Abstract
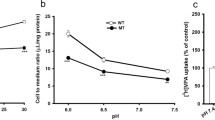
Amyotrophic lateral sclerosis (ALS) is a progressive motor neuron disease caused by the death of the neurons regulating the voluntary muscles which leads to the progressive paralysis. We investigated the difference of transport function of L-citrulline in ALS disease model (NSC-34/hSOD1G93A, MT) and a control model (NSC-34/hSOD1wt, WT). The [14C]L-citrulline uptake was significantly reduced in MT cells as compared with that of control. The Michaelis-Menten constant (Km) for MT cells was 0.67 ± 0.05 mM, whereas it was 1.48 ± 0.21 mM for control. On the other hand, the Vmax values for MT and control were 10.9 ± 0.8 nmol/mg protein/min and 18.3 ± 2.9 nmol/mg protein/min, respectively. The Km and Vmax values showed the high affinity and low capacity for MT as compared with control. Moreover, the uptake of [14C]L-citrulline was significantly inhibited by 2-aminobicyclo-(2,2,1)-heptane-2-carboxylic acid (BCH) and harmaline which is the inhibitor of the large neutral amino acid transporter1 (LAT1) in NSC-34 cell lines. Furthermore, [14C]L-citrulline uptakes took place in Na+-independent manner. It was also inhibited by the neutral amino acids such as citrulline and phenylalanine. Likewise, L-dopa, gabapentin, and riluzole significantly inhibited the [14C]L-citrulline uptake. It shows the competitive inhibition for L-dopa in ALS cell lines. On the other hand, [14C]L-citrulline uptake in the presence of riluzole showed competitive inhibition in WT cells, whereas it was uncompetitive for MT cells. The small interfering RNA experiments showed that LAT1 is involved in the [14C]L-citrulline uptake in NSC-34 cell lines. On the other hand, in the examination of the alteration in the expression level of LAT1, it was significantly lower in MT cells as compared with that of control. Similarly, in the spinal cord of ALS, transgenic mice revealed a slight but significant decrease in LAT1 immunoreactivity in motor neurons of ALS mice compared with control. However, the LAT1 immunoreactivity in non-motor neurons and in astrocytes was relatively increased in the spinal cord gray matter of ALS mice. The experimental evidences of our results suggest that the change of transport activity of [14C]L-citrulline may be partially responsible for the pathological alteration in ALS.





Similar content being viewed by others
Abbreviations
- ALS:
-
Amyotrophic lateral sclerosis
- NSC-34 cells:
-
Motor neuron–like cells
- BCH:
-
2-aminobicyclo-(2,2,1)-heptane-2-carboxylic acid
- ECF:
-
Extracellular fluid
- MeAIB:
-
α-(methylamino) isobutyric acid
- siRNA:
-
Small interfering RNA
- mRNA:
-
Messenger RNA
- MT:
-
Mutant type
- WT:
-
Wild type
- ECF:
-
Extracellular fluid
References
Luiking YC, Engelen MP, Deutz NE (2010) Regulation of nitric oxide production in health and disease. Curr Opin Clin Nutr Metab Care 13(1):97–104
Rimando AM, Perkins-Veazie PM (2005) Determination of citrulline in watermelon rind. J Chromatogr A 1078(1–2):196–200
Olsen I, Singhrao SK, Potempa JJ (2018) Citrullination as a plausible link to periodontitis, rheumatoid arthritis, atherosclerosis and Alzheimer's disease. J Oral Microbiol 10(1):1487742
Allerton TD, Proctor DN, Stephens JM, Dugas T, Spielmann G, Irving B (2018) L-Citrulline supplementation: impact on cardiometabolic health. Nutrients 10(7):921
Bahri S, Zerrouk N, Aussel C, Moinard C, Crenn P, Curis E, Chaumeil JC, Cynober L et al (2013) Citrulline: from metabolism to therapeutic use. Nutrition 29(3):479–484
Lameu C, Trujillo CA, Schwindt TT, Negraes PD, Pillat MM, Morais KL, Lebrun I, Ulrich H (2012) Interactions between the NO-citrulline cycle and brain-derived neurotrophic factor in differentiation of neural stem cells. J Biol Chem 287(35):29690–29701
Liu C, Liang MC, Soong TW (2019) Nitric oxide, iron and neurodegeneration. Front Neurosci 13:114
