Abstract
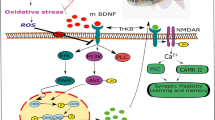
Melanin-concentrating hormone (MCH) is a highly conserved neuropeptide known to exhibit important functions in the brain. Some studies have reported that MCH improves memory by promoting memory retention. However, the precise molecular mechanisms by which MCH enhances memory impairment have yet to be fully elucidated. In this study, MCH was administered to the scopolamine-induced memory-impaired mice via the nasal cavity to examine the acute effects of MCH and Alzheimer’s disease (AD) mouse models to evaluate the chronic effects of MCH. MCH improved memory impairment in both models and reduced soluble amyloid beta in the cerebral cortex of APP/PS1 transgenic mice. In vitro assays also showed that MCH inhibits amyloid beta-induced cytotoxicity. Furthermore, MCH increased long-term potentiation (LTP) in the hippocampus of wild-type and 5XFAD AD mouse model. To further elucidate the mechanisms of the chronic effect of MCH, the levels of phosphorylated CREB and GSK3β, and the expression of BDNF, TrkB and PSD95 were examined in the cerebral cortex and hippocampus. Our findings indicate that MCH might have neuroprotective effects via downstream pathways associated with the enhancement of neuronal synapses and LTP. This suggests a therapeutic potential of MCH for the treatment of neurodegenerative diseases such as AD.







Similar content being viewed by others
References
Nahon JL (1994) The melanin-concentrating hormone: from the peptide to the gene. Crit Rev Neurobiol 8:221–262
Griffond B, Baker BI (2002) Cell and molecular cell biology of melanin-concentrating hormone. Int Rev Cytol 213:233–277
Bittencourt JC, Presse F, Arias C, Peto C, Vaughan J, Nahon JL, Vale W, Sawchenko PE (1992) The melanin-concentrating hormone system of the rat brain: an immuno- and hybridization histochemical characterization. J Comp Neurol 319:218–245. https://doi.org/10.1002/cne.903190204
Balleine BW, Delgado MR, Hikosaka O (2007) The role of the dorsal striatum in reward and decision-making. J Neurosci 27:8161–8165. https://doi.org/10.1523/JNEUROSCI.1554-07.2007
Yin HH, Knowlton BJ (2006) The role of the basal ganglia in habit formation. Nat Rev Neurosci 7:464–476
Adamantidis A, Thomas E, Foidart A, Tyhon A, Coumans B, Minet A, Tirelli E, Seutin V et al (2005) Disrupting the melanin-concentrating hormone receptor 1 in mice leads to cognitive deficits and alterations of NMDA receptor function. Eur J Neurosci 21:2837–2844. https://doi.org/10.1111/j.1460-9568.2005.04100.x
Monzon ME, De Souza MM, Izquierdo LA et al (1999) Melanin-concentrating hormone (MCH) modifies memory retention in rats. Peptides 20:1517–1519. https://doi.org/10.1016/S0196-9781(99)00164-3
Varas M, Pérez M, Monzón ME, De Barioglio SR (2002) Melanin-concentrating hormone, hippocampal nitric oxide levels and memory retention. Peptides 23:2213–2221. https://doi.org/10.1016/S0196-9781(02)00252-8
Varas M, Pérez M, Ramírez O, De Barioglio SR (2002) Melanin concentrating hormone increase hippocampal synaptic transmission in the rat. Peptides 23:151–155. https://doi.org/10.1016/S0196-9781(01)00591-5
Varas MM, Pérez MF, Ramírez OA, De Barioglio SR (2003) Increased susceptibility to LTP generation and changes in NMDA-NR1 and -NR2B subunits mRNA expression in rat hippocampus after MCH administration. Peptides 24:1403–1411. https://doi.org/10.1016/j.peptides.2003.09.006
Tan CP, Sano H, Iwaasa H, Pan J, Sailer AW, Hreniuk DL, Feighner SD, Palyha OC et al (2002) Melanin-concentrating hormone receptor subtypes 1 and 2: species-specific gene expression. Genomics 79:785–792. https://doi.org/10.1006/geno.2002.6771
Borowsky B, Durkin MM, Ogozalek K, Marzabadi MR, DeLeon J, Heurich R, Lichtblau H, Shaposhnik Z et al (2002) Antidepressant, anxiolytic and anorectic effects of a melanin-concentrating hormone-1 receptor antagonist. Nat Med 8:825–830. https://doi.org/10.1038/nm741
Hervieu GJ, Cluderay JE, Harrison D, Meakin J, Maycox P, Nasir S, Leslie RA (2000) The distribution of the mRNA and protein products of the melanin-concentrating hormone (MCH) receptor gene, slc-1, in the central nervous system of the rat. Eur J Neurosci 12:1194–1216. https://doi.org/10.1046/j.1460-9568.2000.00008.x
Saito Y, Cheng M, Leslie FM, Civelli O (2001) Expression of the melanin-concentrating hormone (MCH) receptor mRNA in the rat brain. J Comp Neurol 435:26–40. https://doi.org/10.1002/cne.1191
Park JY, Kim SN, Yoo J, Jang J, Lee A, Oh JY, Kim H, Oh ST et al (2017) Novel neuroprotective effects of melanin-concentrating hormone in Parkinson’s disease. Mol Neurobiol 54:7706–7721. https://doi.org/10.1007/s12035-016-0258-8
Schmidt FM, Kratzsch J, Gertz H-J, Tittmann M, Jahn I, Pietsch UC, Kaisers UX, Thiery J et al (2013) Cerebrospinal fluid melanin-concentrating hormone (MCH) and hypocretin-1 (HCRT-1, orexin-a) in Alzheimer’s disease. PLoS One 8:e63136. https://doi.org/10.1371/journal.pone.0063136
