Abstract
Multiple players are involved in motor and sensory dysfunctions after spinal cord injury (SCI). Therefore, therapeutic approaches targeting these various players in the damage cascade hold considerable promise for the treatment of traumatic spinal cord injury. Soluble epoxide hydrolase (sEH) is an endogenous key enzyme in the metabolic conversion and degradation of P450 eicosanoids called epoxyeicosatrienoic acids (EETs). sEH inhibition has been shown to provide neuroprotective effects upon multiple elements of neurovascular unit under cerebral ischemia. However, its role in the pathological process after SCI remains unclear. In this study, we tested the hypothesis that sEH inhibition may have therapeutic effects in preventing secondary damage in rats after traumatic SCI. sEH was widely expressed in spinal cord tissue, mainly confined to astrocytes, and neurons. Administration of sEH inhibitor AUDA significantly suppressed local inflammatory responses as indicated by the reduced microglia activation and IL-1 β expression, as well as the decreased infiltration of neutrophils and T lymphocytes. Meanwhile, reactive astrogliosis was remarkably attenuated. Furthermore, treatment of AUDA improved angiogenesis, inhibited neuron cells apoptosis, alleviated demyelination and formation of cavity and improved motor recovery. Together, these results provide the first in vivo evidence that sEH inhibition could exert multiple targets protective effects after SCI in rats. sEH may thereby serve as a promising multi-mechanism therapeutic target for the treatment of SCI.









Similar content being viewed by others
References
Tohda C, Kuboyama T (2011) Current and future therapeutic strategies for functional repair of spinal cord injury. Pharmacol Ther 132(1):57–71
Shih CH, Lacagnina M, Leuer-Bisciotti K, Pröschel C (2014) Astroglial-derived periostin promotes axonal regeneration after spinal cord injury. J Neurosci 34(7):2438–2443
Su Z, Yuan Y, Cao L, Zhu Y, Gao L, Qiu Y, He C (2010) Triptolide promotes spinal cord repair by inhibiting astrogliosis and inflammation. Glia 58(8):901–915
Iliff JJ, Jia J, Nelson J, Goyagi T, Klaus J, Alkayed NJ (2010) Epoxyeicosanoid signaling in CNS function and disease. Prostaglandins Lipid Mediat 91(3–4):68–84
Xu X, Zhang XA, Wang DW (2011) The roles of CYP450 epoxygenases and metabolites, epoxyeicosatrienoic acids, in cardiovascular and malignant diseases. Adv Drug Deliv Rev 63(8):597–609
Koerner IP, Jacks R, DeBarber AE, Koop D, Mao P, Grant DF, Alkayed NJ (2007) Polymorphisms in the human soluble epoxide hydrolase gene EPHX2 linked to neuronal survival after ischemic injury. J Neurosci 27:4642–4649
Medhora M, Dhanasekaran A, Gruenloh SK, Dunn LK, Gabrilovich M, Falck JR, Miettinen M, Lindenmayer AE et al (1994) Endothelial cell markers CD31, CD34, and BNH9 antibody to H- and Y-antigens—evaluation of their specificity and sensitivity in the diagnosis of vascular tumors and comparison with von Willebrand factor. Mod Pathol 7(1):82–90
Liu M, Alkayed NJ (2005) Hypoxic preconditioning and tolerance via hypoxia inducible factor (HIF) 1alpha linked induction of P450 2C11 epoxygenase in astrocytes. J Cereb Blood Flow Metab 25:939–948