Moinard C, Maccario J, Walrand S, Lasserre V, Marc J, Boirie Y, Cynober L (2016) Arginine behaviour after arginine or citrulline administration in older subjects. Br J Nutr 115(3):399–404
Gubert F, Bonacossa-Pereira I, Decotelli AB, Furtado M, Vasconcelos-dos-Santos A, Mendez-Otero R, Santiago MF (2019) Bone-marrow mononuclear cell therapy in a mouse model of amyotrophic lateral sclerosis: functional outcomes from different administration routes. Brain Res 1712:73–81
Renton AE, Chiò A, Traynor BJ (2014) State of play in amyotrophic lateral sclerosis genetics. Nat Neurosci 17(1):17–23
Stuerenburg HJ, Stangneth B, Schoser BG (2006) Age related profiles of free amino acids in human skeletal muscle. Neuro Endocrinol Lett 27(1–2):133–136
Sau D, De Biasi S, Vitellaro-Zuccarello L et al (2007) Mutation of SOD1 in ALS: a gain of a loss of function. Hum Mol Genet 16(13):1604–1618
Lee J, Ryu H, Kowall NW (2009) Motor neuronal protection by l-arginine prolongs survival of mutant SOD1 (G93A) ALS mice. Biochem Biophys Res Commun 384(4):524–529
Liu D, Bao F, Wen J, Liu J (2007) Mutation of superoxide dismutase elevates reactive species: comparison of nitration and oxidation of proteins in different brain regions of transgenic mice with amyotrophic lateral sclerosis. Neuroscience 146(1):255–264
Yabuki Y, Shioda N, Yamamoto Y, Shigano M, Kumagai K, Morita M, Fukunaga K (2013) Oral L-Citrulline administration improves memory deficits following transient brain ischemia through cerebrovascular protection. Brain Res 1520:157–167
Lee KE, Kang YS (2017) Characteristics of L-citrulline transport through blood-brain barrier in the brain capillary endothelial cell line (TR-BBB cells). J Biomed Sci 24:28
Lee J, Ryu H, Kowall NW (2009) Differential regulation of neuronal and inducible nitric oxide synthase (NOS) in the spinal cord of mutant SOD1 (G93A) ALS mice. Biochem Biophys Res Commun 387(1):202–206
Gyawali A, Kang YS (2019) Blood-to-retina transport of imperatorin involves the carrier-mediated transporter system at the inner blood-retinal barrier. J Pharm Sci 108(4):1619–1626
Lee NY, Kim Y, Ryu H, Kang YS (2017) The alteration of serine transporter activity in a cell line model of amyotrophic lateral sclerosis (ALS). Biochem Biophys Res Commun 483(1):135–141
Kang YS, Ohtsuki S, Takanaga H, Tomi M, Hosoya KI, Terasaki T (2002) Regulation of taurine transport at the blood–brain barrier by tumor necrosis factor-alpha, taurine and hypertonicity. J Neurochem 83(5):1188–1195
Gyawali A, Krol S, Kang YS (2019) Involvement of a novel organic cation transporter in paeonol transport across the blood-brain barrier. Biomol Ther 27(3):290–301
Lee NY, Kang YS (2016) In vivo and in vitro evidence for brain uptake of 4-phenylbutyrate by the monocarboxylate transporter 1 (MCT1). Pharm Res 33(7):1711–1722
Ryu H, Lee J, Hagerty SW, Soh BY, McAlpin SE, Cormier KA, Smith KM, Ferrante RJ (2006) ESET/SETDB1 gene expression and histone H3 (K9) trimethylation in Huntington’s disease. Proc Natl Acad Sci U S A 103(50):19176–19181
Jung MK, Kim KY, Lee NY, Kang YS, Hwang YJ, Kim Y, Sung JJ, McKee A et al (2013) Expression of taurine transporter (TauT) is modulated by heat shock factor 1 (HSF1) in motor neurons of ALS. Mol Neurobiol 47(2):699–710
Sasabe J, Chiba T, Yamada M, Okamoto K, Nishimoto I, Matsuoka M, Aiso S (2007) D-serine is a key determinant of glutamate toxicity in amyotrophic lateral sclerosis. EMBO J 26(18):4149–4159
Sasabe J, Miyoshi Y, Suzuki M et al (2012) D-amino acid oxidase controls motoneuron degeneration through D serine. Proc Natl Acad Sci U S A 109(2):627–632
O’Kane RL, Viña JR, Simpson I et al (2006) Cationic amino acid transport across the blood–brain barrier is mediated exclusively by system y+. Am J Physiol Endocrinol Metab 291(2):412–419
