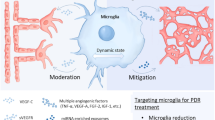
Abstract
Microglia are immune cells in the central nervous system (CNS) that originate from the yolk sac in an embryo. The renewal of the microglia pool in the adult eye consists of two components. In addition to the self-proliferation of resident cells, microglia in the CNS also derive from the bone marrow (BM). BM-derived cells pass through the blood–brain barrier (BBB) or blood–retina barrier (BRB) and differentiate into microglia under specific conditions which involves a complex mechanism. Recent studies have widely investigated the role of resident microglia and BM-derived microglia in the retinal degenerative disease. Both two cell types play dual roles and share many similar functions. However, resident microglia tend to polarize to the M1 phenotype which is pro-inflammatory and neurotoxic, whereas BM-derived microglia mainly polarize to the neuroprotective M2 phenotype in retinal degeneration. The molecular mechanism that underlines the invasion of peripheral cells has led to extensive discussions. In addition to the BBB and BRB disruption, many signaling pathways are involved in this process. Based on these studies, we discuss the roles of these two types of microglia in retinal degeneration disease and the potential clinical application of BM-derived microglia, which may benefit future therapies.


Similar content being viewed by others
Abbreviations
- Aβ:
-
Amyloid β
- AD:
-
Alzheimer’s disease
- AMD:
-
Age-related macular degeneration
- BBB:
-
Blood–brain barrier
- BCECs:
-
Brain capillary endothelial cells
- BM:
-
Bone marrow
- BRB:
-
Blood–retina barrier
- CNS:
-
Central nervous system
- CNV:
-
Choroidal neovascularization
- DR:
-
Diabetic retinopathy
- EMP:
-
Erythromyeloid precursors
- GCL:
-
Ganglion cell layer
- HIF1α:
-
Hypoxia inducible factor 1 alpha
- HSC:
-
Hematopoietic stem cells
- IGF-1:
-
Insulin-like growth factor-1
- IPL:
-
Inner plexiform layer
- MCP:
-
Monocyte chemoattractant protein
- MHC:
-
Major histocompatibility complex
- MIP:
-
Macrophage inflammatory protein
- MMP:
-
Matrix metalloproteinases
- NFL:
-
Nerve fiber layer
- NV:
-
Neovascularization
- ONL:
-
Outer nuclear layer
- OPL:
-
Outer plexiform layer
- RD:
-
Retinal degeneration
- ROS:
-
Reactive oxygen species
- RP:
-
Retinitis pigmentosa
- RPE:
-
Retinal pigment epithelium
- SDF-1:
-
Stromal-derived factor-1
- VEGF:
-
Vascular endothelial growth factor
- YS:
-
Yolk sac
- 7KCh:
-
7-Ketocholesterol
References
Fu R, Shen Q, Xu P, Luo JJ, Tang Y (2014) Phagocytosis of microglia in the central nervous system diseases. Mol Neurobiol 49(3):1422–1434. doi:10.1007/s12035-013-8620-6
Alliot F, Godin I, Pessac B (1999) Microglia derive from progenitors, originating from the yolk sac, and which proliferate in the brain. Brain Res Dev Brain Res 117(2):145–152
Lampron A, Elali A, Rivest S (2013) Innate immunity in the CNS: redefining the relationship between the CNS and Its environment. Neuron 78(2):214–232. doi:10.1016/j.neuron.2013.04.005
Kettenmann H, Hanisch UK, Noda M, Verkhratsky A (2011) Physiology of microglia. Physiol Rev 91(2):461–553. doi:10.1152/physrev.00011.2010
Boche D, Perry VH, Nicoll JA (2013) Review: activation patterns of microglia and their identification in the human brain. Neuropathol Appl Neurobiol 39(1):3–18. doi:10.1111/nan.12011
Kaur C, Rathnasamy G, Ling EA (2013) Roles of activated microglia in hypoxia induced neuroinflammation in the developing brain and the retina. J Neuroimmune Pharmacol 8(1):66–78. doi:10.1007/s11481-012-9347-2
Amantea D, Micieli G, Tassorelli C, Cuartero MI, Ballesteros I, Certo M, Moro MA, Lizasoain I et al (2015) Rational modulation of the innate immune system for neuroprotection in ischemic stroke. Front Neurosci 9:147. doi:10.3389/fnins.2015.00147
Taylor RA, Sansing LH (2013) Microglial responses after ischemic stroke and intracerebral hemorrhage. Clin Dev Immunol 2013:746068. doi:10.1155/2013/746068
Ginhoux F, Prinz M (2015) Origin of microglia: current concepts and past controversies. Cold Spring Harb Perspect Biol 7(8):a020537. doi:10.1101/cshperspect.a020537
Kierdorf K, Erny D, Goldmann T, Sander V, Schulz C, Perdiguero EG, Wieghofer P, Heinrich A et al (2013) Microglia emerge from erythromyeloid precursors via Pu.1- and Irf8-dependent pathways. Nat Neurosci 16(3):273–280. doi:10.1038/nn.3318
Hanisch UK, Kettenmann H (2007) Microglia: active sensor and versatile effector cells in the normal and pathologic brain. Nat Neurosci 10(11):1387–1394. doi:10.1038/nn1997
Bruttger J, Karram K, Wortge S, Regen T, Marini F, Hoppmann N, Klein M, Blank T et al (2015) Genetic cell ablation reveals clusters of local self-renewing microglia in the mammalian central nervous system. Immunity 43(1):92–106. doi:10.1016/j.immuni.2015.06.012
