Abstract
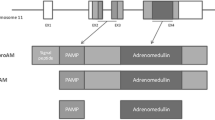
Alzheimer’s disease (AD) is characterized by the loss of synaptic contacts caused in part by cytoskeleton disruption. Adrenomedullin (AM) is involved in physiological functions such as vasodilation, hormone secretion, antimicrobial activity, cellular growth, and angiogenesis. In neurons, AM and related peptides are associated with some structural and functional cytoskeletal proteins, causing microtubule destabilization. Here, we describe the relationships between AM and other signs of AD in clinical specimens. Frontal cortex from AD patients and controls were studied for AM, acetylated tubulin, NCAM, Ox-42, and neurotransmitters. AM was increased in AD compared with controls, while levels of acetylated tubulin, NCAM, and neurotransmitters were decreased. Interestingly, increases in AM statistically correlated with the decrease in these markers. Furthermore, Ox42 overexpression in AD correlated with levels of AM. It is proposed that AD patients may have neural cytoskeleton failure associated with increase of AM levels, resulting in axon transport collapse and synaptic loss. These observations suggest that reducing AM expression may constitute a new avenue to prevent/treat AD.




Similar content being viewed by others
References
Hernandez F, Lucas JJ, Avila J (2013) GSK3 and tau: two convergence points in Alzheimer’s disease. J Alzheimers Dis 33(Suppl 1):S141–S144
Querfurth HW, LaFerla FM (2010) Alzheimer’s disease. N Engl J Med 362:329–344
Buerger K, Uspenskaya O, Hartmann O et al (2011) Prediction of Alzheimer’s disease using midregional proadrenomedullin and midregional proatrial natriuretic peptide: a retrospective analysis of 134 patients with mild cognitive impairment. J Clin Psychiatry 72:556–563
Henriksen K, O’Bryant SE, Hampel H et al (2014) The future of blood-based biomarkers for Alzheimer’s disease. Alzheimers Dement 10:115–131
Pérez-Castells J, Martín-Santamaría S, Nieto L et al (2012) Structure of micelle-bound adrenomedullin: a first step toward the analysis of its interactions with receptors and small molecules. Biopolymers 97:45–53
López J, Martínez A (2002) Cell and molecular biology of the multifunctional peptide, adrenomedullin. Int Rev Cytol 221:1–92
Serrano J, Uttenthal LO, Martínez A et al (2000) Distribution of adrenomedullin-like immunoreactivity in the rat central nervous system by light and electron microscopy. Brain Res 853:245–268
Xu Y, Krukoff TL (2004) Adrenomedullin in the rostral ventrolateral medulla increases arterial pressure and heart rate: roles of glutamate and nitric oxide. Am J Physiol Regul Integr Comp Physiol 287:R729–R734
Sackett DL, Ozbun L, Zudaire E et al (2008) Intracellular proadrenomedullin-derived peptides decorate the microtubules and contribute to cytoskeleton function. Endocrinology 149:2888–2898
Larráyoz IM, Martínez A (2012) Proadrenomedullin N-terminal 20 peptide increases kinesin’s velocity both in vitro and in vivo. Endocrinology 153:1734–1742
Vergaño-Vera E, Fernández AP, Hurtado-Chong A et al (2010) Lack of adrenomedullin affects growth and differentiation of adult neural stem/progenitor cells. Cell Tissue Res 340:1–11
Fernandez AP, Masa JS, Guedan MA et al (2016) Adrenomedullin expression in Alzheimer’s brain. Curr Alzheimer Res 13:428–438
Kirvell SL, Esiri M, Francis PT (2006) Down-regulation of vesicular glutamate transporters precedes cell loss and pathology in Alzheimer’s disease. J Neurochem 98:939–950
Aisa B, Gil-Bea FJ, Solas M et al (2010) Altered NCAM expression associated with the cholinergic system in Alzheimer’s disease. J Alzheimers Dis 20:659–668
Garcia-Alloza M, Gil-Bea FJ, Diez-Ariza M et al (2005) Cholinergic-serotonergic imbalance contributes to cognitive and behavioral symptoms in Alzheimer’s disease. Neuropsychologia 43:442–449
Morgenthaler NG, Struck J, Alonso C, Bergmann A (2005) Measurement of midregional proadrenomedullin in plasma with an immunoluminometric assay. Clin Chem 51:1823–1829
Strzyz P (2016) Post-translational modifications: extension of the tubulin code. Nat Rev Mol Cell Biol 17:609
Mungas D (1991) In-office mental status testing: a practical guide. Geriatrics 46:54–58 63, 66
Vu HT, Akatsu H, Hashizume Y et al (2017) Increase in α-tubulin modifications in the neuronal processes of hippocampal neurons in both kainic acid-induced epileptic seizure and Alzheimer’s disease. Sci Rep 7:40205
Rønn LC, Berezin V, Bock E The neural cell adhesion molecule in synaptic plasticity and ageing. Int J Dev Neurosci 18:193–199