Sura P, Sura R, Enayetallah AE, Grant DF (2008) Distribution and expression of soluble epoxide hydrolase in human brain. J Histochem Cytochem 56(6):551–559
Iliff JJ, Close LN, Selden NR, Alkayed NJ (2007) A novel role for P450 eicosanoids in the neurogenic control of cerebral blood flow in the rat. Exp Physiol 92(4):653–658
Zhang W, Koerner IP, Noppens R, Grafe M, Tsai HJ, Morisseau C, Luria A, Hammock BD et al (2007) Soluble epoxide hydrolase: a novel therapeutic target in stroke. J Cereb Blood Flow Metab 27(12):1931–1940
Marowsky A, Burgener J, Falck JR, Fritschy JM, Arand M (2009) Distribution of soluble and microsomal epoxide hydrolase in the mouse brain and its contribution to cerebral epoxyeicosatrienoic acid metabolism. Neuroscience 163(2):646–661
Dorrance AM, Rupp N, Pollock DM, Newman JW, Hammock BD, Imig JD (2005) An epoxide hydrolase inhibitor, 12-(3-adamantan-1-yl-ureido)dodecanoic acid (AUDA), reduces ischemic cerebral infarct size in stroke-prone spontaneously hypertensive rats. J Cardiovasc Pharmacol 46:842–848
Zhang W, Otsuka T, Sugo N, Ardeshiri A, Alhadid YK, Iliff JJ, DeBarber AE, Koop DR et al (2008) Soluble epoxide hydrolase gene deletion is protective against experimental cerebral ischemia. Stroke 39(7):2073–2078
Iliff JJ, Alkayed NJ (2009) Soluble epoxide hydrolase inhibition: targeting multiple mechanisms of ischemic brain injury with a single agent. Future Neurol 4(2):179–199
Perot PL Jr, Lee WA, Hsu CY, Hogan EL, Cox RD, Gross AJ (1987) Therapeutic model for experimental spinal cord injury in the rat: I. Mortality and motor deficit. Cent Nerv Syst Trauma 4:149–159
Lee JP, Yang SH, Lee HY, Kim B, Cho JY, Paik JH, Oh YJ, Kim DK et al (2012) Soluble epoxide hydrolase activity determines the severity of ischemia-reperfusion injury in kidney. PLoS One 7(5):e37075
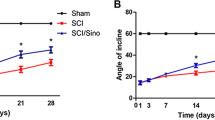
Basso DM, Beattie MS, Bresnahan JC (1995) A sensitive and reliable locomotor rating scale for open field testing in rat. J Neurotrauma 12:1–21
Fitch MT, Silver J (2008) CNS injury, glial scars, and inflammation: inhibitory extracellular matrices and regeneration failure. Exp Neurol 209(2):294–301
Ito D, Imai Y, Ohsawa K, Nakajima K, Fukuuchi Y, Kohsaka S (1998) Microglia-specific localisation of a novel calcium binding protein, Iba1. Brain Res Mol Brain Res 57(1):1–9
Esposito E, Cuzzocrea S (2011) Anti-TNF therapy in the injured spinal cord. Trends Pharmacol Sci 32(2):107–115
Taoka Y, Okajima K (1998) Spinal cord injury in the rat. Prog Neurobiol 56(3):341–358
Mautes AE, Weinzierl MR, Donovan F, Noble LJ (2000) Vascular events after spinal cord injury: contribution to secondary pathogenesis. Phys Ther 80(7):673–687
Benowitz LI, Routtenberg A (1997) GAP-43: an intrinsic determinant of neuronal development and plasticity. Trends Neurosci 20(2):84–91
Schwab ME, Bartholdi D (1996) Degeneration and regeneration of axons in the lesioned spinal cord. Physiol Rev 76:319–370
Fawcett JW, Asher RA (1999) The glial scar and central nervous system repair. Brain Res Bull 49:377–391
Silver J, Miller JH (2004) Regeneration beyond the glial scar. Nat Rev Neurosci 5:146–156
Bunge MB (2008) Novel combination strategies to repair the injured mammalian spinal cord. J Spinal Cord Med 31:262–269