Ajami B, Bennett JL, Krieger C, Tetzlaff W, Rossi FM (2007) Local self-renewal can sustain CNS microglia maintenance and function throughout adult life. Nat Neurosci 10(12):1538–1543. doi:10.1038/nn2014
Simard AR, Soulet D, Gowing G, Julien JP, Rivest S (2006) Bone marrow-derived microglia play a critical role in restricting senile plaque formation in Alzheimer’s disease. Neuron 49(4):489–502. doi:10.1016/j.neuron.2006.01.022
Sasahara M, Otani A, Oishi A, Kojima H, Yodoi Y, Kameda T, Nakamura H, Yoshimura N (2008) Activation of bone marrow-derived microglia promotes photoreceptor survival in inherited retinal degeneration. Am J Pathol 172(6):1693–1703. doi:10.2353/ajpath.2008.080024
Yang Y, Shiao C, Hemingway JF, Jorstad NL, Shalloway BR, Chang R, Keene CD (2013) Suppressed retinal degeneration in aged wild type and APPswe/PS1DeltaE9 mice by bone marrow transplantation. PLoS One 8(6):e64246. doi:10.1371/journal.pone.0064246
Mildner A, Schmidt H, Nitsche M, Merkler D, Hanisch UK, Mack M, Heikenwalder M, Bruck W et al (2007) Microglia in the adult brain arise from Ly-6ChiCCR2+ monocytes only under defined host conditions. Nat Neurosci 10(12):1544–1553. doi:10.1038/nn2015
Lawson LJ, Perry VH, Gordon S (1992) Turnover of resident microglia in the normal adult mouse brain. Neuroscience 48(2):405–415
Tanaka R, Komine-Kobayashi M, Mochizuki H, Yamada M, Furuya T, Migita M, Shimada T, Mizuno Y et al (2003) Migration of enhanced green fluorescent protein expressing bone marrow-derived microglia/macrophage into the mouse brain following permanent focal ischemia. Neuroscience 117(3):531–539. doi:10.1016/S0306-4522(02)00954-5
Ajami B, Bennett JL, Krieger C, McNagny KM, Rossi FM (2011) Infiltrating monocytes trigger EAE progression, but do not contribute to the resident microglia pool. Nat Neurosci 14(9):1142–1149. doi:10.1038/nn.2887
Kaneko H, Nishiguchi KM, Nakamura M, Kachi S, Terasaki H (2008) Characteristics of bone marrow-derived microglia in the normal and injured retina. Invest Ophthalmol Vis Sci 49(9):4162–4168. doi:10.1167/iovs.08-1738
Ginhoux F, Greter M, Leboeuf M, Nandi S, See P, Gokhan S, Mehler MF, Conway SJ et al (2010) Fate mapping analysis reveals that adult microglia derive from primitive macrophages. Science 330(6005):841–845. doi:10.1126/science.1194637
Hoeffel G, Chen J, Lavin Y, Low D, Almeida FF, See P, Beaudin AE, Lum J et al (2015) C-Myb(+) erythro-myeloid progenitor-derived fetal monocytes give rise to adult tissue-resident macrophages. Immunity 42(4):665–678. doi:10.1016/j.immuni.2015.03.011
Saijo K, Glass CK (2011) Microglial cell origin and phenotypes in health and disease. Nat Rev Immunol 11(11):775–787. doi:10.1038/nri3086
Bertrand JY, Jalil A, Klaine M, Jung S, Cumano A, Godin I (2005) Three pathways to mature macrophages in the early mouse yolk sac. Blood 106(9):3004–3011. doi:10.1182/blood-2005-02-0461
Solomon JN, Lewis CA, Ajami B, Corbel SY, Rossi FM, Krieger C (2006) Origin and distribution of bone marrow-derived cells in the central nervous system in a mouse model of amyotrophic lateral sclerosis. Glia 53(7):744–753. doi:10.1002/glia.20331
Daneman R, Zhou L, Kebede AA, Barres BA (2010) Pericytes are required for blood–brain barrier integrity during embryogenesis. Nature 468(7323):562–566. doi:10.1038/nature09513
Diaz-Araya CM, Provis JM, Penfold PL, Billson FA (1995) Development of microglial topography in human retina. J Comp Neurol 363(1):53–68. doi:10.1002/cne.903630106
Chen L, Yang P, Kijlstra A (2002) Distribution, markers, and functions of retinal microglia. Ocul Immunol Inflamm 10(1):27–39
Provis JM, Diaz CM, Penfold PL (1996) Microglia in human retina: a heterogeneous population with distinct ontogenies. Perspect Dev Neurobiol 3(3):213–222
Santos AM, Calvente R, Tassi M, Carrasco MC, Martin-Oliva D, Marin-Teva JL, Navascues J, Cuadros MA (2008) Embryonic and postnatal development of microglial cells in the mouse retina. J Comp Neurol 506(2):224–239. doi:10.1002/cne.21538
Navascues J, Moujahid A, Almendros A, Marin-Teva JL, Cuadros MA (1995) Origin of microglia in the quail retina: central-to-peripheral and vitreal-to-scleral migration of microglial precursors during development. J Comp Neurol 354(2):209–228. doi:10.1002/cne.903540205
Yuan TF, Liang YX, Peng B, Lin B, So KF (2015) Local proliferation is the main source of rod microglia after optic nerve transection. Sci Rep 5:10788. doi:10.1038/srep10788
Varvel NH, Grathwohl SA, Baumann F, Liebig C, Bosch A, Brawek B, Thal DR, Charo IF et al (2012) Microglial repopulation model reveals a robust homeostatic process for replacing CNS myeloid cells. Proc Natl Acad Sci U S A 109(44):18150–18155. doi:10.1073/pnas.1210150109