Kiss JZ, Muller D (2001) Contribution of the neural cell adhesion molecule to neuronal and synaptic plasticity. Rev Neurosci 12:297–310
Walmod PS, Kolkova K, Berezin V, Bock E (2004) Zippers make signals: NCAM-mediated molecular interactions and signal transduction. Neurochem Res 29:2015–2035
Kleene R, Schachner M (2004) Glycans and neural cell interactions. Nat Rev Neurosci 5:195–208
Montag-Sallaz M, Schachner M, Montag D (2002) Misguided axonal projections, neural cell adhesion molecule 180 mRNA upregulation, and altered behavior in mice deficient for the close homolog of L1. Mol Cell Biol 22:7967–7981
Aisa B, Elizalde N, Tordera R et al (2009) Effects of neonatal stress on markers of synaptic plasticity in the hippocampus: implications for spatial memory. Hippocampus 19:1222–1231
Arendt T (2009) Synaptic degeneration in Alzheimer’s disease. Acta Neuropathol 118:167–179
Mikkonen M, Soininen H, Tapiola T et al (1999) Hippocampal plasticity in Alzheimer’s disease: changes in highly polysialylated NCAM immunoreactivity in the hippocampal formation. Eur J Neurosci 11:1754–1764
Mikkonen M, Soininen H, Alafuzof I, Miettinen R (2001) Hippocampal plasticity in Alzheimer’s disease. Rev Neurosci 12:311–325
Gillian AM, Brion JP, Breen KC (1994) Expression of the neural cell adhesion molecule (NCAM) in Alzheimer’s disease. Neurodegeneration 3:283–291
Jin K, Peel AL, Mao XO et al (2004) Increased hippocampal neurogenesis in Alzheimer’s disease. Proc Natl Acad Sci 101:343–347
Strekalova H, Buhmann C, Kleene R et al (2006) Elevated levels of neural recognition molecule L1 in the cerebrospinal fluid of patients with Alzheimer disease and other dementia syndromes. Neurobiol Aging 27:1–9
Todaro L, Puricelli L, Gioseffi H et al (2004) Neural cell adhesion molecule in human serum. Increased levels in dementia of the Alzheimer type. Neurobiol Dis 15:387–393
Yew DT, Li WP, Webb SE et al (1999) Neurotransmitters, peptides, and neural cell adhesion molecules in the cortices of normal elderly humans and Alzheimer patients: a comparison. Exp Gerontol 34:117–133
Calsolaro V, Edison P (2016) Neuroinflammation in Alzheimer’s disease: Current evidence and future directions. Alzheimers Dement 12:719–732
Francis PT (2003) Glutamatergic systems in Alzheimer’s disease. Int J Geriatr Psychiatry 18:S15–S21
Francis PT, Ramírez MJ, Lai MK (2010) Neurochemical basis for symptomatic treatment of Alzheimer’s disease. Neuropharmacology 59:221–229
Braak H, Braak E (1991) Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol 82:239–259
Garcia-Alloza M, Tsang SW, Gil-Bea FJ et al (2006) Involvement of the GABAergic system in depressive symptoms of Alzheimer’s disease. Neurobiol Aging 27:1110–1117
Cheung BMY, Ong KL, Tso AWK et al (2011) Plasma adrenomedullin level is related to a single nucleotide polymorphism in the adrenomedullin gene. Eur J Endocrinol 165:571–577
Martínez-Herrero S, Martínez A (2013) Cancer protection elicited by a single nucleotide polymorphism close to the adrenomedullin gene. J Clin Endocrinol Metab 98:E807–E810
Martínez A, Weaver C, López J et al (1996) Regulation of insulin secretion and blood glucose metabolism by adrenomedullin. Endocrinology 137:2626–2632
Ishikawa T, Chen J, Wang J et al (2003) Adrenomedullin antagonist suppresses in vivo growth of human pancreatic cancer cells in SCID mice by suppressing angiogenesis. Oncogene 22:1238–1242
Martínez A, Julián M, Bregonzio C et al (2004) Identification of vasoactive nonpeptidic positive and negative modulators of adrenomedullin using a neutralizing antibody-based screening strategy. Endocrinology 145:3858–3865
Roldós V, Carbajo RJ, Schott A-K et al (2012) Identification of first proadrenomedullin N-terminal 20 peptide (PAMP) modulator by means of virtual screening and NMR interaction experiments. Eur J Med Chem 55:262–272
Hickman RA, Faustin A, Wisniewski T (2016) Alzheimer disease and its growing epidemic: risk factors, biomarkers, and the urgent need for therapeutics. Neurol Clin 34:941–953
Barbe C, Morrone I, Novella JL et al (2016) Predictive factors of rapid cognitive decline in patients with Alzheimer disease. Dement Geriatr Cogn Dis Extra 6:549–558
Acknowledgements
H.F. is a recipient of a fellowship from Ministerio de Educación y Ciencia (FPU). I.M.L. and A.M. are funded by Fundación Rioja Salud (FRS).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Ferrero, H., Larrayoz, I.M., Martisova, E. et al. Increased Levels of Brain Adrenomedullin in the Neuropathology of Alzheimer’s Disease. Mol Neurobiol 55, 5177–5183 (2018). https://doi.org/10.1007/s12035-017-0700-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-017-0700-6