Abstract
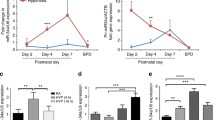
Bronchopulmonary dysplasia (BPD), caused by hyperoxia exposure, is the most common complication affecting preterm infants. The C-X-C motif chemokine ligand 17 (CXCL17) belongs to the chemokine family that plays important roles in various processes, but the function in BPD is unknown. Elevated serum CXCL17 levels were observed in human premature infants with hyperoxia-induced lung injury, suggesting that CXCL17 might be involved in BPD. To further validate our speculation, studies were conducted in a hyperoxia-induced lung injury mouse model and primary murine alveolar epithelial cells Type II (T2AEC) cells exposed to hyperoxia. RT-qPCR and western blot were used to validate CXCL17 expression in newborn mice. Hyperoxia exposure-induced lung injury was determined by assessing the lung wet-weight/dry-weight ratio and histological changes. Oxidative stress and inflammatory factors were examined by ELISA assay and RT-qPCR. Reactive oxygen species (ROS) level was evaluated by DHE staining. Apoptosis was assessed by TUNEL staining and western blot. The results showed that hyperoxia exposure increased CXCL17 levels in newborn mice pups. Hyperoxia exposure increased lung wet-weight/dry-weight ratio, increased alveolar diameter and enlarged alveoli, and reduced surfactant protein C expression. However, recombinant CXCL17 (rCXCL17) treatment alleviated hyperoxia-induced lung injury. rCXCL17 treatment inhibited hyperoxia-induced inflammation, oxidative stress, and apoptosis in neonatal mice. These results were further verified in T2AEC cells. Additionally, rCXCL17 treatment activated the AKT pathway, which is a protective pathway in BPD. Collectively, rCXCL17 alleviates hyperoxia-induced lung injury in neonatal mice by activating the AKT pathway, indicating that CXCL17 may be a promising target for BPD therapy.








Similar content being viewed by others
Data Availability
Data will be made available upon reasonable request.
References
Gong, J., Feng, Z., Peterson, A. L., Carr, J. F., Vang, A., Braza, J., Choudhary, G., Dennery, P. A., & Yao, H. (2020). Endothelial to mesenchymal transition during neonatal hyperoxia-induced pulmonary hypertension. The Journal of Pathology, 252, 411–422.
Gilfillan, M., Bhandari, A., & Bhandari, V. (2021). Diagnosis and management of bronchopulmonary dysplasia. Bmj. https://doi.org/10.1136/bmj.n1974
Jobe, A. H., & Abman, S. H. (2019). Bronchopulmonary dysplasia: A Continuum of Lung Disease from the Fetus to the adult. American Journal of Respiratory and Critical Care Medicine, 200, 659–660.
Nicchi, S., Giusti, F., Carello, S., Utrio Lanfaloni, S., Tavarini, S., Frigimelica, E., Ferlenghi, I., Rossi Paccani, S., Merola, M., Delany, I., Scarlato, V., Maione, D., & Brettoni, C. (2022). Moraxella catarrhalis evades neutrophil oxidative stress responses providing a safer niche for nontypeable Haemophilus influenzae. iScience, 25, 103931.
Cannavo, L., Perrone, S., Viola, V., Marseglia, L., Di Rosa, G., & Gitto, E. (2021). Oxidative stress and respiratory diseases in preterm newborns. International Journal Of Molecular Sciences, 22(55), 12504.
Denisov, S. S. (2021). CXCL17: The Black Sheep in the chemokine flock. Frontiers in Immunology, 12, 712897.
Weinstein, E. J., Head, R., Griggs, D. W., Sun, D., Evans, R. J., Swearingen, M. L., Westlin, M. M., & Mazzarella, R. (2006). VCC-1, a novel chemokine, promotes tumor growth. Biochemical and Biophysical Research Communications, 350, 74–81.
Oka, T., Sugaya, M., Takahashi, N., Takahashi, T., Shibata, S., Miyagaki, T., Asano, Y., & Sato, S. (2017). CXCL17 attenuates imiquimod-induced psoriasis-like skin inflammation by recruiting myeloid-derived suppressor cells and regulatory T cells. Journal of Immunology, 198(10), 3897–3908.