Fortun J, Hill CE, Bunge MB (2009) Combinatorial strategies with Schwann cell transplantation to improve repair of the injured spinal cord. Neurosci Lett 456:124–132
Inceoglu B, Schmelzer KR, Morisseau C, Jinks SL, Hammock BD (2007) Soluble epoxide hydrolase inhibition reveals novel biological functions of epoxyeicosatrienoic acids (EETs). Prostaglandins Lipid Mediat 82(1–4):42–49
Karimi-Abdolrezaee S, Billakanti R (2012) Reactive astrogliosis after spinal cord injury-beneficial and detrimental effects. Mol Neurobiol 46(2):251–264
Teshigawara K, Kuboyama T, Shigyo M, Nagata A, Sugimoto K, Matsuya Y, Tohda C (2013) A novel compound, denosomin, ameliorates spinal cord injury via axonal growth associated with astrocyte-secreted vimentin. Br J Pharmacol 168(4):903–919
Herrmann JE, Imura T, Song B, Qi J, Ao Y, Nguyen TK, Korsak RA, Takeda K et al (2008) STAT3 is a critical regulator of astrogliosis and scar formation after spinal cord injury. J Neurosci 28:7231–7243
Karimi-Abdolrezaee S, Eftekharpour E, Wang J, Schut D, Fehlings MG (2010) Synergistic effects of transplanted adult neural stem/progenitor cells, chondroitinase, and growth factors promote functional repair and plasticity of the chronically injured spinal cord. J Neurosci 30:1657–1676
Bradbury EJ, Moon LD, Popat RJ, King VR, Bennett GS, Patel PN, Fawcett JW, McMahon SB (2002) Chondroitinase ABC promotes functional recovery after spinal cord injury. Nature 416:636–640
Tian DS, Yu ZY, Xie MJ, Bu BT, Witte OW, Wang W (2006) Suppression of astroglial scar formation and enhanced axonal regeneration associated with functional recovery in a spinal cord injury rat model by the cell cycle inhibitor olomoucine. J Neurosci Res 84(5):1053–1063
Schwab JM, Zhang Y, Kopp MA, Brommer B, Popovich PG (2014) The paradox of chronic neuroinflammation, systemic immune suppression, autoimmunity after traumatic chronic spinal cord injury. Exp Neurol 258C:121–129
Yuan Y, Su Z, Pu Y, Liu X, Chen J, Zhu F, Zhu Y, Zhang H et al (2012) Ethyl pyruvate promotes spinal cord repair by ameliorating the glial microenvironment. Br J Pharmacol 166(2):749–763
Node K, Huo Y, Ruan X, Yang B, Spiecker M, Ley K, Zeldin DC, Liao JK (1999) Anti-inflammatory properties of cytochrome P450 epoxygenase-derived eicosanoids. Science 285:1276–1279
Schmelzer KR, Kubala L, Newman JW, Kim IH, Eiserich JP, Hammock BD (2005) Soluble epoxide hydrolase is a therapeutic target for acute inflammation. Proc Natl Acad Sci U S A 102:9772–9777
Koerner IP, Zhang W, Cheng J, Parker S, Hurn PD, Alkayed NJ (2008) Soluble epoxide hydrolase: regulation by estrogen and role in the inflammatory response to cerebral ischemia. Front Biosc 13:2833–2841
Huang C, Han X, Li X, Lam E, Peng W, Lou N, Torres A, Yang M et al (2012) Critical role of connexin 43 in secondary expansion of traumatic spinal cord injury. J Neurosci 32(10):3333–3338
Trivedi A, Olivas AD, Noble-Haeusslein LJ (2006) Inflammation and spinal cord injury: infiltrating leukocytes as determinants of injury and repair processes. Clin Neurosci Res 6(5):283–292
Tator CH, Fehlings MG (1991) Review of the secondary injury theory of acute spinal cord trauma with emphasis on vascular mechanisms. J Neurosurg 75:15–26
Glaser J, Gonzalez R, Sadr E, Keirstead HS (2006) Neutralization of the chemokine CXCL10 reduces apoptosis and increases axon sprouting after spinal cord injury. J Neurosci Res 84:724–734
