Abstract
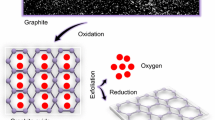
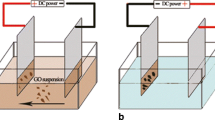
Graphene has superior electrical conductivity than graphite and other allotropes of carbon because of its high surface area and chemical tolerance. Electrochemically processed graphene sheets were obtained through the reduction of graphene oxide from hydrazine hydrate. The prepared samples were heated to different temperatures such as 673 and 873 K. X-ray diffraction (XRD), fourier transform infrared spectroscopy (FTIR), scanning electron microscopy (SEM), energy dispersive X-ray spectroscopy (EDXS), transmission electron microscopy (TEM), Raman spectra and conductivity measurements were made for as-prepared and heat-treated graphene samples. XRD pattern of graphene shows a sharp and intensive peak centred at a diffraction angle (2θ) of 26·350. FTIR spectra of as-prepared and heated graphene were used to confirm the oxidation of graphite. TEM results indicated that the defect density and number of layers of graphene sheets were varied with heating temperature. The hexagonal sheet morphology and purity of as-prepared and heat treated samples were confirmed by SEM–EDX and Raman spectroscopy. The conductivity measurements revealed that the conductivity of graphene was decreased with an increase in heating temperature. The present study explains that graphene with enhanced functional properties can be achieved from the as-prepared sample.






Similar content being viewed by others
References
Choucair M, Thordarson P and Stride J A 2009 Nat. Nanotechnol. 4 30
Dong L X and Chen Q 2010 Chem. Mater. Sci. 4 45
Eda G, Fanchini G and Chhowalla M 2008 Nat. Nanotechnol. 3 270
Ferrari A C and Robertson J 2000 Phys. Rev. B61 14095
Ferrari A C et al 2006 Phys. Rev. Lett. 97 187401
Ghosh D, Chandra S, Chakraborty A, Ghosh S K and Pramanik P 2010 Int. J. Pharm. Sci. Drug. Res. 2 127
Gijie S, Han S, Wang M, Wang K L and Kaner R B 2007 Nano. Lett. 7 3394
Ju H M, Choi S H and Huh S H 2010 J. Korean Phys. Soc. 57 1649
Kaniyoor A, Baby T and Ramaprabhu S 2010 J. Mater. Chem. 20 8467
Kumar A et al 2011 ACS Nano. 5 4345
Liu N, Luo F, Wu H, Liu Y, Zhang C and Chen J 2008 Adv. Funct. Mater. 18 1518
Luo Z, Lu Y, Somers L A and Johnson A T 2009 J. Am. Chem. Soc. 131 898
Ni Z, Wang Y, Yu T and Shen Z 2008 Nano. Res. 1 273
Novoselov K S, Geim A K, Morozov S V, Jiang D, Zhang Y and Dubonos S V 2004 Science 306 666
O’Brien M and Nichols B 2010 Army research laboratory ARL-TR- 5047
Park S and Ruoff R S 2009 Nat. Nanotechnol. 4 217
Parthasarathy G, Sreedhar B and Chetty T R K 2006 Curr. Sci. 90 995
Ramesha G K and Sampath S 2009 J. Phys. Chem. C113 7985
Reina A, Jia X T, Ho J, Nezich D, Son H, Bulovic V, Dresselhaus M S and Kong J 2009a Nano. Lett. 9 1752
Reina A et al 2009b Nano. Lett. 9 30
Schwab T, Burg B R, Schirmar N C and Poulikakos D 2009 Nanotechnol. 20 405704
Seger B and Kamat P V 2009 J. Phys. Chem. C113 7990
Shinde D B, Debgupta J, Kushwaha A, Aslam M and Pillai V K 2011 J. Am. Chem. Soc. 133 4168
Sima M, Enculescu I and Sima A 2011 Optoelectron. Adv. Mater. 5 414
Singh D K, Iyery P K and Giriz P K 2011 Int. J. Nanosci. 10 1
Stankovich S, Dikin D A, Dommett G H B, Rohlhaas K M, Zimney E J and Stach E A 2006 Nature 442 282
Su C Y, Lu A Y, Xu Y, Chen F R, Khlobystov A N and Li L J 2011 ACS Nano. 5 2332
Tang L, Wang Y, Li Y, Feng H, Lu J and Li J 2009 Adv. Funct. Mater. 19 2782
Titelman G I, Gelman V, Bron S, Khalfin R L, Cohen Y and Bianco-Peled H 2005 Carbon 43 641
Wang C, Li D, Too C O and Wallace G G 2009 Chem. Mater. 21 2604
Wang F et al 2011 ACS Nano. 5 1190
Wang G, Wang B, Park J, Wang Y, Sun B and Yao J 2009 Carbon 47 3242
Wang J J, Zhu M Y, Outlaw R A, Zhao X, Manos D M, Holloway B C and Mammana V P 2004 Appl. Phys. Lett. 85 1265
Wu H, Liu J, Aksay I A and Lin Y 2010 Electroanal. 22 1027
Wu J B, Becerril H A, Bao Z N, Liu Z F, Chen Y S and Peumans P 2008 Appl. Phys. Lett. 92 263302
Yoo E J, Kim J, Hosono E, Zhou H S, Kudo T and Honma I 2008 Nano. Lett. 8 2277
Zhang L L, Wei T, Wang W and Zhao X S 2009 Micropor. Mesopor. Mater. 123 260
Acknowledgement
One of the authors (M S) is thankful to the Department of Science and Technology (DST), New Delhi, for providing the Inspire Fellowship to carry out the research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
SELVAM, M., SAKTHIPANDI, K., SURIYAPRABHA, R. et al. Synthesis and characterization of electrochemically-reduced graphene. Bull Mater Sci 36, 1315–1321 (2013). https://doi.org/10.1007/s12034-013-0581-x
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12034-013-0581-x