Lebson L, Nash K, Kamath S, Herber D, Carty N, Lee DC, Li Q, Szekeres K et al (2010) Trafficking CD11b-positive blood cells deliver therapeutic genes to the brain of amyloid-depositing transgenic mice. J Neurosci 30(29):9651–9658. doi:10.1523/JNEUROSCI.0329-10.2010
Prinz M, Priller J, Sisodia SS, Ransohoff RM (2011) Heterogeneity of CNS myeloid cells and their roles in neurodegeneration. Nat Neurosci 14(10):1227–1235. doi:10.1038/nn.2923
Simard AR, Rivest S (2004) Bone marrow stem cells have the ability to populate the entire central nervous system into fully differentiated parenchymal microglia. FASEB J 18(9):998–1000. doi:10.1096/fj.04-1517fje
Okonogi N, Nakamura K, Suzuki Y, Suto N, Suzue K, Kaminuma T, Nakano T, Hirai H (2014) Cranial irradiation induces bone marrow-derived microglia in adult mouse brain tissue. J Radiat Res 55(4):713–719. doi:10.1093/jrr/rru015
Priller J, Flugel A, Wehner T, Boentert M, Haas CA, Prinz M, Fernandez-Klett F, Prass K et al (2001) Targeting gene-modified hematopoietic cells to the central nervous system: use of green fluorescent protein uncovers microglial engraftment. Nat Med 7(12):1356–1361. doi:10.1038/nm1201-1356
Keene CD, Chang RC, Lopez-Yglesias AH, Shalloway BR, Sokal I, Li X, Reed PJ, Keene LM et al (2010) Suppressed accumulation of cerebral amyloid {beta} peptides in aged transgenic Alzheimer’s disease mice by transplantation with wild-type or prostaglandin E2 receptor subtype 2-null bone marrow. Am J Pathol 177(1):346–354. doi:10.2353/ajpath.2010.090840
Jack CS, Arbour N, Blain M, Meier UC, Prat A, Antel JP (2007) Th1 polarization of CD4+ T cells by Toll-like receptor 3-activated human microglia. J Neuropathol Exp Neurol 66(9):848–859. doi:10.1097/nen.0b013e3181492a7
Mildner A, Mack M, Schmidt H, Bruck W, Djukic M, Zabel MD, Hille A, Priller J et al (2009) CCR2 + Ly-6Chi monocytes are crucial for the effector phase of autoimmunity in the central nervous system. Brain 132(Pt 9):2487–2500. doi:10.1093/brain/awp144
Yang LP, Sun HL, Wu LM, Guo XJ, Dou HL, Tso MO, Zhao L, Li SM (2009) Baicalein reduces inflammatory process in a rodent model of diabetic retinopathy. Invest Ophthalmol Vis Sci 50(5):2319–2327. doi:10.1167/iovs.08-2642
Shi G, Chen S, Wandu WS, Ogbeifun O, Nugent LF, Maminishkis A, Hinshaw SJ, Rodriguez IR et al (2015) Inflammasomes induced by 7-ketocholesterol and other stimuli in RPE and in bone marrow-derived cells differ markedly in their production of IL-1beta and IL-18. Invest Ophthalmol Vis Sci 56(3):1658–1664. doi:10.1167/iovs.14-14557
Wang H, Shi H, Zhang J, Wang G, Zhang J, Jiang F, Xiao Q (2014) Toll-like receptor 4 in bone marrow-derived cells contributes to the progression of diabetic retinopathy. Mediat Inflamm 2014:858763. doi:10.1155/2014/858763
Qin L, Liu Y, Cooper C, Liu B, Wilson B, Hong JS (2002) Microglia enhance beta-amyloid peptide-induced toxicity in cortical and mesencephalic neurons by producing reactive oxygen species. J Neurochem 83(4):973–983
Song M, Jue SS, Cho YA, Kim EC (2015) Comparison of the effects of human dental pulp stem cells and human bone marrow-derived mesenchymal stem cells on ischemic human astrocytes in vitro. J Neurosci Res 93(6):973–983. doi:10.1002/jnr.23569
Gupta N, Brown KE, Milam AH (2003) Activated microglia in human retinitis pigmentosa, late-onset retinal degeneration, and age-related macular degeneration. Exp Eye Res 76(4):463–471
Kokovay E, Cunningham LA (2005) Bone marrow-derived microglia contribute to the neuroinflammatory response and express iNOS in the MPTP mouse model of Parkinson’s disease. Neurobiol Dis 19(3):471–478. doi:10.1016/j.nbd.2005.01.023
Joly S, Francke M, Ulbricht E, Beck S, Seeliger M, Hirrlinger P, Hirrlinger J, Lang KS et al (2009) Cooperative phagocytes: resident microglia and bone marrow immigrants remove dead photoreceptors in retinal lesions. Am J Pathol 174(6):2310–2323. doi:10.2353/ajpath.2009.090023
Kaur C, Sivakumar V, Ang LS, Sundaresan A (2006) Hypoxic damage to the periventricular white matter in neonatal brain: role of vascular endothelial growth factor, nitric oxide and excitotoxicity. J Neurochem 98(4):1200–1216. doi:10.1111/j.1471-4159.2006.03964.x
Zhao L, Zabel MK, Wang X, Ma W, Shah P, Fariss RN, Qian H, Parkhurst CN et al (2015) Microglial phagocytosis of living photoreceptors contributes to inherited retinal degeneration. EMBO Mol Med 7(9):1179–1197. doi:10.15252/emmm.201505298
Brown GC, Neher JJ (2014) Microglial phagocytosis of live neurons. Nat Rev Neurosci 15(4):209–216. doi:10.1038/nrn3710
Koronyo Y, Salumbides BC, Sheyn J, Pelissier L, Li S, Ljubimov V, Moyseyev M, Daley D et al (2015) Therapeutic effects of glatiramer acetate and grafted CD115(+) monocytes in a mouse model of Alzheimer’s disease. Brain 138(Pt 8):2399–2422. doi:10.1093/brain/awv150