Yoshihara T, Ohta M, Itokazu Y, Matsumoto N, Dezawa M, Suzuki Y, Taguchi A, Watanabe Y et al (2007) Neuroprotective effect of bone marrow–derived mononuclear cells promoting functional recovery from spinal cord injury. J Neurotrauma 24:1026–1036
Figley SA, Liu Y, Karadimas SK, Satkunendrarajah K, Fettes P, Spratt SK, Lee G, Ando D et al (2014) Delayed administration of a bio-engineered zinc-finger VEGF-A gene therapy is neuroprotective and attenuates allodynia following traumatic spinal cord injury. PLoS One 9(5):e96137
Fleming I (2007) Epoxyeicosatrienoic acids, cell signaling and angiogenesis. Prostaglandins Lipid Mediat 82(1–4):60–67
Munzenmaier DH, Harder DR (2000) Cerebral microvascular endothelial cell tube formation: role of astrocytic epoxyeicosatrienoic acid release. Am J Physiol Heart Circ Physiol 278:H1163–H1167
Zhang C, Harder DR (2002) Cerebral capillary endothelial cell mitogenesis and morphogenesis induced by astrocytic epoxyeicosatrienoic Acid. Stroke 33:2957–2964
Wang Y, Wei X, Xiao X, Hui R, Card JW, Carey MA, Wang DW, Zeldin DC (2005) Arachidonic acid epoxygenase metabolites stimulate endothelial cell growth and angiogenesis via mitogen-activated protein kinase and phosphatidylinositol 3-kinase/Akt signaling pathways. J Pharmacol Exp Ther 314(2):522–532
Simpkins AN, Rudic RD, Schreihofer DA, Roy S, Manhiani M, Tsai HJ, Hammock BD, Imig JD (2009) Soluble epoxide inhibition is protective against cerebral ischemia via vascular and neural protection. Am J Pathol 174(6):2086–2095
Almad A, Sahinkaya FR, McTigue DM (2011) Oligodendrocyte fate after spinal cord injury. Neurotherapeutics 8:262–273
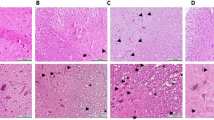
Liu XZ, Xu XM, Hu R, Du C, Zhang SX, McDonald JW, Dong HX, Wu YJ et al (1997) Neuronal and glial apoptosis after traumatic spinal cord injury. J Neurosci 17:5395–5406
Beattie MS, Li Q, Bresnahan JC (2000) Cell death and plasticity after experimental spinal cord injury. Prog Brain Res 128:9–21
McTigue DM, Tripathi RB (2008) The life, death, and replacement of oligodendrocytes in the adult CNS. J Neurochem 107(1):1–19
Li ZW, Tang RH, Zhang JP, Tang ZP, Qu WS, Zhu WH, Li JJ, Xie MJ et al (2011) Inhibiting epidermal growth factor receptor attenuates reactive astrogliosis and improves functional outcome after spinal cord injury in rats. Neurochem Int 58(7):812–819
Acknowledgments
The investigation was supported by the National Natural Science Foundation of China (81371312, 61327902, 30971007), National Basic Research Development Program (973 Program) of China (2011CB504403) and Natural Science Foundation of Hubei Province (2014CFB445). The authors declare no competing financial interests.
Author information
Authors and Affiliations
Corresponding author
Additional information
Xiaojing Chen and Xiaoqi Chen contributed equally to this work.
Rights and permissions
About this article
Cite this article
Chen, X., Chen, X., Huang, X. et al. Soluble epoxide hydrolase inhibition provides multi-target therapeutic effects in rats after spinal cord injury. Mol Neurobiol 53, 1565–1578 (2016). https://doi.org/10.1007/s12035-015-9118-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-015-9118-1