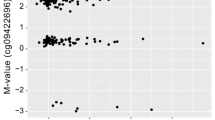
Abstract
Epigenetic dysregulation has been known to be involved in neurodegenerative diseases, including amnestic mild cognitive impairment (MCI). The aim of this study was to investigate the genome-wide DNA methylation analysis, in order to identify epigenetic dysregulation in blood from patients with MCI. Here, we investigated whether epigenetic dysregulation in MCI and whether such an aberration could be detected in blood circulation. Genome-wide bisulfite sequencing targeted 84 million bases covering 3.7 million CpG sites was comparatively analyzed in MCI and control groups. And correlation between DNA methylation and transcriptomic changes was sought. Significant differentially methylated regions (DMRs) distinguishing the MCI and control groups were identified and functionally annotated. Most DMRs specific to MCI were enriched between – 2 kb and + 2 kb of the CpG island start sites located within or near gene promoters. Representative hypo- and hypermethylated DMRs in MCI were confirmed to be correlated to mRNA expression changes with the comparative delta Ct method. DNA methylation aberrations involving metal ion homeostasis, axon growth, inflammasome, and others in this study may be less-invasive, easily measurable blood biomarker candidates for MCI.



Similar content being viewed by others
References
Ahn H-J, Chin J, Park A, Lee BH, Suh MK, Seo SW, Na DL (2010) Seoul Neuropsychological Screening Battery-Dementia Version (SNSB-D): a useful tool for assessing and monitoring cognitive impairments in dementia patients. J Korean Med Sci 25:1071–1076. https://doi.org/10.3346/jkms.2010.25.7.1071
Almen MS, Nilsson EK, Jacobsson JA, Kalnina I, Klovins J, Fredriksson R, Schioth HB (2014) Genome-wide analysis reveals DNA methylation markers that vary with both age and obesity. Gene 548:61–67. https://doi.org/10.1016/j.gene.2014.07.009
Atamna H, Killilea DW, Killilea AN, Ames BN (2002) Heme deficiency may be a factor in the mitochondrial and neuronal decay of aging. Proc Natl Acad Sci U S A 99:14807–14812. https://doi.org/10.1073/pnas.192585799
Bae JN, Cho MJ (2004) Development of the Korean version of the Geriatric Depression Scale and its short form among elderly psychiatric patients. J Psychosom Res 57:297–305. https://doi.org/10.1016/j.jpsychores.2004.01.004
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B Methodol 57:289–300
Bird TD (2008) Genetic aspects of Alzheimer disease. Genet Med 10:231–239. https://doi.org/10.1097/GIM.0b013e31816b64dc
Chen W, Paradkar PN, Li L, Pierce EL, Langer NB, Takahashi-Makise N, Hyde BB, Shirihai OS, Ward DM, Kaplan J, Paw BH (2009) Abcb10 physically interacts with mitoferrin-1 (Slc25a37) to enhance its stability and function in the erythroid mitochondria. Proc Natl Acad Sci U S A 106:16263–16268. https://doi.org/10.1073/pnas.0904519106
Chen J, Xu X (2010) Diet, epigenetic, and cancer prevention. Adv Genet 71:237–255. https://doi.org/10.1016/B978-0-12-380864-6.00008-0
Chetelat G, La Joie R, Villain N, Perrotin A, de La Sayette V, Eustache F, Vandenberghe R (2013) Amyloid imaging in cognitively normal individuals, at-risk populations and preclinical Alzheimer’s disease. Neuroimage Clin 2:356–365. https://doi.org/10.1016/j.nicl.2013.02.006
Chouliaras L, Pishva E, Haapakoski R, Zsoldos E, Mahmood A, Filippini N, Burrage J, Mill J, Kivimäki M, Lunnon K, Ebmeier KP (2018) Peripheral DNA methylation, cognitive decline and brain aging: pilot findings from the Whitehall II imaging study. Epigenomics 10:585–595. https://doi.org/10.2217/epi-2017-0132
Cisse M et al (2011) Reversing EphB2 depletion rescues cognitive functions in Alzheimer model. Nature 469:47–52. https://doi.org/10.1038/nature09635