Indaram M, Ma W, Zhao L, Fariss RN, Rodriguez IR, Wong WT (2015) 7-Ketocholesterol increases retinal microglial migration, activation, and angiogenicity: a potential pathogenic mechanism underlying age-related macular degeneration. Sci Rep 5:9144. doi:10.1038/srep09144
Fukuda S, Nagano M, Yamashita T, Kimura K, Tsuboi I, Salazar G, Ueno S, Kondo M et al (2013) Functional endothelial progenitor cells selectively recruit neurovascular protective monocyte-derived F4/80(+)/Ly6c(+) macrophages in a mouse model of retinal degeneration. Stem Cells 31(10):2149–2161. doi:10.1002/stem.1469
Marneros AG (2013) NLRP3 inflammasome blockade inhibits VEGF-A-induced age-related macular degeneration. Cell Rep 4(5):945–958. doi:10.1016/j.celrep.2013.08.002
Caicedo A, Espinosa-Heidmann DG, Pina Y, Hernandez EP, Cousins SW (2005) Blood-derived macrophages infiltrate the retina and activate Muller glial cells under experimental choroidal neovascularization. Exp Eye Res 81(1):38–47. doi:10.1016/j.exer.2005.01.013
Ishikawa K, Yoshida S, Nakao S, Sassa Y, Asato R, Kohno R, Arima M, Kita T et al (2012) Bone marrow-derived monocyte lineage cells recruited by MIP-1beta promote physiological revascularization in mouse model of oxygen-induced retinopathy. Lab Investig 92(1):91–101. doi:10.1038/labinvest.2011.141
Combadiere C, Feumi C, Raoul W, Keller N, Rodero M, Pezard A, Lavalette S, Houssier M et al (2007) CX3CR1-dependent subretinal microglia cell accumulation is associated with cardinal features of age-related macular degeneration. J Clin Invest 117(10):2920–2928. doi:10.1172/JCI31692
Sennlaub F, Auvynet C, Calippe B, Lavalette S, Poupel L, Hu SJ, Dominguez E, Camelo S et al (2013) CCR2(+) monocytes infiltrate atrophic lesions in age-related macular disease and mediate photoreceptor degeneration in experimental subretinal inflammation in Cx3cr1 deficient mice. EMBO Mol Med 5(11):1775–1793. doi:10.1002/emmm.201302692
Li L, Eter N, Heiduschka P (2015) The microglia in healthy and diseased retina. Exp Eye Res 136:116–130. doi:10.1016/j.exer.2015.04.020
Rymo SF, Gerhardt H, Wolfhagen Sand F, Lang R, Uv A, Betsholtz C (2011) A two-way communication between microglial cells and angiogenic sprouts regulates angiogenesis in aortic ring cultures. PLoS One 6(1), e15846. doi:10.1371/journal.pone.0015846
Lee JE, Liang KJ, Fariss RN, Wong WT (2008) Ex vivo dynamic imaging of retinal microglia using time-lapse confocal microscopy. Invest Ophthalmol Vis Sci 49(9):4169–4176. doi:10.1167/iovs.08-2076
Kezic JM, McMenamin PG (2013) The effects of CX3CR1 deficiency and irradiation on the homing of monocyte-derived cell populations in the mouse eye. PLoS One 8(7):e68570. doi:10.1371/journal.pone.0068570
Chen M, Zhao J, Luo C, Pandi SP, Penalva RG, Fitzgerald DC, Xu H (2012) Para-inflammation-mediated retinal recruitment of bone marrow-derived myeloid cells following whole-body irradiation is CCL2 dependent. Glia 60(5):833–842. doi:10.1002/glia.22315
Pierce EA (2001) Pathways to photoreceptor cell death in inherited retinal degenerations. Bioessays 23(7):605–618. doi:10.1002/bies.1086
Hartong DT, Berson EL, Dryja TP (2006) Retinitis pigmentosa. Lancet 368(9549):1795–1809. doi:10.1016/S0140-6736(06)69740-7
Noailles A, Fernandez-Sanchez L, Lax P, Cuenca N (2014) Microglia activation in a model of retinal degeneration and TUDCA neuroprotective effects. J Neuroinflammation 11:186. doi:10.1186/s12974-014-0186-3
Yoshida N, Ikeda Y, Notomi S, Ishikawa K, Murakami Y, Hisatomi T, Enaida H, Ishibashi T (2013) Laboratory evidence of sustained chronic inflammatory reaction in retinitis pigmentosa. Ophthalmology 120(1):e5–e12. doi:10.1016/j.ophtha.2012.07.008
Iezzi R, Guru BR, Glybina IV, Mishra MK, Kennedy A, Kannan RM (2012) Dendrimer-based targeted intravitreal therapy for sustained attenuation of neuroinflammation in retinal degeneration. Biomaterials 33(3):979–988. doi:10.1016/j.biomaterials.2011.10.010
Zeng H, Ding M, Chen XX, Lu Q (2014) Microglial NADPH oxidase activation mediates rod cell death in the retinal degeneration in rd mice. Neuroscience 275:54–61. doi:10.1016/j.neuroscience.2014.05.065
Zhou WT, Ni YQ, Jin ZB, Zhang M, Wu JH, Zhu Y, Xu GZ, Gan DK (2012) Electrical stimulation ameliorates light-induced photoreceptor degeneration in vitro via suppressing the proinflammatory effect of microglia and enhancing the neurotrophic potential of Muller cells. Exp Neurol 238(2):192–208. doi:10.1016/j.expneurol.2012.08.029
Kohno H, Chen Y, Kevany BM, Pearlman E, Miyagi M, Maeda T, Palczewski K, Maeda A (2013) Photoreceptor proteins initiate microglial activation via Toll-like receptor 4 in retinal degeneration mediated by all-trans-retinal. J Biol Chem 288(21):15326–15341. doi:10.1074/jbc.M112.448712