Zhang, K., Liang, Y., Feng, Y., Wu, W., Zhang, H., He, J., Hu, Q., Zhao, J., Xu, Y., Liu, Z., & Zhen, G. (2018). Decreased epithelial and sputum mir-221-3p associates with airway eosinophilic inflammation and CXCL17 expression in asthma. American Journal of Physiology Lung Cellular and Molecular Physiology, 315, L253–L264.
Hernandez-Ruiz, M., Othy, S., Herrera, C., Nguyen, H. T., Arrevillaga-Boni, G., Catalan-Dibene, J., Cahalan, M. D., & Zlotnik, A. (2019). Cxcl17(-/-) mice develop exacerbated disease in a T cell-dependent autoimmune model. Journal of Leukocyte Biology, 105, 1027–1039.
Shimada, S., Makino, K., Jinnin, M., Sawamura, S., Kawano, Y., Ide, M., Kajihara, I., Makino, T., Fukushima, S., & Ihn, H. (2020). CXCL17-mediated downregulation of type I collagen via MMP1 and miR-29 in skin fibroblasts possibly contributes to the fibrosis in systemic sclerosis. Journal of Dermatological Science, 100, 183–191.
Zhou, Z., Lu, X., Zhu, P., Zhu, W., Mu, X., Qu, R., & Li, M. (2012). VCC-1 over-expression inhibits cisplatin-induced apoptosis in HepG2 cells. Biochemical and Biophysical Research Communications, 420, 336–342.
Hsu, Y. L., Yen, M. C., Chang, W. A., Tsai, P. H., Pan, Y. C., Liao, S. H., & Kuo, P. L. (2019). CXCL17-derived CD11b(+)Gr-1(+) myeloid-derived suppressor cells contribute to lung metastasis of breast cancer through platelet-derived growth factor-BB. Breast Cancer Research, 21, 23.
Wang, J., Ito, T., Udaka, N., Okudela, K., Yazawa, T., & Kitamura, H. (2005). PI3K-AKT pathway mediates growth and survival signals during development of fetal mouse lung. Tissue and Cell, 37, 25–35.
Reddy, N. M., Potteti, H. R., Vegiraju, S., Chen, H. J., Tamatam, C. M., & Reddy, S. P. (2015). PI3K-AKT signaling via Nrf2 protects against hyperoxia-induced acute lung injury, but promotes inflammation post-injury independent of Nrf2 in mice. PLoS One, 10, e0129676.
Pisabarro, M. T., Leung, B., Kwong, M., Corpuz, R., Frantz, G. D., Chiang, N., Vandlen, R., Diehl, L. J., Skelton, N., Kim, H. S., Eaton, D., & Schmidt, K. N. (2006). Cutting edge: Novel human dendritic cell- and monocyte-attracting chemokine-like protein identified by fold recognition methods. Journal of Immunology, 176, 2069–2073.
Matsui, A., Yokoo, H., Negishi, Y., Endo-Takahashi, Y., Chun, N. A., Kadouchi, I., Suzuki, R., Maruyama, K., Aramaki, Y., Semba, K., Kobayashi, E., Takahashi, M., & Murakami, T. (2012). CXCL17 expression by tumor cells recruits CD11b + Gr1 high F4/80- cells and promotes tumor progression. PloS One, 7, e44080.
Lee, W. Y., Wang, C. J., Lin, T. Y., Hsiao, C. L., & Luo, C. W. (2013). CXCL17, an orphan chemokine, acts as a novel angiogenic and anti-inflammatory factor. American Journal of Physiology Endocrinology and Metabolism, 304, E32–40.
Corti, M., Brody, A. R., & Harrison, J. H. (1996). Isolation and primary culture of murine alveolar type II cells. American Journal of Respiratory cell and Molecular Biology, 14, 309–315.