Coppede F, Migliore L (2010) Evidence linking genetics, environment, and epigenetics to impaired DNA repair in Alzheimer’s disease. J Alzheimers Dis 20:953–966. https://doi.org/10.3233/JAD-2010-1415
Davies MN, Volta M, Pidsley R, Lunnon K, Dixit A, Lovestone S, Coarfa C, Harris RA, Milosavljevic A, Troakes C, al-Sarraj S, Dobson R, Schalkwyk LC, Mill J (2012) Functional annotation of the human brain methylome identifies tissue-specific epigenetic variation across brain and blood. Genome Biol 13:R43. https://doi.org/10.1186/gb-2012-13-6-r43
Davis CD, Milner J (2004) Frontiers in nutrigenomics, proteomics, metabolomics and cancer prevention. Mutat Res 551:51–64. https://doi.org/10.1016/j.mrfmmm.2004.01.012
Egger G, Liang G, Aparicio A, Jones PA (2004) Epigenetics in human disease and prospects for epigenetic therapy. Nature 429:457–463. https://doi.org/10.1038/nature02625
Feil R, Fraga MF (2012) Epigenetics and the environment: emerging patterns and implications. Nat Rev Genet 13:97–109. https://doi.org/10.1038/nrg3142
Geda YE, Ragossnig M, Roberts LA, Roberts RO, Pankratz VS, Christianson TJH, Mielke MM, Levine JA, Boeve BF, Sochor O, Tangalos EG, Knopman DS, Petersen RC (2013) Caloric intake, aging, and mild cognitive impairment: a population-based study. J Alzheimers Dis 34:501–507. https://doi.org/10.3233/JAD-121270
Hannon E, Lunnon K, Schalkwyk L, Mill J (2015) Interindividual methylomic variation across blood, cortex, and cerebellum: implications for epigenetic studies of neurological and neuropsychiatric phenotypes. Epigenetics 10:1024–1032. https://doi.org/10.1080/15592294.2015.1100786
Heneka MT, Kummer MP, Stutz A, Delekate A, Schwartz S, Vieira-Saecker A, Griep A, Axt D, Remus A, Tzeng TC, Gelpi E, Halle A, Korte M, Latz E, Golenbock DT (2013) NLRP3 is activated in Alzheimer’s disease and contributes to pathology in APP/PS1 mice. Nature 493:674–678. https://doi.org/10.1038/nature11729
Jimenez-Chillaron JC, Diaz R, Martinez D, Pentinat T, Ramon-Krauel M, Ribo S, Plosch T (2012) The role of nutrition on epigenetic modifications and their implications on health. Biochimie 94:2242–2263. https://doi.org/10.1016/j.biochi.2012.06.012
Jones PA, Baylin SB (2002) The fundamental role of epigenetic events in cancer. Nat Rev Genet 3:415–428. https://doi.org/10.1038/nrg816
Kang Y, Na DL, Seung H (1997) A validity study on the Korean Mini-Mental State Examination (K-MMSE) in dementia patients. J Korean Neurol Assoc 15:300–308
Kobayashi N, Shinagawa S, Nagata T, Shimada K, Shibata N, Ohnuma T, Kasanuki K, Arai H, Yamada H, Nakayama K, Kondo K (2016) Usefulness of DNA methylation levels in COASY and SPINT1 gene promoter regions as biomarkers in diagnosis of Alzheimer’s disease and amnestic mild cognitive impairment. PLoS One 11:e0168816. https://doi.org/10.1371/journal.pone.0168816
Krueger F, Andrews SR (2011) Bismark: a flexible aligner and methylation caller for Bisulfite-Seq applications. Bioinformatics 27:1571–1572. https://doi.org/10.1093/bioinformatics/btr167
Ku HM et al (2004) A study on the reliability and validity of Seoul-Instrumental Activities of Daily Living(S-IADL). J Korean Neuropsychiatr Assoc 43:11
Liesa M, Qiu W, Shirihai OS (2012) Mitochondrial ABC transporters function: the role of ABCB10 (ABC-me) as a novel player in cellular handling of reactive oxygen species. Biochim Biophys Acta 1823:1945–1957. https://doi.org/10.1016/j.bbamcr.2012.07.013
Lunnon K, Smith RG, Cooper I, Greenbaum L, Mill J, Beeri MS (2015) Blood methylomic signatures of presymptomatic dementia in elderly subjects with type 2 diabetes mellitus. Neurobiol Aging 36:1600.e1-4. https://doi.org/10.1016/j.neurobiolaging.2014.12.023