Aziz A, Fronczek R, Maat-Schieman M, Unmehopa U, Roelandse F, Overeem S, van Duinen S, Lammers GJ et al (2008) Hypocretin and melanin-concentrating hormone in patients with Huntington disease. Brain Pathol 18:474–483. https://doi.org/10.1111/j.1750-3639.2008.00135.x
Chauhan NB, Siegel GJ, Lichtor T (2001) Distribution of intraventricularly administered antiamyloid-beta peptide (Aβ) antibody in the mouse brain. J Neurosci Res 66:231–235. https://doi.org/10.1002/jnr.1215
Klinkenberg I, Blokland A (2010 Jul) The validity of scopolamine as a pharmacological model for cognitive impairment: a review of animal behavioral studies. Neurosci Biobehav Rev 34(8):1307–1350
Ogura H, Kosasa T, Kuriya Y, Yamanishi Y (2000) Donepezil, a centrally acting acetylcholinesterase inhibitor, alleviates learning deficits in hypocholinergic models in rats. Methods Find Exp Clin Model Pharmacol 22:89–95
Adamantidis A, de Lecea L (2009) A role for melanin-concentrating hormone in learning and memory. Peptides 30:2066–2070. https://doi.org/10.1016/j.peptides.2009.06.024
Le Barillier L, Léger L, Luppi PH et al (2015) Genetic deletion of melanin-concentrating hormone neurons impairs hippocampal short-term synaptic plasticity and hippocampal-dependent forms of short-term memory. Hippocampus 25:1361–1373. https://doi.org/10.1002/hipo.22442
Haider S, Tabassum S, Perveen T (2016) Scopolamine induced greater alterations in neurochemical profile and increased oxidative stress demonstrated a better model of dementia: a comparative study. Brain Res Bull 127:234–247
Cammarota M, Levi De Stein M, Paratcha G et al (2000) Rapid and transient learning-associated increase in NMDA NR1 subunit in the rat hippocampus. Neurochem Res 25:567–572. https://doi.org/10.1023/A:1007590415556
Patterson SL, Abel T, Deuel TAS, Martin KC, Rose JC, Kandel ER (1996) Recombinant BDNF rescues deficits in basal synaptic transmission and hippocampal LTP in BDNF knockout mice. Neuron 16:1137–1145. https://doi.org/10.1016/S0896-6273(00)80140-3
Minichiello L, Korte M, Wolfer D, Kühn R, Unsicker K, Cestari V, Rossi-Arnaud C, Lipp HP et al (1999) Essential role for TrkB receptors in hippocampus-mediated learning. Neuron 24:401–414. https://doi.org/10.1016/S0896-6273(00)80853-3
Zhang P, Lisman JE (2012) Activity-dependent regulation of synaptic strength by PSD-95 in CA1 neurons. J Neurophysiol 107:1058–1066. https://doi.org/10.1152/jn.00526.2011
Slutsky I, Abumaria N, Wu LJ, Huang C, Zhang L, Li B, Zhao X, Govindarajan A et al (2010) Enhancement of learning and memory by elevating brain magnesium. Neuron 65:165–177. https://doi.org/10.1016/j.neuron.2009.12.026
Adams JP, Sweatt JD (2002) Molecular psychology: roles for the ERK MAP kinase cascade in memory. Annu Rev Pharmacol Toxicol 42:135–163. https://doi.org/10.1146/annurev.pharmtox.42.082701.145401
Carlezon WA Jr, Duman RS, Nestler EJ (2005) The many faces of CREB. Trends Neurosci 28:436–445
Lonze BE, Ginty DD (2002) Function and regulation of CREB family transcription factors in the nervous system. Neuron 35:605–623
Morens C, Nørregaard P, Receveur JM, van Dijk G, Scheurink AJW (2005) Effects of MCH and a MCH1-receptor antagonist on (palatable) food and water intake. Brain Res 1062:32–38. https://doi.org/10.1016/j.brainres.2005.09.005
Torterolo P, Lagos P, Monti JM (2011) Melanin-concentrating hormone: a new sleep factor? Front Neurol 2:14. https://doi.org/10.3389/fneur.2011.00014
Jang JH, Park JY, Oh JY, Bae SJ, Jang H, Jeon S, Kim J, Park HJ (2018) Novel analgesic effects of melanin-concentrating hormone on persistent neuropathic and inflammatory pain in mice. Sci Rep 8:707. https://doi.org/10.1038/s41598-018-19145-z
Ma C, Yin Z, Zhu P, Luo J, Shi X, Gao X (2017) Blood cholesterol in late-life and cognitive decline: a longitudinal study of the Chinese elderly. Mol Neurodegener 12(24). https://doi.org/10.1186/s13024-017-0167-y
Funding
This research was supported by grants from the National Research Foundation (2017R1A2B4009963) and the Korean Health Technology R&D Project founded by the Ministry of Health and Welfare (HI16C0405, HI18C1077) of the Republic of Korea.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
All animal experiments in this study followed the “Guide for Animal Experiments” provided by the Korean Academy of Medical Sciences. All animal protocols were approved by the Institutional Animal Care and Use Committee of Dongguk University, South Korea (IACUC-2016-1193), and Chonnam National University Medical School (CNU IACUC-H-2018-1).
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Oh, S.T., Liu, Q.F., Jeong, H.J. et al. Nasal Cavity Administration of Melanin-Concentrating Hormone Improves Memory Impairment in Memory-Impaired and Alzheimer’s Disease Mouse Models. Mol Neurobiol 56, 8076–8086 (2019). https://doi.org/10.1007/s12035-019-01662-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-019-01662-1