Alejandre-Alcazar, M. A., Kwapiszewska, G., Reiss, I., Amarie, O. V., Marsh, L. M., Sevilla-Perez, J., Wygrecka, M., Eul, B., Kobrich, S., Hesse, M., Schermuly, R. T., Seeger, W., Eickelberg, O., & Morty, R. E. (2007). Hyperoxia modulates TGF-beta/BMP signaling in a mouse model of bronchopulmonary dysplasia. American Journal of Physiology Lung Cellular and Molecular Physiology, 292, L537–549.
Ota, C., Yamada, M., Fujino, N., Motohashi, H., Tando, Y., Takei, Y., Suzuki, T., Takahashi, T., Kamata, S., Makiguchi, T., Yamaya, M., & Kubo, H. (2015). Histone deacetylase inhibitor restores surfactant protein-C expression in alveolar-epithelial type II cells and attenuates bleomycin-induced pulmonary fibrosis in vivo. Experimental lung Research, 41, 422–434.
Hughes, C. E., & Nibbs, R. J. B. (2018). A guide to chemokines and their receptors. The FEBS Journal, 285, 2944–2971.
Harijith, A., Choo-Wing, R., Cataltepe, S., Yasumatsu, R., Aghai, Z. H., Janer, J., Andersson, S., Homer, R. J., & Bhandari, V. (2011). A role for matrix metalloproteinase 9 in IFNgamma-mediated injury in developing lungs: Relevance to bronchopulmonary dysplasia. American Journal of Respiratory cell and Molecular Biology, 44, 621–630.
Tang, X. (2018). Interleukin-33 (IL-33) increases hyperoxia-induced bronchopulmonary dysplasia in newborn mice by regulation of inflammatory mediators. Medical Science Monitor: International Medical Journal of Experimental and Clinical Research, 24, 6717–6728.
Martin, C. R., Zaman, M. M., Gilkey, C., Salguero, M. V., Hasturk, H., Kantarci, A., Van Dyke, T. E., & Freedman, S. D. (2014). Resolvin D1 and lipoxin A4 improve alveolarization and normalize septal wall thickness in a neonatal murine model of hyperoxia-induced lung injury. PloS One, 9, e98773.
Sun, C., Zhang, S., Wang, J., Jiang, W., Xin, Q., Chen, X., Zhang, Z., & Luan, Y. (2019). EPO enhances the protective effects of MSCs in experimental hyperoxia-induced neonatal mice by promoting angiogenesis. Aging (Albany Ny), 11, 2477–2487.
Fu, H., Zhang, T., Huang, R., Yang, Z., Liu, C., Li, M., Fang, F., & Xu, F. (2017). Calcitonin gene-related peptide protects type II alveolar epithelial cells from hyperoxia-induced DNA damage and cell death. Experimental and Therapeutic Medicine, 13, 1279–1284.
Auten, R. L., Mason, S. N., Auten, K. M., & Brahmajothi, M. (2009). Hyperoxia impairs postnatal alveolar epithelial development via NADPH oxidase in newborn mice. American Journal of Physiology Lung Cellular and Molecular Physiology, 297, L134–142.
Balasubramaniam, V., Mervis, C. F., Maxey, A. M., Markham, N. E., & Abman, S. H. (2007). Hyperoxia reduces bone marrow, circulating, and lung endothelial progenitor cells in the developing lung: Implications for the pathogenesis of bronchopulmonary dysplasia. American Journal of Physiology Lung Cellular and Molecular Physiology, 292, L1073–1084.
Kalikkot Thekkeveedu, R., Guaman, M. C., & Shivanna, B. (2017). Bronchopulmonary dysplasia: A review of pathogenesis and pathophysiology. Respiratory Medicine, 132, 170–177.
Abdel Ghany, E. A., Alsharany, W., Ali, A. A., Youness, E. R., & Hussein, J. S. (2016). Anti-oxidant profiles and markers of oxidative stress in preterm neonates. Paediatrics and International Child Health, 36, 134–140.
Sun, C., Shen, H., Cai, H., Zhao, Z., Gan, G., Feng, S., Chu, P., Zeng, M., Deng, J., Ming, F., Ma, M., Jia, J., He, R., Cao, D., Chen, Z., Li, J., & Zhang, L. (2021). Intestinal guard: Human CXCL17 modulates protective response against mycotoxins and CXCL17-mimetic peptides development. Biochemical Pharmacology, 188, 114586.