Morris JC (1993) The Clinical Dementia Rating (CDR): current version and scoring rules. Neurology 43:2412–2414
Petersen RC, Smith GE, Waring SC, Ivnik RJ, Tangalos EG, Kokmen E (1999) Mild cognitive impairment: clinical characterization and outcome. Arch Neurol 56:303–308
Pietrzak M, Rempala GA, Nelson PT, Hetman M (2016) Non-random distribution of methyl-CpG sites and non-CpG methylation in the human rDNA promoter identified by next generation bisulfite sequencing. Gene 585:35–43. https://doi.org/10.1016/j.gene.2016.03.028
Rama N, Goldschneider D, Corset V, Lambert J, Pays L, Mehlen P (2012) Amyloid precursor protein regulates netrin-1-mediated commissural axon outgrowth. J Biol Chem 287:30014–30023. https://doi.org/10.1074/jbc.M111.324780
Rao JS, Keleshian VL, Klein S, Rapoport SI (2012) Epigenetic modifications in frontal cortex from Alzheimer’s disease and bipolar disorder patients. Transl Psychiatry 2:e132. https://doi.org/10.1038/tp.2012.55
Saresella M, la Rosa F, Piancone F, Zoppis M, Marventano I, Calabrese E, Rainone V, Nemni R, Mancuso R, Clerici M (2016) The NLRP3 and NLRP1 inflammasomes are activated in Alzheimer’s disease. Mol Neurodegener 11:23. https://doi.org/10.1186/s13024-016-0088-1
Simon AM et al (2009) Early changes in hippocampal Eph receptors precede the onset of memory decline in mouse models of Alzheimer’s disease. J Alzheimers Dis 17:773–786. https://doi.org/10.3233/JAD-2009-1096
Snitz BE, Weissfeld LA, Lopez OL, Kuller LH, Saxton J, Singhabahu DM, Klunk WE, Mathis CA, Price JC, Ives DG, Cohen AD, McDade E, DeKosky ST (2013) Cognitive trajectories associated with beta-amyloid deposition in the oldest-old without dementia. Neurology 80:1378–1384. https://doi.org/10.1212/WNL.0b013e31828c2fc8
Spilman PR, Corset V, Gorostiza O, Poksay KS, Galvan V, Zhang J, Rao R, Peters-Libeu C, Vincelette J, McGeehan A, Dvorak-Ewell M, Beyer J, Campagna J, Bankiewicz K, Mehlen P, John V, Bredesen DE (2016) Netrin-1 interrupts amyloid-beta amplification, increases sAbetaPPalpha in vitro and in vivo, and improves cognition in a mouse model of Alzheimer’s disease. J Alzheimers Dis 52:223–242. https://doi.org/10.3233/JAD-151046
Sung HY, Choi BO, Jeong JH, Kong KA, Hwang J, Ahn JH (2016) Amyloid beta-mediated hypomethylation of heme oxygenase 1 correlates with cognitive impairment in Alzheimer’s disease. PLoS One 11:e0153156. https://doi.org/10.1371/journal.pone.0153156
Umar A, Dunn BK, Greenwald P (2012) Future directions in cancer prevention. Nat Rev Cancer 12:835–848. https://doi.org/10.1038/nrc3397
Van Battum EY, Brignani S, Pasterkamp RJ (2015) Axon guidance proteins in neurological disorders. Lancet Neurol 14:532–546. https://doi.org/10.1016/S1474-4422(14)70257-1
Wen KX, Miliç J, el-Khodor B, Dhana K, Nano J, Pulido T, Kraja B, Zaciragic A, Bramer WM, Troup J, Chowdhury R, Ikram MA, Dehghan A, Muka T, Franco OH (2016) The role of DNA methylation and histone modifications in neurodegenerative diseases: a systematic review. PLoS One 11:e0167201. https://doi.org/10.1371/journal.pone.0167201
Acknowledgements
The authors give special thanks to S.Y. Han and S. J. Song for assistance in the management of subjects.
Funding
This study was supported by Small and Medium Business Administration of South Korea (Grant No. S2261274).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflicts of interest.
Electronic Supplementary Material
ESM 1
(DOCX 34 kb)
Rights and permissions
About this article
Cite this article
Park, MK., Lee, JW., Lee, JC. et al. NLRP1 and NTN1, Deregulated Blood Differentially Methylated Regions in Mild Cognitive Impairment Patients. J Mol Neurosci 66, 561–571 (2018). https://doi.org/10.1007/s12031-018-1180-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-018-1180-5