Peng B, Xiao J, Wang K, So KF, Tipoe GL, Lin B (2014) Suppression of microglial activation is neuroprotective in a mouse model of human retinitis pigmentosa. J Neurosci 34(24):8139–8150. doi:10.1523/JNEUROSCI.5200-13.2014
Kohno H, Maeda T, Perusek L, Pearlman E, Maeda A (2014) CCL3 production by microglial cells modulates disease severity in murine models of retinal degeneration. J Immunol 192(8):3816–3827. doi:10.4049/jimmunol.1301738
Gehrs KM, Anderson DH, Johnson LV, Hageman GS (2006) Age-related macular degeneration--emerging pathogenetic and therapeutic concepts. Ann Med 38(7):450–471. doi:10.1080/07853890600946724
Ambati J, Fowler BJ (2012) Mechanisms of age-related macular degeneration. Neuron 75(1):26–39. doi:10.1016/j.neuron.2012.06.018
Kinnunen K, Petrovski G, Moe MC, Berta A, Kaarniranta K (2012) Molecular mechanisms of retinal pigment epithelium damage and development of age-related macular degeneration. Acta Ophthalmol 90(4):299–309. doi:10.1111/j.1755-3768.2011.02179.x
Thurman JM, Renner B, Kunchithapautham K, Ferreira VP, Pangburn MK, Ablonczy Z, Tomlinson S, Holers VM et al (2009) Oxidative stress renders retinal pigment epithelial cells susceptible to complement-mediated injury. J Biol Chem 284(25):16939–16947. doi:10.1074/jbc.M808166200
Noel A, Jost M, Lambert V, Lecomte J, Rakic JM (2007) Anti-angiogenic therapy of exudative age-related macular degeneration: current progress and emerging concepts. Trends Mol Med 13(8):345–352. doi:10.1016/j.molmed.2007.06.005
Doyle SL, Adamson P, Lopez FJ, Humphries P, Campbell M (2014) Reply to IL-18 is not therapeutic for neovascular age-related macular degeneration. Nat Med 20(12):1376–1377. doi:10.1038/nm.3741
Doyle SL, Ozaki E, Brennan K, Humphries MM, Mulfaul K, Keaney J, Kenna PF, Maminishkis A et al (2014) IL-18 attenuates experimental choroidal neovascularization as a potential therapy for wet age-related macular degeneration. Sci Transl Med 6(230):230ra244. doi:10.1126/scitranslmed.3007616
Koronyo-Hamaoui M, Koronyo Y, Ljubimov AV, Miller CA, Ko MK, Black KL, Schwartz M, Farkas DL (2011) Identification of amyloid plaques in retinas from Alzheimer’s patients and noninvasive in vivo optical imaging of retinal plaques in a mouse model. Neuroimage 54(Suppl 1):S204–S217. doi:10.1016/j.neuroimage.2010.06.020
Iseri PK, Altinas O, Tokay T, Yuksel N (2006) Relationship between cognitive impairment and retinal morphological and visual functional abnormalities in Alzheimer disease. J Neuroophthalmol 26(1):18–24. doi:10.1097/01.wno.0000204645.56873.26
Gao L, Liu Y, Li X, Bai Q, Liu P (2015) Abnormal retinal nerve fiber layer thickness and macula lutea in patients with mild cognitive impairment and Alzheimer’s disease. Arch Gerontol Geriatr 60(1):162–167. doi:10.1016/j.archger.2014.10.011
Blanks JC, Hinton DR, Sadun AA, Miller CA (1989) Retinal ganglion cell degeneration in Alzheimer’s disease. Brain Res 501(2):364–372
Danesh-Meyer HV, Birch H, Ku JY, Carroll S, Gamble G (2006) Reduction of optic nerve fibers in patients with Alzheimer disease identified by laser imaging. Neurology 67(10):1852–1854. doi:10.1212/01.wnl.0000244490.07925.8b
Meda L, Cassatella MA, Szendrei GI, Otvos L Jr, Baron P, Villalba M, Ferrari D, Rossi F (1995) Activation of microglial cells by beta-amyloid protein and interferon-gamma. Nature 374(6523):647–650. doi:10.1038/374647a0
Dong N, Chang L, Wang B, Chu L (2014) Retinal neuronal MCP-1 induced by AGEs stimulates TNF-alpha expression in rat microglia via p38, ERK, and NF-kappaB pathways. Mol Vis 20:616–628
Sun D, Chen J, Bao X, Cai Y, Zhao J, Huang J, Huang W, Fan X et al (2015) Protection of radial glial-like cells in the hippocampus of APP/PS1 mice: a novel mechanism of memantine in the treatment of Alzheimer’s disease. Mol Neurobiol 52(1):464–477. doi:10.1007/s12035-014-8875-6
Gao L, Chen X, Tang Y, Zhao J, Li Q, Fan X, Xu H, Yin ZQ (2015) Neuroprotective effect of memantine on the retinal ganglion cells of APPswe/PS1DeltaE9 mice and its immunomodulatory mechanisms. Exp Eye Res 135:47–58. doi:10.1016/j.exer.2015.04.013
Malm TM, Koistinaho M, Parepalo M, Vatanen T, Ooka A, Karlsson S, Koistinaho J (2005) Bone-marrow-derived cells contribute to the recruitment of microglial cells in response to beta-amyloid deposition in APP/PS1 double transgenic Alzheimer mice. Neurobiol Dis 18(1):134–142. doi:10.1016/j.nbd.2004.09.009
Cheung N, Mitchell P, Wong TY (2010) Diabetic retinopathy. Lancet 376(9735):124–136. doi:10.1016/S0140-6736(09)62124-3
Grossniklaus HE, Kang SJ, Berglin L (2010) Animal models of choroidal and retinal neovascularization. Prog Retin Eye Res 29(6):500–519. doi:10.1016/j.preteyeres.2010.05.003