Lim, G., Kim, Y. J., Chung, S., Park, Y. M., Kim, K. S., & Park, H. W. (2022). Association of maternal hypertensive disorders during pregnancy with severe bronchopulmonary dysplasia: A systematic review and Meta-analysis. Journal of Korean Medical Science, 37, e127.
Nayak, P. S., Wang, Y., Najrana, T., Priolo, L. M., Rios, M., Shaw, S. K., & Sanchez-Esteban, J. (2015). Mechanotransduction via TRPV4 regulates inflammation and differentiation in fetal mouse distal lung epithelial cells. Respiratory Research, 16, 60.
Wang, M., Tang, S., Yang, X., Xie, X., Luo, Y., He, S., Li, X., & Feng, X. (2022). Identification of key genes and pathways in chronic rhinosinusitis with nasal polyps and asthma comorbidity using bioinformatics approaches. Frontiers in Immunology, 13, 941547.
Newton, D. A., Baatz, J. E., Chetta, K. E., Walker, P. W., Washington, R. O., Shary, J. R., & Wagner, C. L. (2022). Maternal vitamin D status correlates to leukocyte antigenic responses in breastfeeding infants. Nutrients , 14(6), 1266.
Cui, T. X., Maheshwer, B., Hong, J. Y., Goldsmith, A. M., Bentley, J. K., & Popova, A. P. (2016). Hyperoxic exposure of immature mice increases the inflammatory response to subsequent Rhinovirus infection: Association with Danger signals. The Journal of Immunology, 196, 4692–4705.
Alphonse, R. S., Vadivel, A., Coltan, L., Eaton, F., Barr, A. J., Dyck, J. R., & Thebaud, B. (2011). Activation of akt protects alveoli from neonatal oxygen-induced lung injury. American Journal of Respiratory cell and Molecular Biology, 44, 146–154.
Xu, D., Guthrie, J. R., Mabry, S., Sack, T. M., & Truog, W. E. (2006). Mitochondrial aldehyde dehydrogenase attenuates hyperoxia-induced cell death through activation of ERK/MAPK and PI3K-Akt pathways in lung epithelial cells. American Journal of Physiology. Lung Cellular and Molecular Physiology, 291, L966–975.
Huang, J., Zheng, L., Wang, F., Su, Y., Kong, H., & Xin, H. (2020). Mangiferin ameliorates placental oxidative stress and activates PI3K/Akt/mTOR pathway in mouse model of preeclampsia. Archives of Pharmacal Research, 43, 233–241.
Delaney, C., Sherlock, L., Fisher, S., Maltzahn, J., Wright, C., & Nozik-Grayck, E. (2018). Serotonin 2A receptor inhibition protects against the development of pulmonary hypertension and pulmonary vascular remodeling in neonatal mice. American Journal of Physiology. Lung Cellular and Molecular Physiology, 314, L871–L881.
Du, F. L., Dong, W. B., Zhang, C., Li, Q. P., Kang, L., Lei, X. P., & Zhai, X. S. (2019). Budesonide and Poractant Alfa prevent bronchopulmonary dysplasia via triggering SIRT1 signaling pathway. European Review for Medical Pharmacological Sciences, 23(24), 11032–11042.
Acknowledgements
No.
Funding
No.
Author information
Authors and Affiliations
Contributions
Conceptualization, writing with review and editing: PC; Methodology, software, and validation: YC and JH; Data analysis: RF; Supervision and conceptualization: LQY.
Corresponding author
Ethics declarations
Conflict of interest
The authors have not disclosed any competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, P., Cheng, Y., Hu, J. et al. Recombinant CXCL17 Treatment Alleviates Hyperoxia-Induced Lung Apoptosis and Inflammation In Vivo and Vitro by Activating the AKT Pathway: A Possible Therapeutic Approach for Bronchopulmonary Dysplasia. Mol Biotechnol (2023). https://doi.org/10.1007/s12033-023-00866-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12033-023-00866-0