoarthritis of the knee in a rat model. Theranostics 7(1):180–195. https://doi.org/10.7150/thno.17133
Geng W, Tang H, Luo S et al (2019) Exosomes from miRNA-126-modified ADSCs promotes functional recovery after stroke in rats by improving neurogenesis and suppressing microglia activation. Am J Transl Res 11(2):780–792
Samuel W, Kutty RK, Vijayasarathy C, Pascual I, Duncan T, Redmond TM (2010) Decreased expression of insulin-like growth factor binding protein-5 during N-(4-hydroxyphenyl)retinamide-induced neuronal differentiation of ARPE-19 human retinal pigment epithelial cells: regulation by CCAAT/enhancer-binding protein. J Cell Physiol 224(3):827–836. https://doi.org/10.1002/jcp.22191
Funding
This study is supported by the National Natural Science Foundation of China (81870944; 81771233); the Specific Research Projects for Capital Health Development (2018–2-2041); the Beijing Science and Technology Planning Project (Z181100009618035); the Beijing Municipal Administration of Hospitals’ Ascent Plan (DFL20190501); and the Research and Promotion Program of Appropriate Techniques for Intervention of Chinese High-Risk Stroke People (GN-2020R0007).
Author information
Authors and Affiliations
Contributions
Pinyan Wang and Yanan Xue designed the study. Yuchun Zuo and Yinan Xue collated the data, Pinyan Wang, John H Zhang, and Jiajia Duan carried out data analyses and produced the initial draft of the manuscript. Fei Liu and Aihua Liu contributed to drafting the manuscript. All authors have read and approved the final submitted manuscript.
Corresponding authors
Ethics declarations
Ethics Approval
Animal experimentation conformed to the animal ethics protocol ratified by Animal Ethics Committee of Central South University (No: 2019sydw0153). Extensive efforts were undertaken to minimize both the numbers and the respective suffering of the experimental animals.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, P., Xue, Y., Zuo, Y. et al. Exosome-Encapsulated microRNA-140-5p Alleviates Neuronal Injury Following Subarachnoid Hemorrhage by Regulating IGFBP5-Mediated PI3K/AKT Signaling Pathway. Mol Neurobiol 59, 7212–7228 (2022). https://doi.org/10.1007/s12035-022-03007-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-022-03007-x