Madeira MH, Boia R, Santos PF, Ambrosio AF, Santiago AR (2015) Contribution of microglia-mediated neuroinflammation to retinal degenerative diseases. Mediat Inflamm 2015:673090. doi:10.1155/2015/673090
Kern TS (2007) Contributions of inflammatory processes to the development of the early stages of diabetic retinopathy. Exp Diabetes Res 2007:95103. doi:10.1155/2007/95103
Rangasamy S, McGuire PG, Franco Nitta C, Monickaraj F, Oruganti SR, Das A (2014) Chemokine mediated monocyte trafficking into the retina: role of inflammation in alteration of the blood-retinal barrier in diabetic retinopathy. PLoS One 9(10):e108508. doi:10.1371/journal.pone.0108508
Zeng HY, Green WR, Tso MO (2008) Microglial activation in human diabetic retinopathy. Arch Ophthalmol 126(2):227–232. doi:10.1001/archophthalmol.2007.65
Zeng XX, Ng YK, Ling EA (2000) Neuronal and microglial response in the retina of streptozotocin-induced diabetic rats. Vis Neurosci 17(3):463–471
Rungger-Brandle E, Dosso AA, Leuenberger PM (2000) Glial reactivity, an early feature of diabetic retinopathy. Invest Ophthalmol Vis Sci 41(7):1971–1980
Gaucher D, Chiappore JA, Paques M, Simonutti M, Boitard C, Sahel JA, Massin P, Picaud S (2007) Microglial changes occur without neural cell death in diabetic retinopathy. Vis Res 47(5):612–623. doi:10.1016/j.visres.2006.11.017
Grigsby JG, Cardona SM, Pouw CE, Muniz A, Mendiola AS, Tsin AT, Allen DM, Cardona AE (2014) The role of microglia in diabetic retinopathy. J Ophthalmol 2014:705783. doi:10.1155/2014/705783
Ritter MR, Banin E, Moreno SK, Aguilar E, Dorrell MI, Friedlander M (2006) Myeloid progenitors differentiate into microglia and promote vascular repair in a model of ischemic retinopathy. J Clin Invest 116(12):3266–3276. doi:10.1172/JCI29683
Ataka K, Asakawa A, Nagaishi K, Kaimoto K, Sawada A, Hayakawa Y, Tatezawa R, Inui A et al (2013) Bone marrow-derived microglia infiltrate into the paraventricular nucleus of chronic psychological stress-loaded mice. PLoS One 8(11):e81744. doi:10.1371/journal.pone.0081744
Sawada A, Niiyama Y, Ataka K, Nagaishi K, Yamakage M, Fujimiya M (2014) Suppression of bone marrow-derived microglia in the amygdala improves anxiety-like behavior induced by chronic partial sciatic nerve ligation in mice. Pain 155(9):1762–1772. doi:10.1016/j.pain.2014.05.031
Igarashi T, Miyake K, Hayakawa J, Kawabata K, Ishizaki M, Takahashi H, Shimada T (2007) Apoptotic cell death and regeneration in the newborn retina after irradiation prior to bone marrow transplantation. Curr Eye Res 32(6):543–553. doi:10.1080/02713680701389333
Akkus Yildirim B, Cetin E, Topkan E, Ozyigit G, Cengiz M, Surucu S, Usubutun A, Akyol F (2015) Prevention of radiation-induced retinopathy with amifostine in wistar albino rats. Retina 35(7):1458–1464. doi:10.1097/IAE.0000000000000493
Muther PS, Semkova I, Schmidt K, Abari E, Kuebbeler M, Beyer M, Abken H, Meyer KL et al (2010) Conditions of retinal glial and inflammatory cell activation after irradiation in a GFP-chimeric mouse model. Invest Ophthalmol Vis Sci 51(9):4831–4839. doi:10.1167/iovs.09-4923
Wegiel J, Imaki H, Wang KC, Wegiel J, Rubenstein R (2004) Cells of monocyte/microglial lineage are involved in both microvessel amyloidosis and fibrillar plaque formation in APPsw tg mice. Brain Res 1022(1–2):19–29. doi:10.1016/j.brainres.2004.06.058
Mezey E, Chandross KJ, Harta G, Maki RA, McKercher SR (2000) Turning blood into brain: cells bearing neuronal antigens generated in vivo from bone marrow. Science 290(5497):1779–1782
Biju KC, Santacruz RA, Chen C, Zhou Q, Yao J, Rohrabaugh SL, Clark RA, Roberts JL et al (2013) Bone marrow-derived microglia-based neurturin delivery protects against dopaminergic neurodegeneration in a mouse model of Parkinson’s disease. Neurosci Lett 535:24–29. doi:10.1016/j.neulet.2012.12.034
Kierdorf K, Katzmarski N, Haas CA, Prinz M (2013) Bone marrow cell recruitment to the brain in the absence of irradiation or parabiosis bias. PLoS One 8(3):e58544. doi:10.1371/journal.pone.0058544
Capotondo A, Milazzo R, Politi LS, Quattrini A, Palini A, Plati T, Merella S, Nonis A et al (2012) Brain conditioning is instrumental for successful microglia reconstitution following hematopoietic stem cell transplantation. Proc Natl Acad Sci U S A 109(37):15018–15023. doi:10.1073/pnas.1205858109
Bellavance MA, Gosselin D, Yong VW, Stys PK, Rivest S (2015) Patrolling monocytes play a critical role in CX3CR1-mediated neuroprotection during excitotoxicity. Brain Struct Funct 220(3):1759–1776. doi:10.1007/s00429-014-0759-z
King IL, Dickendesher TL, Segal BM (2009) Circulating Ly-6C+ myeloid precursors migrate to the CNS and play a pathogenic role during autoimmune demyelinating disease. Blood 113(14):3190–3197. doi:10.1182/blood-2008-07-168575