Zhan X, Graf WM (2012) Harmaline attenuates voltage--sensitive Ca (2+) currents in neurons of the inferior olive. J Pharm Sci 15(5):657–668
Pingitore P, Pochini L, Scalise M, Galluccio M, Hedfalk K, Indiveri C (2013) Large scale production of the active human ASCT2 (SLC1A5) transporter in Pichia pastoris — functional and kinetic asymmetry revealed in proteoliposomes. Biochim Biophys Acta 1828(9):2238–2246
Fukasawa Y, Segawa H, Kim JY, Chairoungdua A, Kim DK, Matsuo H, Cha SH, Endou H et al (2000) Identification and characterization of a Na(+)-independent neutral amino acid transporter that associates with the 4F2 heavy chain and exhibits substrate selectivity for small neutral D- and L-amino acids. J Biol Chem 275(13):9690–9698
Devés R, Boyd CA (1998) Transporters for cationic amino acids in animal cells: discovery, structure, and function. Physiol Rev 78(2):487–545
Boado RJ, Li JY, Nagaya M et al (1999) Selective expression of the large neutral amino acid transporter at the blood–brain barrier. Proc Natl Acad Sci U S A 96(21):12079–12084
Matsuo H, Tsukada S, Nakata T, Chairoungdua A, Kim DK, Cha SH, Inatomi J, Yorifuji H et al (2000) Expression of a system L neutral amino acid transporter at the blood–brain barrier. Neuroreport 11(16):3507–3511
Dorheim MA, Tracey WR, Pollock JS, Grammas P (1994) Nitric oxide synthase activity is elevated in brain microvessels in Alzheimer’s disease. Biochem Biophys Res Commun 205(1):659–665
Skene DJ, Middleton B, Fraser CK, Pennings JL et al (2017) Metabolic profiling of presymptomatic Huntington’s disease sheep reveals novel biomarkers. Sci Rep 7:43030
Chiang MC, Chen HM, Lee YH, Chang HH, Wu YC, Soong BW, Chen CM, Wu YR et al (2007) Dysregulation of C/EBPα by mutant Huntingtin causes the urea cycle deficiency in Huntington's disease. Hum Mol Genet 16:483–498
Larsen MS, Frølund S, Nøhr MK, Nielsen CU, Garmer M, Kreilgaard M, Holm R (2015) In vivo and in vitro evaluations of intestinal gabapentin absorption: effect of dose and inhibitors on carrier-mediated transport. Pharm Res 32(3):898–909
Wakayama K (1987) Enhancement of the muscle relaxant action of baclofen by glucocorticoids. Nihon Yakurigaku Zasshi 89(2):81–90
Mora JS, Genge A, Chio A et al (2019) Masitinib as an add-on therapy to riluzole in patients with amyotrophic lateral sclerosis: a randomized clinical trial. Amyotroph Lateral Scler Frontotemporal Degener 20:1–10
Collo G, Cavalleri L, Bono F et al (2018) Ropinirole and pramipexole promote structural plasticity in human ipsc-derived dopaminergic neurons via BDNF and mTOR signaling. Neural Plast 2018:15
Lee J, Hyeon SJ, Im HJ, Ryu H, Kim Y, Ryu H (2016) Astrocytes and microglia as non-cell autonomous players in the pathogenesis of ALS. Exp Neurobiol 25(5):233–240
Acknowledgments
We are thankful towards Phuong T. Nguyen and Junghee Lee for performing the in vivo experiments and contributing in data analysis of the same.
Funding
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (No. 2019R1F1A1044048) (YSK) and Grants from Korea Institute of Science and Technology (2E30320) and NRF-2018M3C7A1056894 (HR).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The experiments were carried out in accordance with the Health Guide for the Care and Use of Laboratory Animals and were approved by the Animal Care Committees of Korea Institute of Science and Technology (KIST). Procedures were performed in accordance with Guide for the Care and Use of Laboratory Animals guidelines and were approved by both the Veterans Administration and Boston University animal care committees.
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gyawali, A., Gautam, S., Hyeon, S.J. et al. L-Citrulline Level and Transporter Activity Are Altered in Experimental Models of Amyotrophic Lateral Sclerosis. Mol Neurobiol 58, 647–657 (2021). https://doi.org/10.1007/s12035-020-02143-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-020-02143-6