Humpel C (2008) Basolateral aggregated rat amyloidbeta(1–42) potentiates transmigration of primary rat monocytes through a rat blood–brain barrier. Curr Neurovasc Res 5(3):185–192
Hohsfield LA, Humpel C (2010) Homocysteine enhances transmigration of rat monocytes through a brain capillary endothelial cell monolayer via ICAM-1. Curr Neurovasc Res 7(3):192–200
Haurigot V, Villacampa P, Ribera A, Llombart C, Bosch A, Nacher V, Ramos D, Ayuso E et al (2009) Increased intraocular insulin-like growth factor-I triggers blood-retinal barrier breakdown. J Biol Chem 284(34):22961–22969. doi:10.1074/jbc.M109.014787
Shi C, Pamer EG (2011) Monocyte recruitment during infection and inflammation. Nat Rev Immunol 11(11):762–774. doi:10.1038/nri3070
Wohleb ES, Powell ND, Godbout JP, Sheridan JF (2013) Stress-induced recruitment of bone marrow-derived monocytes to the brain promotes anxiety-like behavior. J Neurosci 33(34):13820–13833. doi:10.1523/JNEUROSCI.1671-13.2013
Mizutani M, Pino PA, Saederup N, Charo IF, Ransohoff RM, Cardona AE (2012) The fractalkine receptor but not CCR2 is present on microglia from embryonic development throughout adulthood. J Immunol 188(1):29–36. doi:10.4049/jimmunol.1100421
Tsou CL, Peters W, Si Y, Slaymaker S, Aslanian AM, Weisberg SP, Mack M, Charo IF (2007) Critical roles for CCR2 and MCP-3 in monocyte mobilization from bone marrow and recruitment to inflammatory sites. J Clin Invest 117(4):902–909. doi:10.1172/JCI29919
Lu H, Huang D, Ransohoff RM, Zhou L (2011) Acute skeletal muscle injury: CCL2 expression by both monocytes and injured muscle is required for repair. FASEB J 25(10):3344–3355. doi:10.1096/fj.10-178939
Zhang ZX, Wang YS, Shi YY, Hou HY, Zhang C, Cai Y, Dou GR, Yao LB et al (2011) Hypoxia specific SDF-1 expression by retinal pigment epithelium initiates bone marrow-derived cells to participate in Choroidal neovascularization in a laser-induced mouse model. Curr Eye Res 36(9):838–849. doi:10.3109/02713683.2011.593107
Chan-Ling T, Baxter L, Afzal A, Sengupta N, Caballero S, Rosinova E, Grant MB (2006) Hematopoietic stem cells provide repair functions after laser-induced Bruch’s membrane rupture model of choroidal neovascularization. Am J Pathol 168(3):1031–1044. doi:10.2353/ajpath.2006.050697
Lima E, Silva R, Shen J, Hackett SF, Kachi S, Akiyama H, Kiuchi K, Yokoi K et al (2007) The SDF-1/CXCR4 ligand/receptor pair is an important contributor to several types of ocular neovascularization. FASEB J 21(12):3219–3230. doi:10.1096/fj.06-7359com
Li H, Yan Z, Cao H, Wang Y (2012) Effective mobilisation of bone marrow-derived cells through proteolytic activity: a new treatment strategy for age-related macular degeneration. Med Hypotheses 78(2):286–290. doi:10.1016/j.mehy.2011.11.003
Morganti JM, Jopson TD, Liu S, Gupta N, Rosi S (2014) Cranial irradiation alters the brain’s microenvironment and permits CCR2+ macrophage infiltration. PLoS One 9(4):e93650. doi:10.1371/journal.pone.0093650
Gragoudas ES, Adamis AP, Cunningham ET Jr, Feinsod M, Guyer DR, Group VISiONCT (2004) Pegaptanib for neovascular age-related macular degeneration. N Engl J Med 351(27):2805–2816. doi:10.1056/NEJMoa042760
Eglitis MA, Mezey E (1997) Hematopoietic cells differentiate into both microglia and macroglia in the brains of adult mice. Proc Natl Acad Sci U S A 94(8):4080–4085
Biffi A, De Palma M, Quattrini A, Del Carro U, Amadio S, Visigalli I, Sessa M, Fasano S et al (2004) Correction of metachromatic leukodystrophy in the mouse model by transplantation of genetically modified hematopoietic stem cells. J Clin Invest 113(8):1118–1129. doi:10.1172/JCI19205
Shi C, Jia T, Mendez-Ferrer S, Hohl TM, Serbina NV, Lipuma L, Leiner I, Li MO et al (2011) Bone marrow mesenchymal stem and progenitor cells induce monocyte emigration in response to circulating toll-like receptor ligands. Immunity 34(4):590–601. doi:10.1016/j.immuni.2011.02.016
Zouggari Y, Ait-Oufella H, Bonnin P, Simon T, Sage AP, Guerin C, Vilar J, Caligiuri G et al (2013) B lymphocytes trigger monocyte mobilization and impair heart function after acute myocardial infarction. Nat Med 19(10):1273–1280. doi:10.1038/nm.3284
Hattori K, Heissig B, Rafii S (2003) The regulation of hematopoietic stem cell and progenitor mobilization by chemokine SDF-1. Leuk Lymphoma 44(4):575–582. doi:10.1080/1042819021000037985
Karlstetter M, Nothdurfter C, Aslanidis A, Moeller K, Horn F, Scholz R, Neumann H, Weber BH et al (2014) Translocator protein (18 kDa) (TSPO) is expressed in reactive retinal microglia and modulates microglial inflammation and phagocytosis. J Neuroinflammation 11:3. doi:10.1186/1742-2094-11-3
Zhang C, Lei B, Lam TT, Yang F, Sinha D, Tso MO (2004) Neuroprotection of photoreceptors by minocycline in light-induced retinal degeneration. Invest Ophthalmol Vis Sci 45(8):2753–2759. doi:10.1167/iovs.03-1344
Karlstetter M, Lippe E, Walczak Y, Moehle C, Aslanidis A, Mirza M, Langmann T (2011) Curcumin is a potent modulator of microglial gene expression and migration. J Neuroinflammation 8:125. doi:10.1186/1742-2094-8-125
Noro T, Namekata K, Kimura A, Guo X, Azuchi Y, Harada C, Nakano T, Tsuneoka H et al (2015) Spermidine promotes retinal ganglion cell survival and optic nerve regeneration in adult mice following optic nerve injury. Cell Death Dis 6:e1720. doi:10.1038/cddis.2015.93
Huang H, Parlier R, Shen JK, Lutty GA, Vinores SA (2013) VEGF receptor blockade markedly reduces retinal microglia/macrophage infiltration into laser-induced CNV. PLoS One 8(8):e71808. doi:10.1371/journal.pone.0071808
Mirza M, Volz C, Karlstetter M, Langiu M, Somogyi A, Ruonala MO, Tamm ER, Jagle H et al (2013) Progressive retinal degeneration and glial activation in the CLN6 (nclf) mouse model of neuronal ceroid lipofuscinosis: a beneficial effect of DHA and curcumin supplementation. PLoS One 8(10):e75963. doi:10.1371/journal.pone.0075963
Wang Y, Neumann H (2010) Alleviation of neurotoxicity by microglial human Siglec-11. J Neurosci 30(9):3482–3488. doi:10.1523/JNEUROSCI.3940-09.2010
Bian M, Du X, Cui J, Wang P, Wang W, Zhu W, Zhang T, Chen Y (2016) Celastrol protects mouse retinas from bright light-induced degeneration through inhibition of oxidative stress and inflammation. J Neuroinflammation 13(1):50. doi:10.1186/s12974-016-0516-8
Zhang-Gandhi CX, Drew PD (2007) Liver X receptor and retinoid X receptor agonists inhibit inflammatory responses of microglia and astrocytes. J Neuroimmunol 183(1–2):50–59. doi:10.1016/j.jneuroim.2006.11.007
Kambhampati SP, Mishra MK, Mastorakos P, Oh Y, Lutty GA, Kannan RM (2015) Intracellular delivery of dendrimer triamcinolone acetonide conjugates into microglial and human retinal pigment epithelial cells. Eur J Pharm Biopharm 95(Pt B):239–249. doi:10.1016/j.ejpb.2015.02.013
Kambhampati SP, Clunies-Ross AJ, Bhutto I, Mishra MK, Edwards M, McLeod DS, Kannan RM, Lutty G (2015) Systemic and intravitreal delivery of dendrimers to activated microglia/macrophage in ischemia/reperfusion mouse retina. Invest Ophthalmol Vis Sci 56(8):4413–4424. doi:10.1167/iovs.14-16250
Aslanidis A, Karlstetter M, Scholz R, Fauser S, Neumann H, Fried C, Pietsch M, Langmann T (2015) Activated microglia/macrophage whey acidic protein (AMWAP) inhibits NFkappaB signaling and induces a neuroprotective phenotype in microglia. J Neuroinflammation 12:77. doi:10.1186/s12974-015-0296-6
Wang K, Peng B, Lin B (2014) Fractalkine receptor regulates microglial neurotoxicity in an experimental mouse glaucoma model. Glia 62(12):1943–1954. doi:10.1002/glia.22715
Couturier A, Bousquet E, Zhao M, Naud MC, Klein C, Jonet L, Tadayoni R, de Kozak Y et al (2014) Anti-vascular endothelial growth factor acts on retinal microglia/macrophage activation in a rat model of ocular inflammation. Mol Vis 20:908–920
Xu YF, Fu LL, Jiang CH, Qin YW, Ni YQ, Fan JW (2012) Naloxone inhibition of lipopolysaccharide-induced activation of retinal microglia is partly mediated via the p38 mitogen activated protein kinase signalling pathway. J Int Med Res 40(4):1438–1448
McVicar CM, Hamilton R, Colhoun LM, Gardiner TA, Brines M, Cerami A, Stitt AW (2011) Intervention with an erythropoietin-derived peptide protects against neuroglial and vascular degeneration during diabetic retinopathy. Diabetes 60(11):2995–3005. doi:10.2337/db11-0026
Kitaoka Y, Hayashi Y, Kumai T, Takeda H, Munemasa Y, Fujino H, Kitaoka Y, Ueno S et al (2009) Axonal and cell body protection by nicotinamide adenine dinucleotide in tumor necrosis factor-induced optic neuropathy. J Neuropathol Exp Neurol 68(8):915–927. doi:10.1097/NEN.0b013e3181afecfa
Ebert S, Schoeberl T, Walczak Y, Stoecker K, Stempfl T, Moehle C, Weber BH, Langmann T (2008) Chondroitin sulfate disaccharide stimulates microglia to adopt a novel regulatory phenotype. J Leukoc Biol 84(3):736–740. doi:10.1189/jlb.0208138
Acknowledgments
This work was supported by the National Basic Research Program of China (973 Program, 2013CB967002) and partially supported by the National Nature Science Foundation of China (No. 81570890).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jin, N., Gao, L., Fan, X. et al. Friend or Foe? Resident Microglia vs Bone Marrow-Derived Microglia and Their Roles in the Retinal Degeneration. Mol Neurobiol 54, 4094–4112 (2017). https://doi.org/10.1007/s12035-016-9960-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-016-9960-9